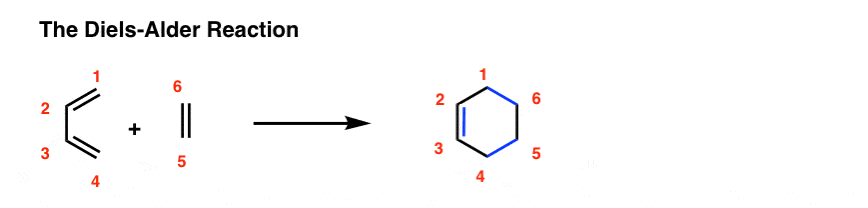
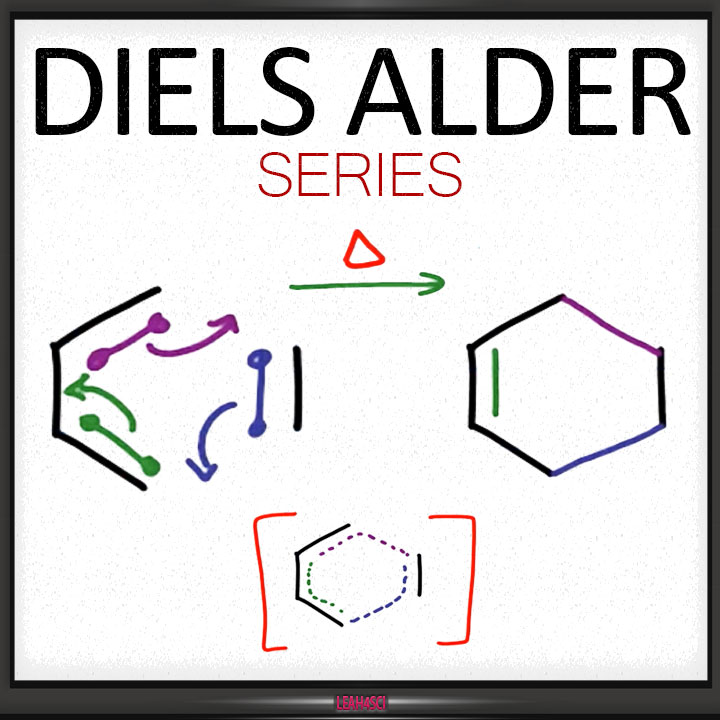
diels alder reaction balanced equation
Why is the Diels-Alder reaction faster?
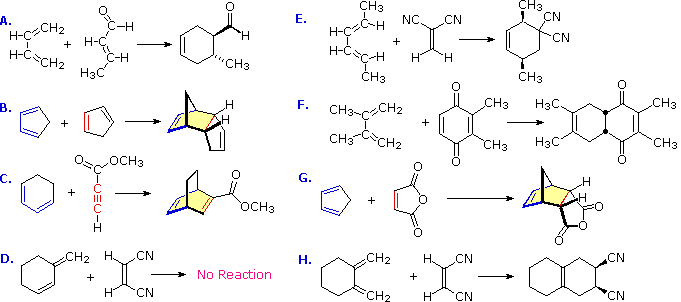
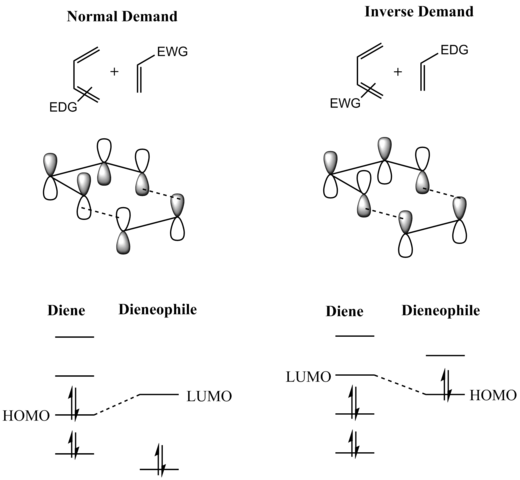
The Diels-Alder reaction is faster when there is an electron-withdrawing group on the pi bond (“dienophile”) and electron donating groups on the diene. Don’t Be Underwhelmed! Four Key Things, Part 3: Substituents On The Diene Or Dienophile Do NOT Affect The Bond-Forming / Bond-Breaking Pattern Of The Diels-Alder Four Key Things, Part 4.
Does 1,3-butadiene undergo a Diels-Alder reaction?
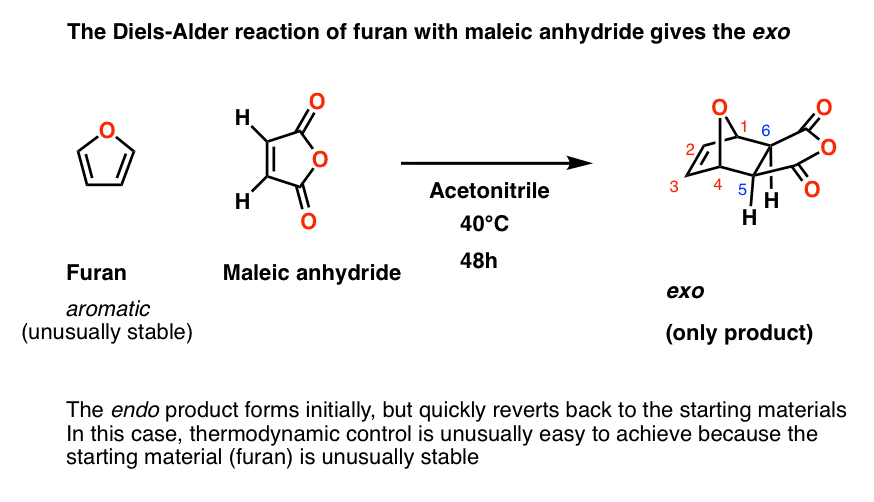
The diene must be conjugated to participate in a Diels-Alder reaction. No conjugation, no Diels-Alder. So while 1,3-butadiene readily undergoes the Diels-Alder reaction, 1,4 pentadiene (below) does not. (If you’re not clear about what constitutes a “conjugated” diene, you might want to visit this post .) 8.
What is an example of a Diels Alder reaction?
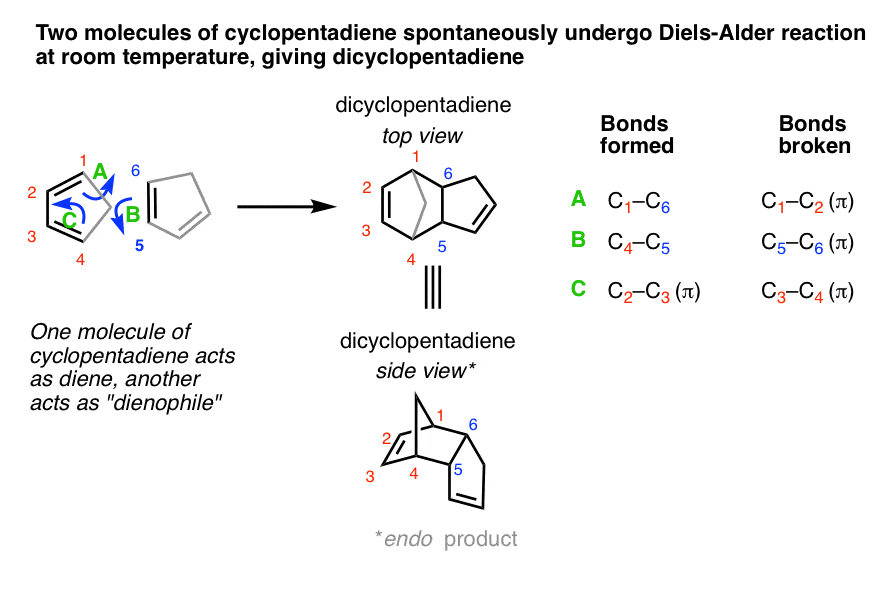
One example occurs in the biosynthesis of the cholesterol-lowering drug lovastatin (trade name Mevacor) isolated from the bacterium Aspergillus terreus. The key step is the intramolecular Diels–Alder reaction of a triene, in which the diene and dienophile components are within the same molecule.
How can a diene predict a Diels-Alder reaction?
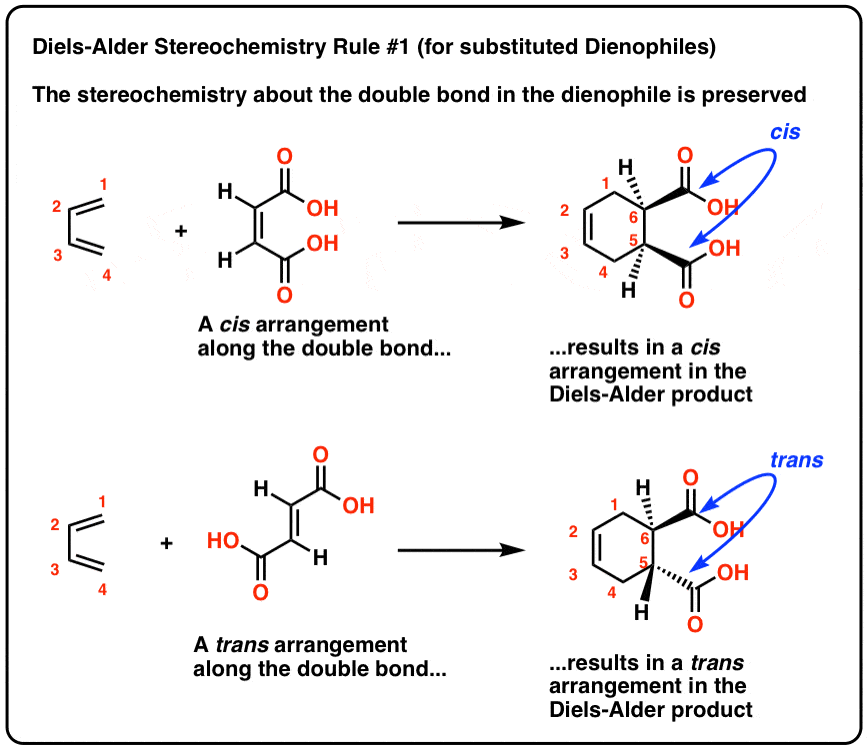
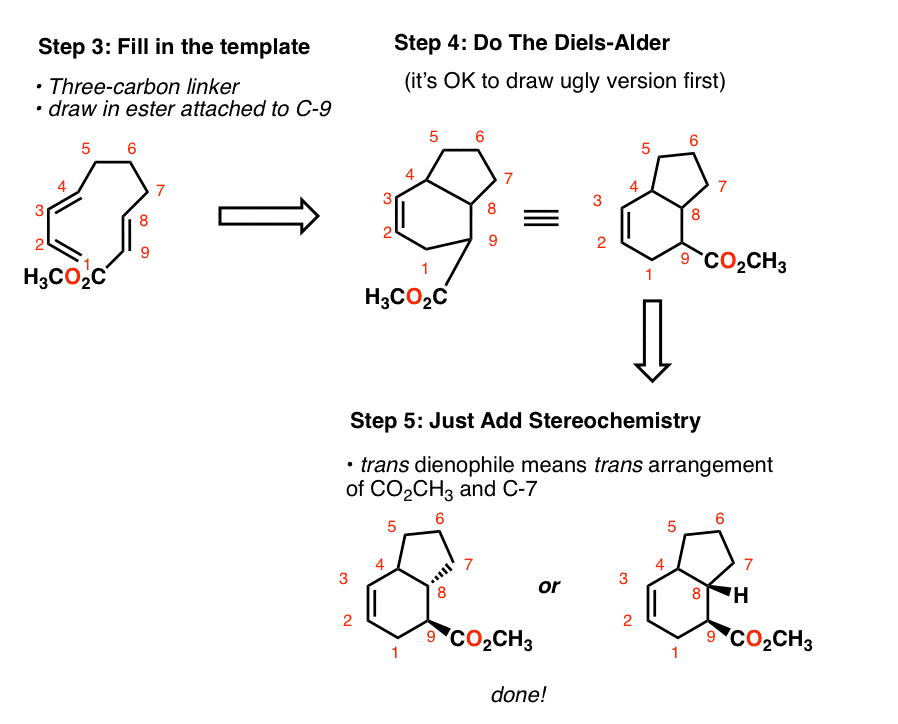
predict the stereochemistry of the product obtained from the reaction of a given diene with a given dienophile. recognize that in order to undergo a Diels-Alder reaction, a diene must be able to assume an s -cis geometry, and determine whether or not a given diene can assume this geometry.

Diels-Alder reaction Organic chemistry Khan Academy

Diels Alder Reaction

Diels-Alder Reaction Organic Chemistry Lessons
|
DIELS-ALDER REACTION OF 13-BUTADIENE AND MALEIC
FIGURE 2 The Diels-Alder reaction between 13-butadiene and maleic anhydride to produce 4- cyclohexene-cis-1 |
|
Tandem Diels–Alder Reaction of Dimethylfuran and Ethylene and
Although it has been shown that these Lewis acids can catalyze the dehydration of the. Diels-Alder cycloadduct the tandem scheme has precluded decoupling of |
|
Diels-Alder Reactions
Diels-Alder Reactions. Paul Schatz and Charles G. Fry. Classic reaction of phellandrene with maleic anhydride is a lab unit in Chem 346:. |
|
Synthesis of 4-Cyclohexene-cis-dicarboxylic Acid Anhydride - (Diels
Figure 1: The overall equation. The Diels-Alder reaction is a member of a larger class of pericyclic reactions. These are reactions with transition states in |
|
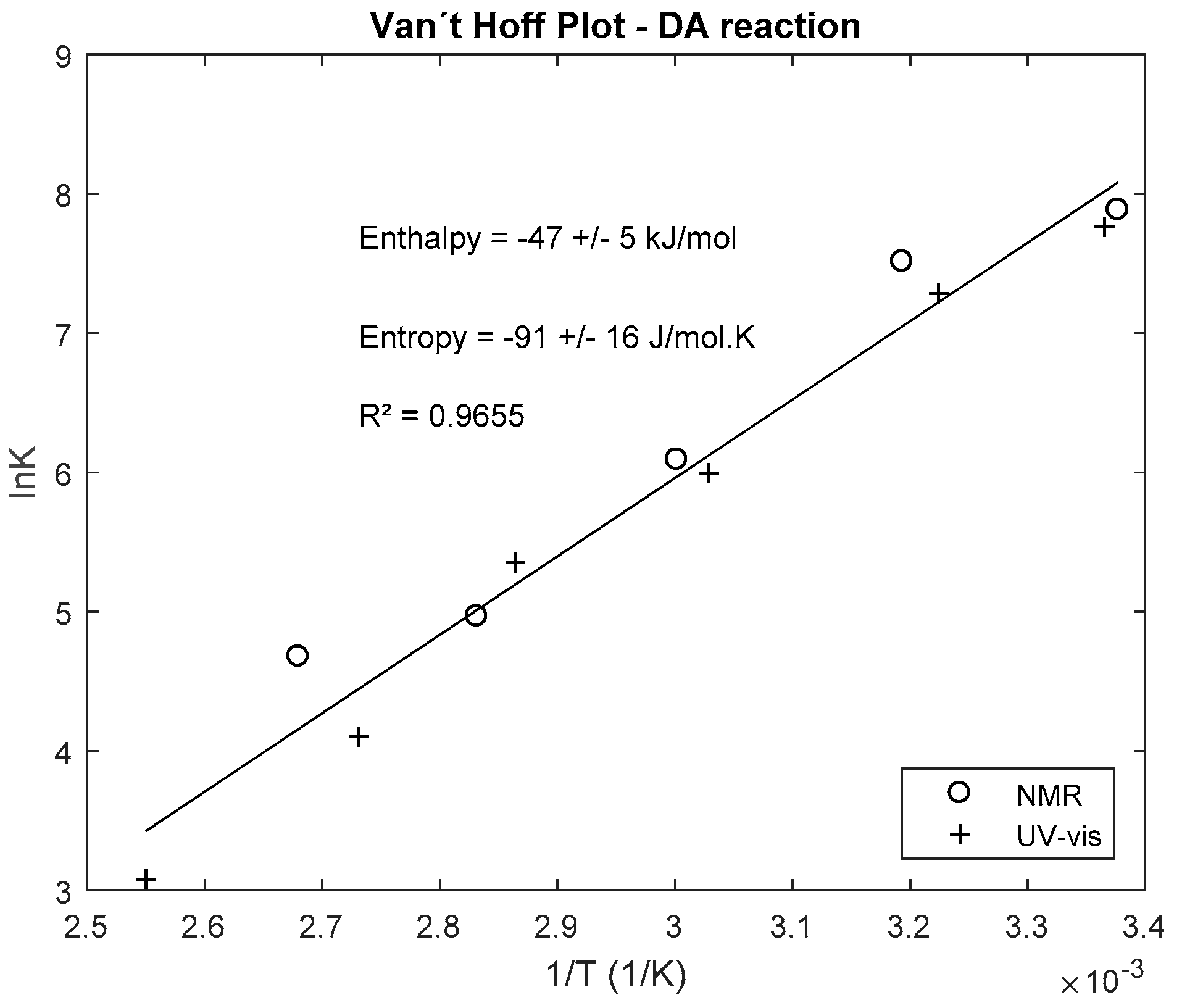
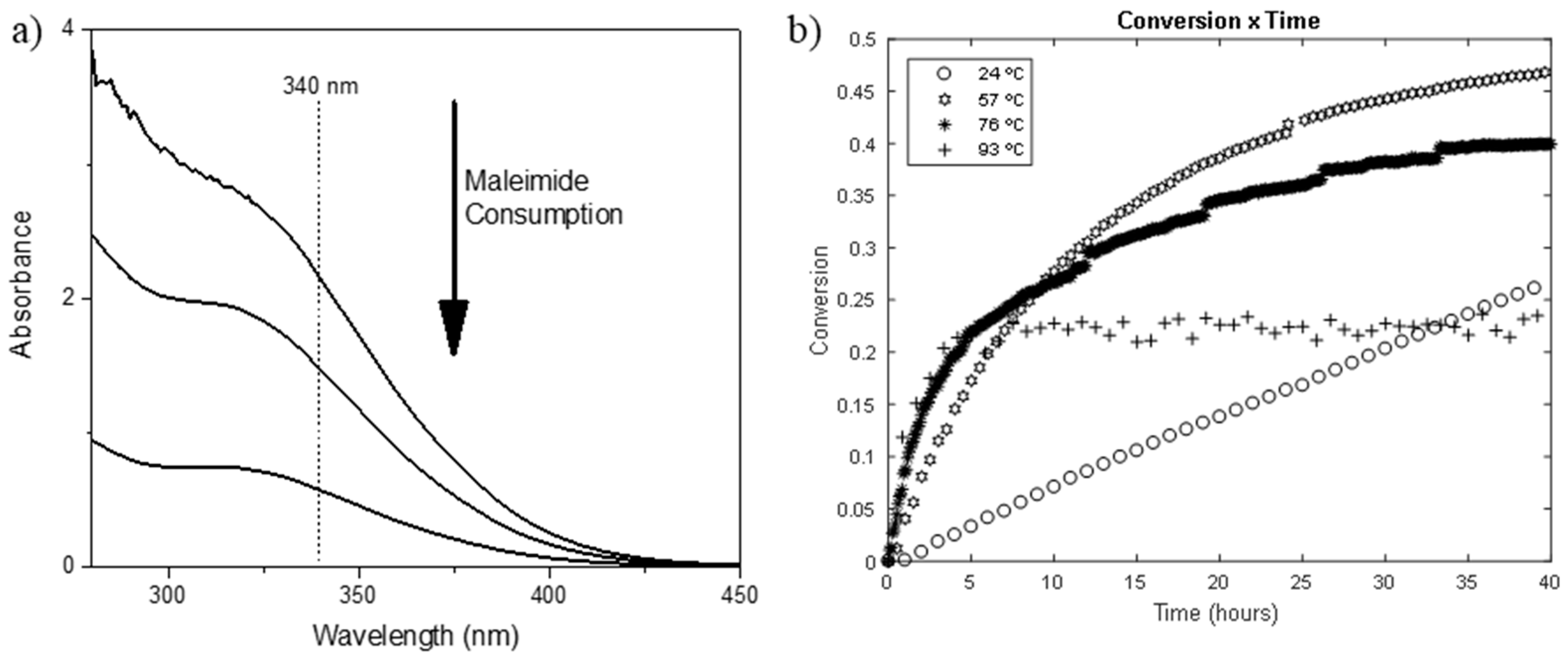
Thermodynamic and Kinetic Study of Diels–Alder Reaction between
7 janv. 2020 Abstract: The study of Diels–Alder reactions in materials science is ... for writing the rate equation as shown above is that there is no ... |
|
Indian National Chemistry Olympiad Theory 2012 - Instructions for
Diels-Alder reaction is an example of [4+2] cycloaddition where 4 and 2 indicate the Write the balanced equations of the reactions involved in. |
|
Fundamentals of Chemical Reaction Engineering
6 mars 1999 stoichiometric equation for a simple reaction can be written as: ... The Diels-Alder reaction of 23-dimethyl-1 |
|
Development of Tubular Chemical Reactor Models for Control
resented by a system of partial differential equations Reaction gas heat balance equation ... serve some pericyclic reactions (the Diels—Alder re-. |
|
Indian National Chemistry Olympiad Theory 2011 - Instructions for
29 janv. 2011 Write balanced equation for the half cell reaction resulting in ... 6 is obtained by dimerization of X through Diels – Alder reaction. |
|
About Percent Yield in the Organic Laboratory - Definitions
Definitions: Theoretical Yield: The maximum amount of product if ALL of the limiting reagent reacted exactly as described by the balanced equation |
|
DIELS-ALDER REACTION OF 1,3-BUTADIENE AND MALEIC
The dienophile, maleic anhydride, attacks the diene forming 4- cyclohexene-cis- dicarboxylic anhydride (Fig 2) The final step of the mechanism is to add water |
|
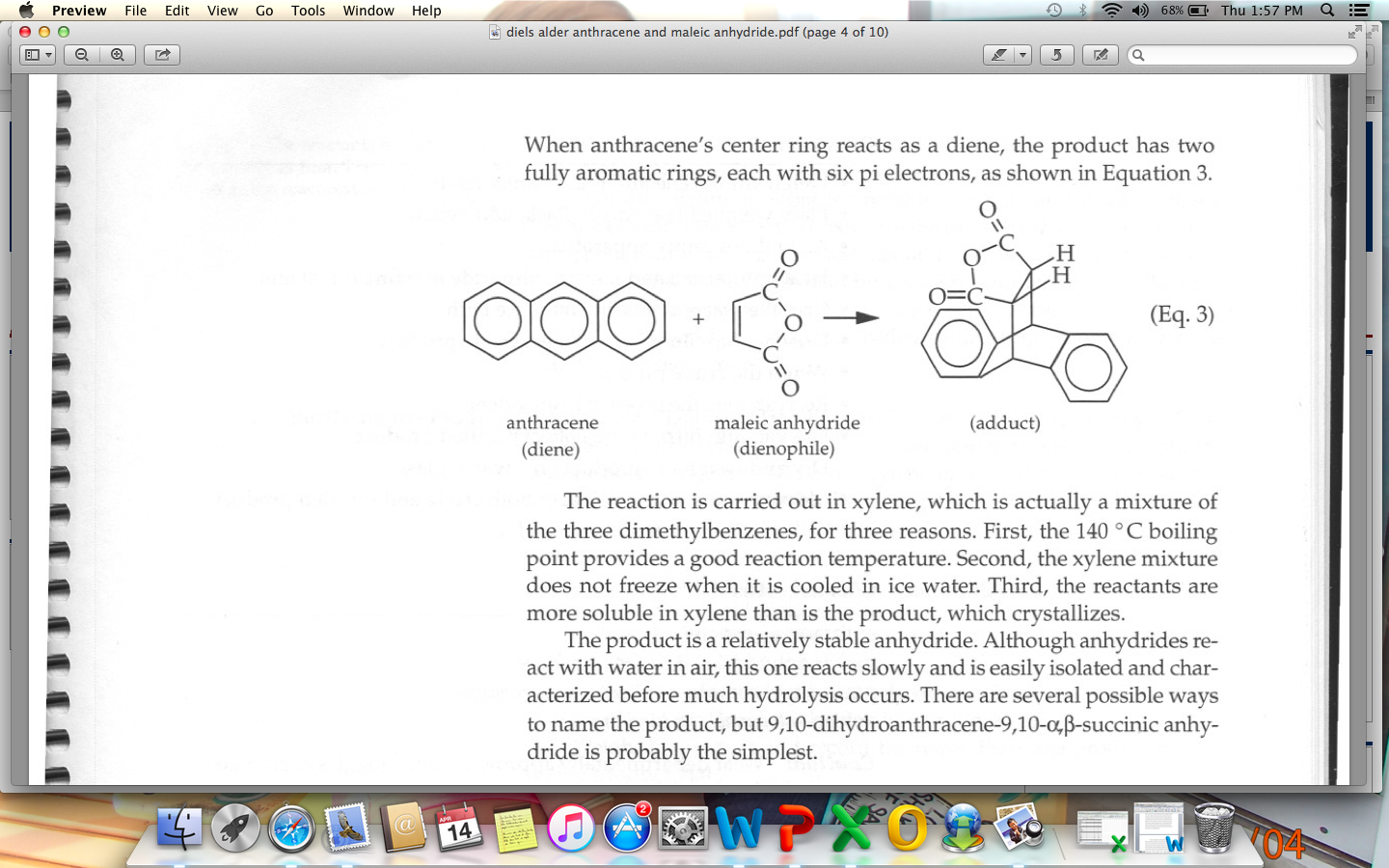
The Diels-Alder Reaction of Anthracene with Maleic Anhydride
by way of a Diels Alder reaction between anthracene and maleic anhydride, as shown in the reaction below ring stand with two utility clamps magnetic stir plate ring stand with filtering flask balance Büchner Compound Chemical formula |
|
Diels-Alder Reaction
Figure 1: The overall equation The Diels-Alder reaction is a member of a larger class of pericyclic reactions These are reactions with transition states in which the |
|
The Diels Alder Reaction - Clemson
The released butadiene from the retro Diels-Alder reaction will react with the dienophile to form an adduct, cis-4-cyclohexene-1,2-dicarboxylic anhydride |
|
The Diels-Alder lab uses two reagents: anthracene and - Chemistry
The Diels-Alder lab uses two reagents: anthracene and maleic anhydride to make the product: 9,10-Dihydro-9,10-ethanoanthracene-11,12-Dicarboxylic |
|
Diels-Alder Reaction - Chem355 Labbook-2020
General Diels-Alder Reaction: A conjugated “diene” reacts with a “dienophile” to produce a cyclohexene ring Draw the chemical equation methyl benzoate you measured a volume by syringe, rather than by weighing on a balance So you |
|
Diels-Alder PDF
The simplest Diels-Alder reaction is between 1,3-butadiene and ethylene: + The mechanism of the reaction begins with the diene assuming the higher energy, |
|
Etude et développement du processus tandem réaction de Diels
réaction présentant la séquence réaction de Diels-Alder/ réarrangement reprochent à la chimie d'influencer de façon négative la balance écologique L' équation simplifiée de la réaction pour l'énol 89b, si l'on considère le réarrangement |


















![Diels Alder Lab - [PDF Document] Diels Alder Lab - [PDF Document]](http://www.scielo.br/img/fbpe/jbchs/v12n5/a04sch29.gif)









![Perfluorobicyclo[220]hex-1(4)-ene as unique partner for Diels Perfluorobicyclo[220]hex-1(4)-ene as unique partner for Diels](https://demo.fdocuments.in/img/742x1000/reader023/reader/2020120122/5750a0f91a28abcf0c8ff828/r-1.jpg?t\u003d1610190539)