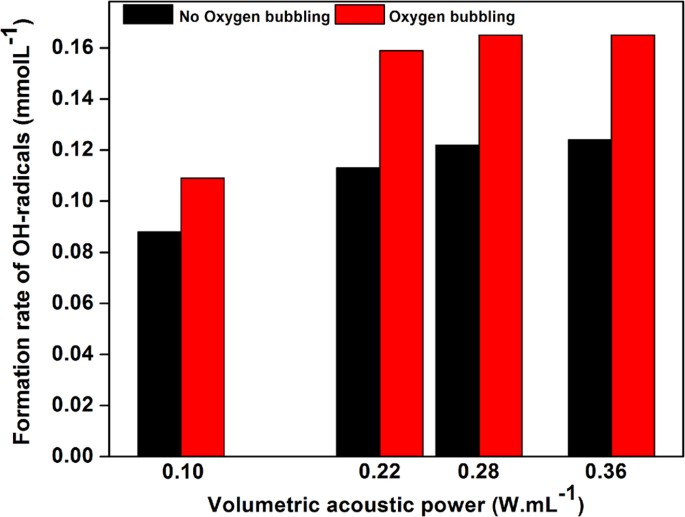
density of glucose solutions table
|
D -(+)-Glucose (G8270)
Molecular Formula: C6H12O6 CAS Number: 50-99-7 Molecular Weight: 180 2 Melting Point : 146 °C for α-D-glucose and 150 °C for β-D-glucose1 pH: 5 9 (0 5 M aqueous)2 Densities of aqueous solutions (w/v) at 17 5 °C with respect to water:2 Concentration ( ) 5 10 20 30 40 Density (g/ml) 1 019 1 038 1 076 1 113 1 149 |
What are the aqueous glycine concentrations of glucose and raffinose?
Density, ρ, viscosity, η, and refractive index, nD of glucose, sucrose, and raffinose have been measured in 0.015 m aqueous glycine at 298.15, 303.15, 308.15, and 313.15 K.
How many grams are in a cup of sugar?
The mass of sugar is 4.00 cups x 200 g/cup = 800 g, and the mass of water is 5.00 cups x 226 g/cup = 1130 g or 1.13 x 10 3 g. So the total mass is 800 g + 1130 g = 1930 g or 1.93 x 10 3 g. The fruit has a density of 1.045 g/ml, while the syrup has a density of 1.15 g/ml, so the fruit will float, being less dense.
How do you estimate the density of a liquid solution?
It is possible to estimate the density of a liquid-liquid solution from the density of the solute and the solvent. However, due to shrinkage, the estimate will be a bit too low. Densities of common liquids like acetone, beer, oil, water and more. Densities and specific volume of liquids vs. pressure and temperature change.
What are RD values in aqueous glycine?
The values of RD are given in Table 8. It indicates that the values of all the three saccharides slightly increase with an increase in the concentration of saccharides. The trend in nD and RD values of the saccharides in aqueous glycine is: glucose < sucrose < raffinose.
|
Refractive indices and densities of aqueous solutions of invert sugar
Five-decimal tables giving therefractive indices and densities of invert sugar solutions containing equal weights of pure D-glucose and D-fructose for. |
|
Temperature and Concentration Dependence of Density of Model
Density of binary solutions and combinations of sucrose glucose |
|
Density Calculation of Sugar Solutions with the SAFT Model
Results with high accuracy have been obtained. Finally the water activity of sorbitol/water and xylitol/water and the normal boiling temperature of glucose/ |
|
Nanofiltration of glucose and sodium lactate solutions: Variations of
27 sept. 2018 solutions. Glu / NaCl. 0.1 M / 0.5 M - 0.1 M / 1 M. 25. Page 27. Table 3: Mean pore radius (rp) and membrane charge density (Xd) obtained from ... |
|
Diffusion in Supersaturated Solutions. II. Glucose Solutions
from standard tables.11 Table I. Diffusion Coefficients of Glucose Solutions. Wt. per cent. ... where p is the density of the solute N the Avogadro. |
|
Density-dependent negative responses by bumblebees to bacteria
27 nov. 2015 glucose solution was negatively correlated with the bacterial density. ... (Table I) a density that has been found in. |
|
Appendix 1: Data on Engineering Properties of Materials Used and
Table A1.5 Density of saturated sugar solutions as a function of tempera- Table A1.14 Boiling point of aqueous glucose syrup solutions at atmos-. |
|
Viscosity of Aqueous Carbohydrate Solutions at Different
[1] presented kinematic viscosity data against molality of sucrose and glucose solutions respectively. Although data were presented in table form |
|
Corn Refiners Association
1 oct. 2018 Data for Density of Dextrose Solutions and Comparison with Interpolation Formula . ... Equivalents Tables for Dextrose/Glucose Solutions . |
|
BRIX - Sugar Determination By Density and Refractometry
There are a lot of different sugars e.g. sucrose |
|
Density Calculation of Sugar Solutions with the SAFT Model
Density is one of the key physicochemical properties for sugar solutions The objective In Table 3, for the solutions of sorbitol/sucrose/water, the results of the |
|
D -(+)-Glucose (G8270) - Product Information Sheet - Sigma-Aldrich
Densities of aqueous solutions (w/v) at 17 5 °C with respect to water:2 Concentration ( ) 5 10 20 30 40 Density (g/ml) 1 019 1 038 1 076 1 113 1 149 Glucose |
|
Refractive indices and densities of aqueous solutions of invert sugar
the sucrose table) gave results which were too low From iais experimental results, de Whalley calcu- lated an average correction of 0 022 for each percent of |
|
1 DETERMINING THE CONCENTRATION OF SUGAR(S) IN A SOFT
Density of a solution can be measured with a device called a hydrometer, a long, Solutions of sucrose (table sugar) in water in concentrations of 3 , 6 , 9 , |































:max_bytes(150000):strip_icc()/guidelines-for-high-cholesterol-and-diabetes-697813-color1b-99ed648078d74592ac667c8533065003.jpg)