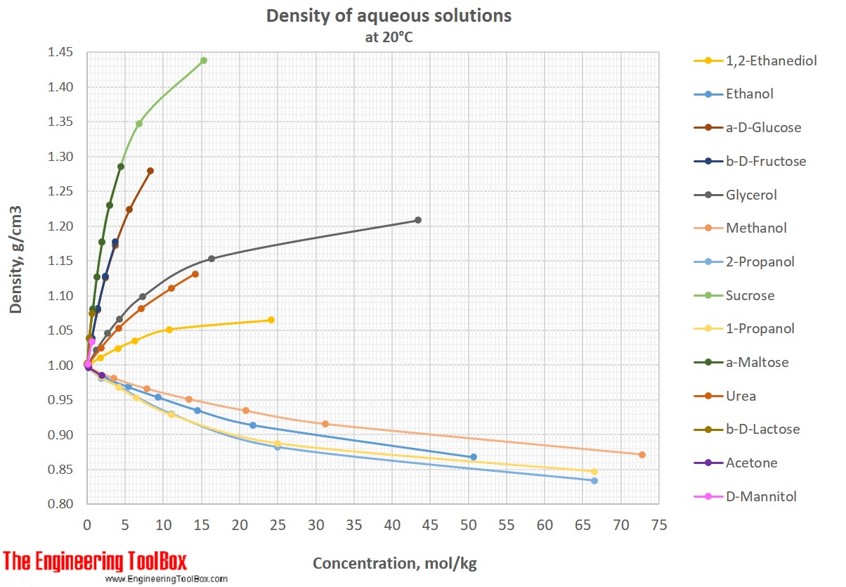
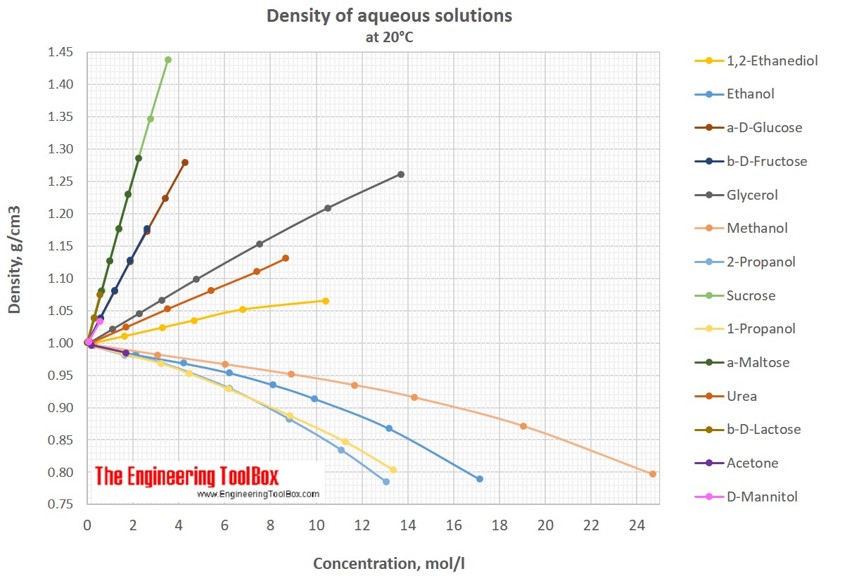
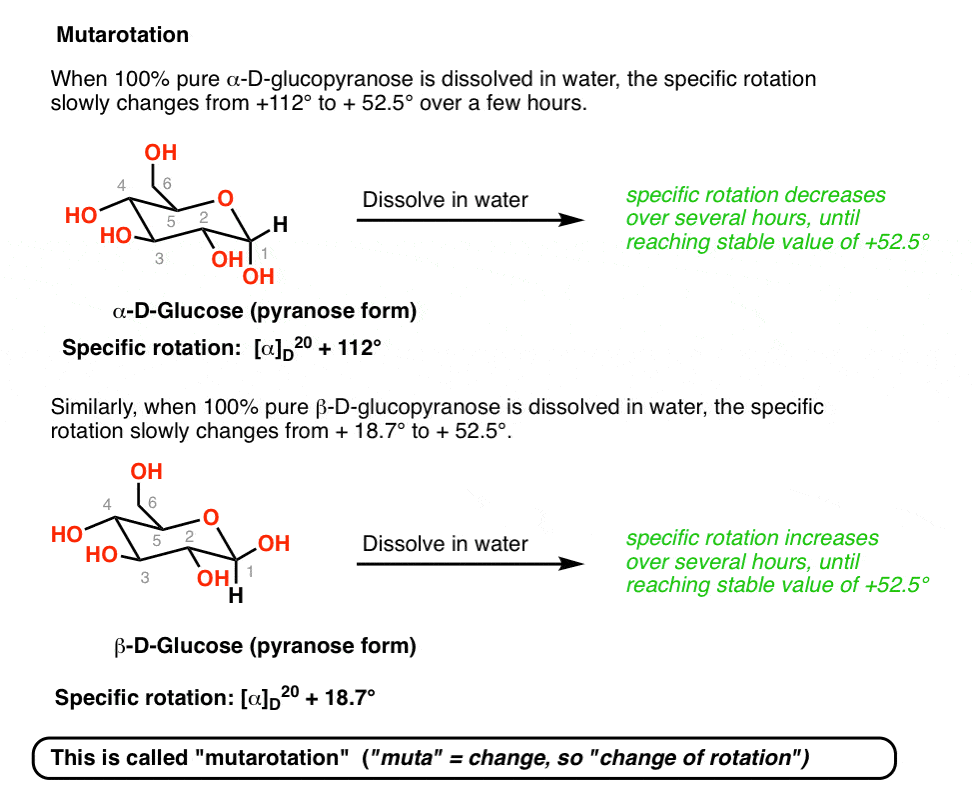
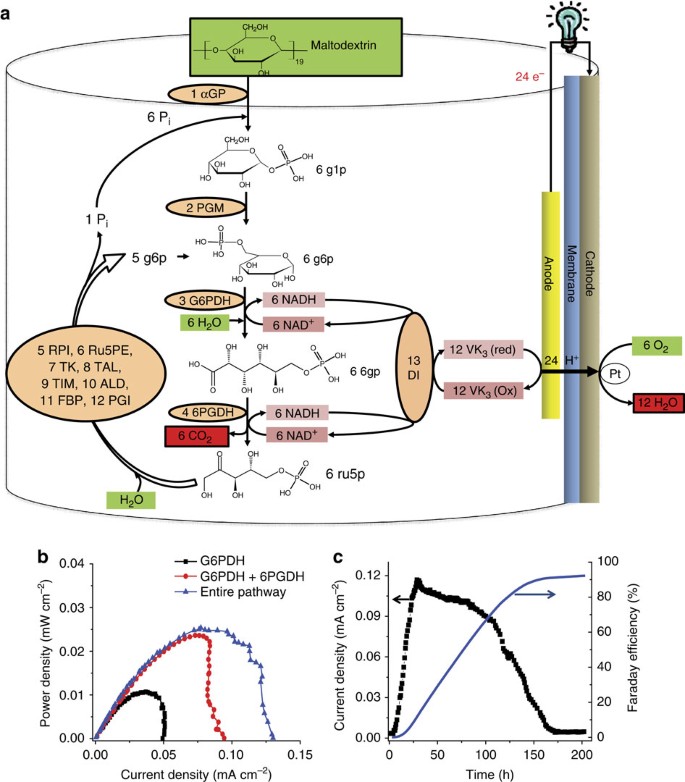
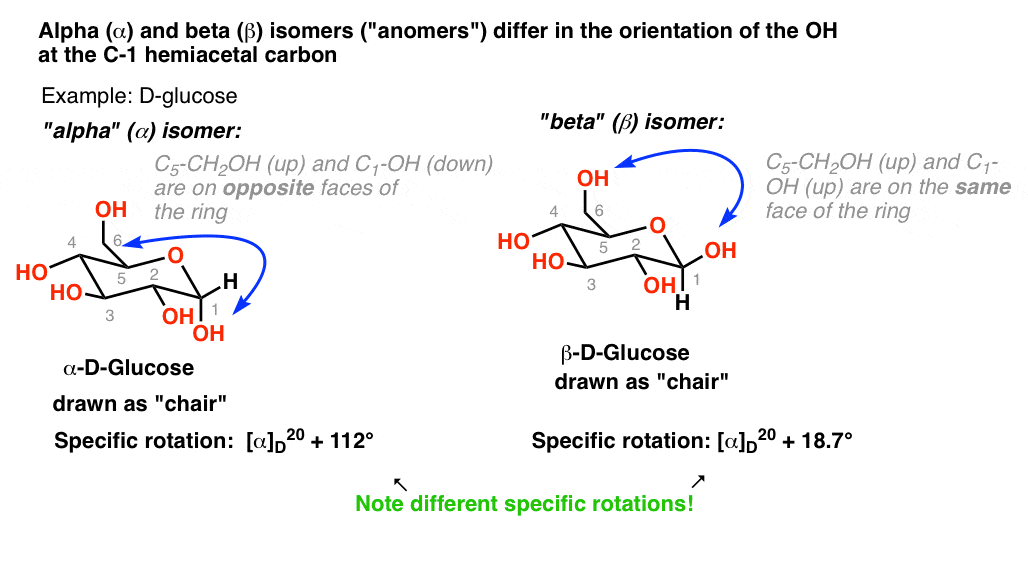
density of glucose in water
How much does glucose weigh per cubic meter?
Glucose weighs 1.56 gram per cubic centimeter or 1 560 kilogram per cubic meter, i.e. density of glucose is equal to 1 560 kg/m³. In Imperial or US customary measurement system, the density is equal to 97.388 pound per cubic foot [lb/ft³], or 0.9017 ounce per cubic inch [oz/inch³] .
Is glucose soluble in water?
It is, however, soluble in nonpolar solvents that have comparable London dispersion forces, such as CS 2 (23 g/100 mL). In contrast, glucose contains five –OH groups that can form hydrogen bonds. Consequently, glucose is very soluble in water (91 g/120 mL of water) but essentially insoluble in nonpolar solvents such as benzene.
What is the density of glucose?
In Imperial or US customary measurement system, the density is equal to 97.388 pound per cubic foot [lb/ft³], or 0.9017 ounce per cubic inch [oz/inch³] . Melting Point (MP), Glucose changes its state from solid to liquid at 146°C (294.8°F or 419.15K) Also known as: Grape sugar.
What does density mean in chemistry?
An introduction to density, specific weight and specific gravity. Physical constants for more than 280 common inorganic compounds. Density is given for the actual state at 25°C and for liquid phase at melting point temperature. Guidelines or solubility rules to predict whether or not a given ionic compound is soluble in water at room temperature.
|
Temperature and Concentration Dependence of Density of Model
10%-fructose designated as A) |
|
D -(+)-Glucose (G8270) - Product Information Sheet
Density (g/ml) 1.019 1.038 1.076 1.113 1.149 Not for drug household or other uses. Preparation Instructions. One gram of glucose dissolves in 1.1 ml of water. |
|
DENSITY OF INTRATHECAL AGENTS
which relates density to water and not CSF and to Solutions of opioid drugs mixed with 5 % glucose varied more in their densities than the same drugs in. |
|
Untitled
The freezing point of a glucose solution (C6H12O6;molar mass= 180.0 g/mole) is 10.3 °C. The density of the solution is 1.50 g/ml. What is the molarity of |
|
Densities and Viscosities of Ternary Systems of Water + Glucose +
The density and dynamic viscosity of aqueous solutions of sodium chloride glucose |
|
1 DETERMINING THE CONCENTRATION OF SUGAR(S) IN A SOFT
The length of the stem that emerges from the liquid is a measure of the density of the liquid. A hydrometer is placed in water and then in a number of carefully |
|
Solubility of fructose in water-ethanol and water-methanol mixtures
They considered 13 sugars and obtained 4 adjustable parameters from solution density and osmotic coefficient of binary sugar-water solutions at. 25°C. They |
|
BRIX - Sugar Determination By Density And Refractometry And
What is Brix ? Brix is the measurement in percentage by weight of sucrose in pure water solution. This designation of Brix degrees is only valid for pure |
|
Effects of Potassium Ions on the Viscosities in the Potassium
The viscosity of potassium chloride-glucose-water ternary system with varying concentrations was investigated in this study. The density of the ternary |
|
Beverage Density Lab
Table sugar is sucrose a disaccharide composed of one molecule of fructose joined with one molecule of glucose after loss of water. Both fructose and sucrose |
|
Temperature and Concentration Dependence of Density of Model
Density of binary solutions and combinations of sucrose glucose |
|
D -(+)-Glucose (G8270) - Product Information Sheet
respect to water:2. Concentration (%) 5 10 20 30 40. Density (g/ml) 1.019 1.038 1.076 1.113 1.149. Glucose is a main source of energy for living organisms. |
|
Sugar Water Density Columns
Sugar Water Density Columns. Standard: 5-PS1-3. Make observations and measurements to identify materials based on their properties. Introduction:. |
|
1 DETERMINING THE CONCENTRATION OF SUGAR(S) IN A SOFT
ON THE BASIS OF DENSITY USING A HYDROMETER. Overview. If a substance such as sugar is dissolved in water the density of the resulting. |
|
BRIX - Sugar Determination By Density and Refractometry
This sugar concentration can be measured with density meters and Brix is the measurement in percentage by weight of sucrose in pure water solution. |
|
Appendix 1: Data on Engineering Properties of Materials Used and
5 Density of saturated sugar solutions as a function of tempera- ture (Antokolskaia 1964 ) . T ( ° C). Sugar content (g/100 cm 3 water). Density (kg/m 3). |
|
Beverage Density Lab
Table sugar is sucrose a disaccharide composed of one molecule of fructose joined with one molecule of glucose after loss of water. Both fructose and sucrose |
|
How many grams of glucose would be dissolved to make 1 liter of a
What is the concentration of the 0.5M glucose solution expressed in %?. 0.5 mole/liter x 180 grams/mole x 1 liter/1000ml = 0.09 g/ml = 9 g/100ml = 9%. |
|
Refractive indices and densities of aqueous solutions of invert sugar
) in distilled water. A suitable quantity of dry D-fructose was placed in a tared glass-stoppered flask and the flask and. |
|
Sample Exercise 13.1 Predicting Solubility Patterns
Calculate the molality of glucose in the solution. Water has a density of 1.00 g/mL. Plan We use the molar mass of C6H12O6 to convert grams to moles. We use |
|
Density Calculation of Sugar Solutions with the SAFT Model
of glucose/water and fructose/water have been modeled Introduction In the food technology and sugar industries, the density of sugar solutions is an important |
|
D -(+)-Glucose (G8270) - Product Information Sheet - Sigma-Aldrich
Densities of aqueous solutions (w/v) at 17 5 °C with respect to water:2 Concentration ( ) 5 10 20 30 40 Density (g/ml) 1 019 1 038 1 076 1 113 1 149 Glucose |
|
1 DETERMINING THE CONCENTRATION OF SUGAR(S) IN A SOFT
measure of the density of the liquid A hydrometer is placed in water and then in a number of carefully prepared sugar solutions of different known concentrations |
|
1 Objective: The mass percent of sugar present in - UCA Faculty
comparing their measured densities to a set of standard sugar-water solutions The density of a solution depends on it part on concentration, which is defined |