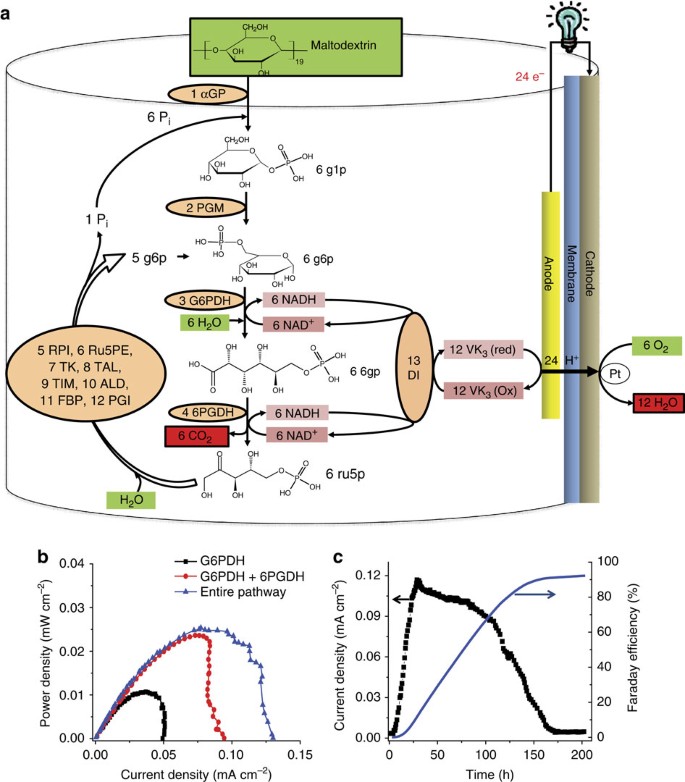
density of glucose solution in kg/m3
|
Glucose solution (G8644)
Molecular Formula: C6H12O6 Molecular Weight: 180 2 pH: 3-4 (25 C) The density of an aqueous 10 solution (w/v) at 17 5 C is 1 038 g/mL 2 This product is a 10 (w/v) solution of glucose Based on a 10 (w/v) concentration and a molecular weight for glucose of 180 Da the solution is 0 56 M glucose |
|
D -(+)-Glucose (G8270)
Molecular Formula: C6H12O6 CAS Number: 50-99-7 Molecular Weight: 180 2 Melting Point : 146 °C for α-D-glucose and 150 °C for β-D-glucose1 pH: 5 9 (0 5 M aqueous)2 Densities of aqueous solutions (w/v) at 17 5 °C with respect to water:2 Concentration ( ) 5 10 20 30 40 Density (g/ml) 1 019 1 038 1 076 1 113 1 149 |
How much glucose is in a D5W solution?
The so-called D5W solution used for the intravenous replacement of body fluids contains 0.310 M glucose. (D5W is an approximately 5% solution of dextrose [the medical name for glucose] in water.) Calculate the mass of glucose necessary to prepare a 500 mL pouch of D5W. Glucose has a molar mass of 180.16 g/mol.
What is the density of sucrose and fructose?
Density of Sugar Solutions. Empirical measurement of the density (g/ml) of fructose, glucose, and sucrose solutions. Note that sucrose is a disaccharide with almost 2x the MW of fructose and glucose; thus 1 M fructose = 180 g/L, while 1 M sucrose = 342 g/L. Mixed in house distilled water, brought to 1 Liter in volumetric flask.
What is a 10 w/v solution of glucose?
This product is a 10% (w/v) solution of glucose. Based on a 10% (w/v) concentration and a molecular weight for glucose of 180 Da, the solution is 0.56 M glucose. This product has been tested with cell lines to verify the product is not cytotoxic.
How do you calculate molar mass of glucose?
Glucose has a molar mass of 180.16 g/mol. A Calculate the number of moles of glucose contained in the specified volume of solution by multiplying the volume of the solution by its molarity. B Obtain the mass of glucose needed by multiplying the number of moles of the compound by its molar mass.
|
Data on Engineering Properties of Materials Used and Made by the
Specific heat capacity of aqueous sugar solutions (Sokolovsky 1958 |
|
Appendix 1: Data on Engineering Properties of Materials Used and
5 Density of saturated sugar solutions as a function of tempera- ture (Antokolskaia 1964 ) . T ( ° C). Sugar content (g/100 cm 3 water). Density (kg/m 3). |
|
BRIX - Sugar Determination By Density and Refractometry
the pure sucrose content in a solution is meant. This sugar concentration can be measured with density meters and ... d = m/V [kg/m3] or [g/cm3]. |
|
1) Find the molarity of all ions in a solution that contains 0.165 moles
6)The freezing point of a glucose solution (C6H12O6;molar mass= 180.0 g/mole) is - 10.3 °C. The density of the solution is 1.50 g/ml. |
|
List of Tables Table A.1 Density and Baume of Pure Sucrose
Table A.2 Sucrose Solubility in Pure Sucrose Solutions Density. Baume. DS. Density. Baume. (% m/m) (kg/m3) (lb/ft3) (lb/ga) (degree) % (m/m) (kg/m3) ... |
|
Measurement of an Analyte Concentration in Test Solution by Using
Mar 5 2019 20 ?C are 998 kg/m3 and 1540 kg/m3 |
|
Some important physical properties of foods
Example 9 : The density of sugar solution prepared by dissolving 43 kg sucrose in 100 kg water is 1127 kg/m3. Calculate the following. |
|
Sample Exercise 13.1 Predicting Solubility Patterns
(a) A solution is made by dissolving 13.5 g of glucose (C6H12O6) in 0.100 kg of water. Sample Exercise 13.6 Calculation of Molarity Using the Density of. |
|
Properties of some particular solutions
Solids dissolved in water: the salt-water and the sugar-water systems are chosen Its density is in the range ?=1005..1035 kg/m3 |
|
There are a number of different ways of expressing solute
Normality N = equivalents of solute/liter of solution Given that the density of dilute aqueous solutions is ~1.00 kg/L |
|
D -(+)-Glucose (G8270) - Product Information Sheet - Sigma-Aldrich
Densities of aqueous solutions (w/v) at 17 5 °C with respect to water:2 Concentration ( ) 5 10 20 30 40 Density (g/ml) 1 019 1 038 1 076 1 113 1 149 Glucose |
|
Density Calculation of Sugar Solutions with the SAFT Model
of glucose/water and fructose/water have been modeled Introduction In the food technology and sugar industries, the density of sugar solutions is an important |
|
1 DETERMINING THE CONCENTRATION OF SUGAR(S) IN A SOFT
ON THE BASIS OF DENSITY USING A HYDROMETER Overview If a substance such as sugar is dissolved in water, the density of the resulting solution is |
|
How many grams of glucose would be dissolved to make - De Anza
What is the concentration of the 0 5M glucose solution expressed in ? 0 5 mole /liter x 180 grams/mole x 1 liter/1000ml = 0 09 g/ml = 9 g/100ml = 9 |
















![High-cell-density cultivation of Escherichia coli - [PDF Document] High-cell-density cultivation of Escherichia coli - [PDF Document]](https://online.fliphtml5.com/gpyl/bbal/files/large/1.jpg)