diels alder reaction ppt
|
1012 The Diels-Alder Reaction Synthetic method for preparing
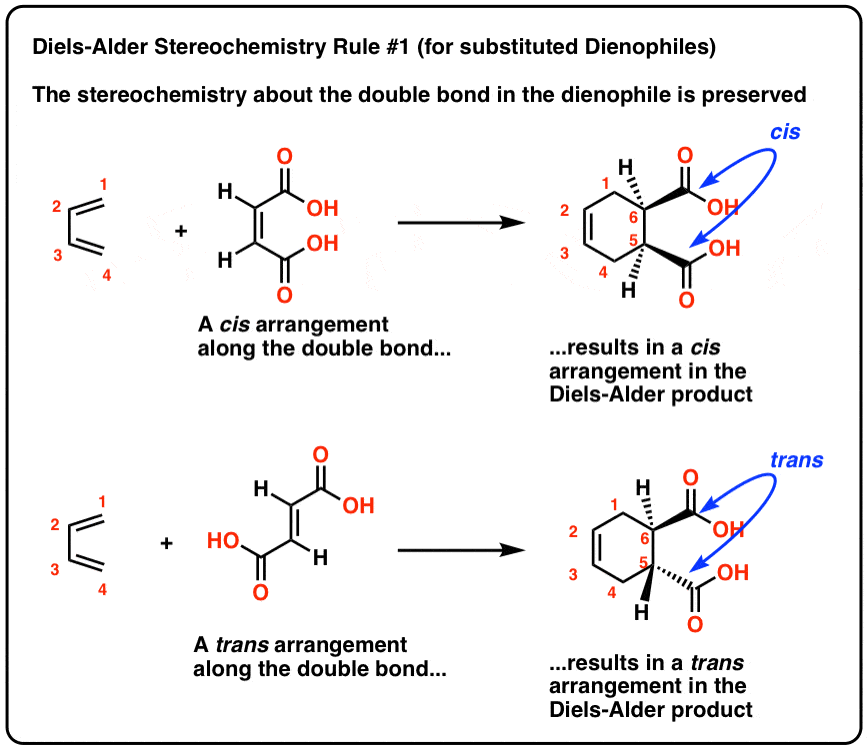
Diels-Alder Reaction is Stereospecific* syn addition to alkene cis-trans relationship of substituents on alkene retained in cyclohexene product *A stereospecific reaction is one in which stereoisomeric starting materials give stereoisomeric products; characterized by terms like syn addition anti elimination inversion of configuration etc |
What is Diels Alder reaction?
HISTORY The Diels-Alder reaction is a method of producing cyclical organic compounds (cycloaddition reaction), and is named for Otto Diels and Kurt Alder who in 1950 received the Nobel Prize for their experiments. Diene : diene are electron rich compound.
Why are hetero-substituted dienes a good substrate for Diels-Alder reaction?
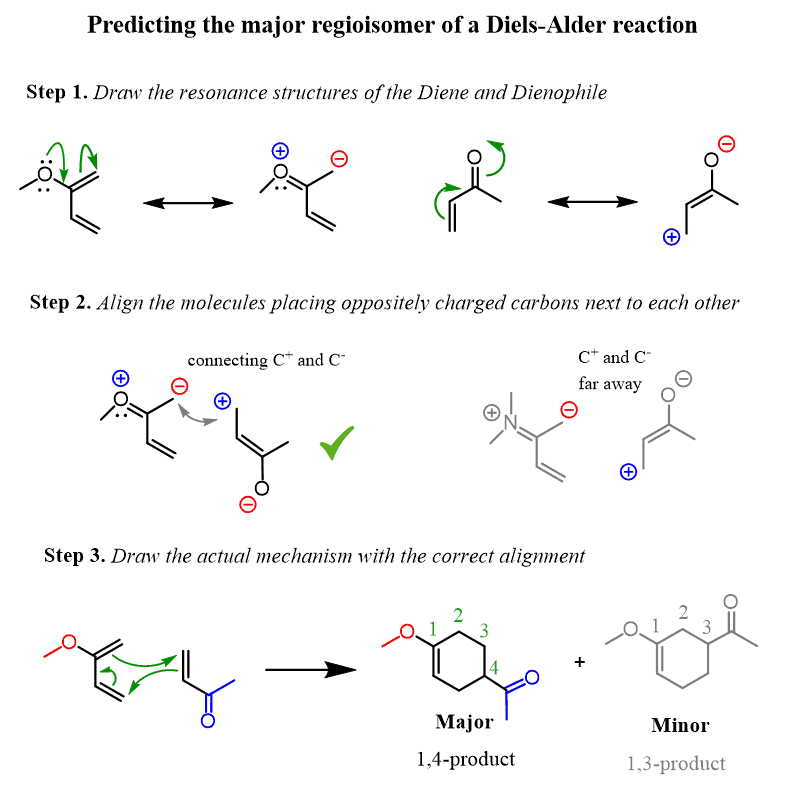
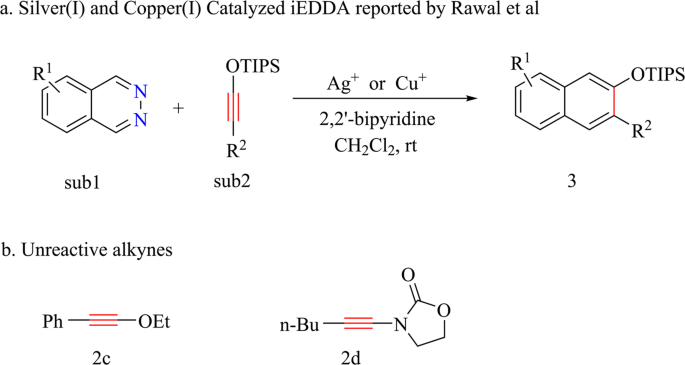
THE DIENE o Hetero-substituted dienes are excellent substrates for the Diels-Alder reaction. The increased rate of reaction is ascribed to the higher energy HOMO of the hetero- substituted diene , which therefore results in the reduction in the energy difference between the HOMO of diene and LUMO of dienophile.
How useful is the Diels-Alder reaction?
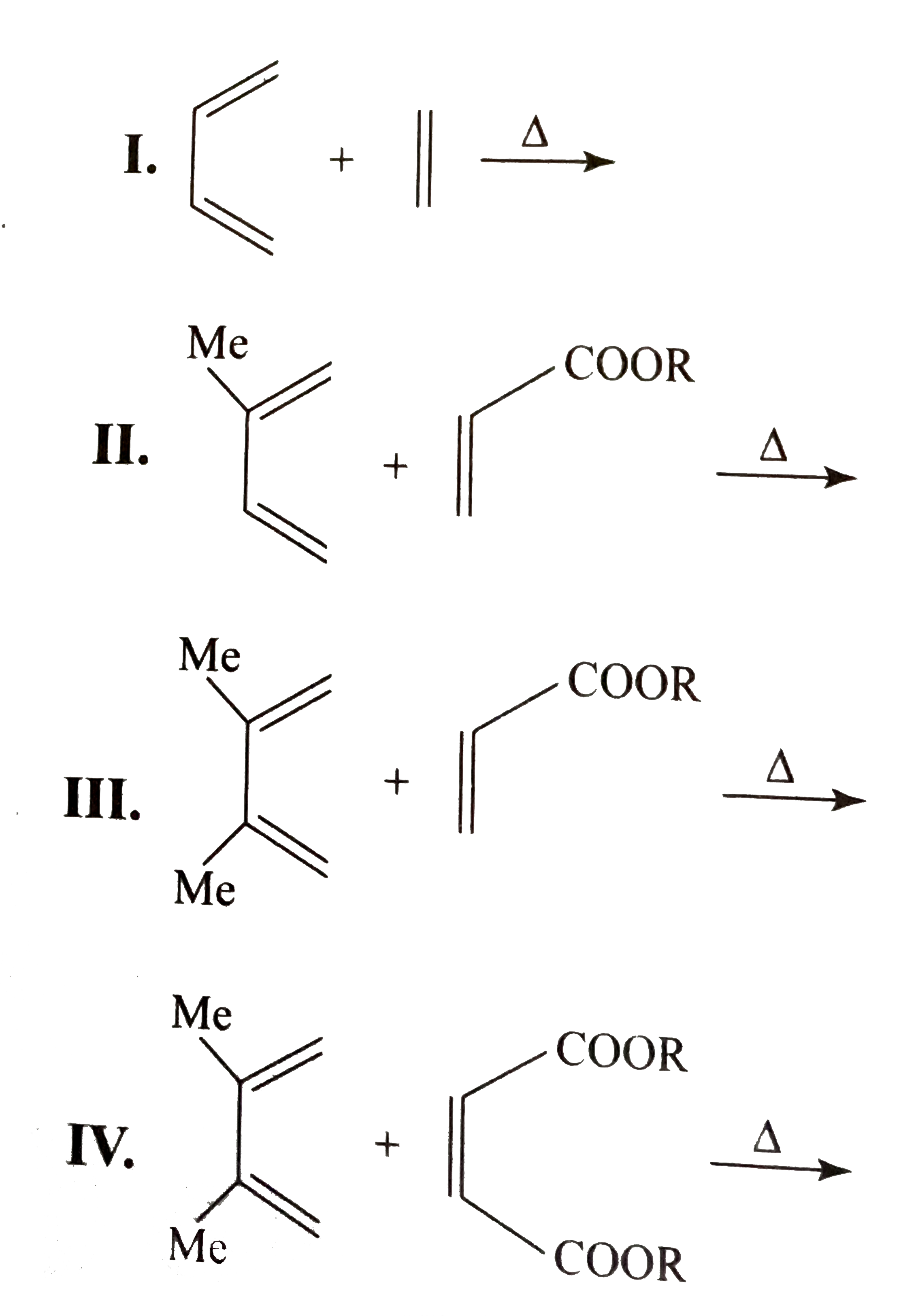
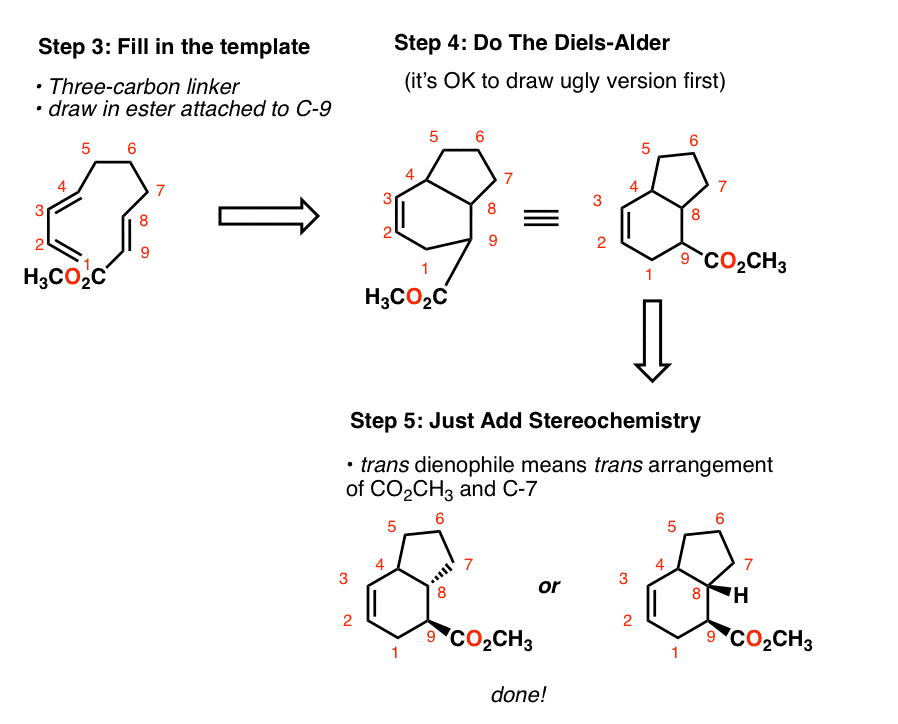
STEREOCHEMISTRY OF THE DIELS- ALDER REACTION The great synthetic usefulness of the Diels-Alder reaction depends not only on the fact that it provides easy access to a variety of six membered ring compounds but also on its remarkable stereoselectivity.
What is 4+2 cycloaddition of a conjugated diene and a dienophile?
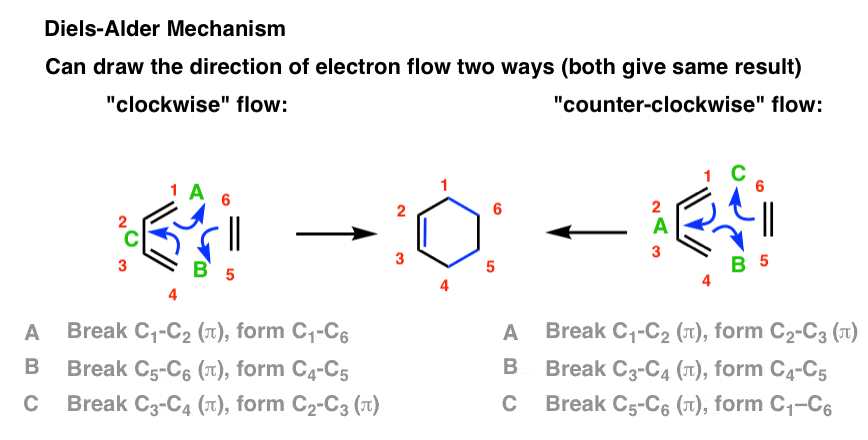
The [4+2]-cycloaddition of a conjugated diene and a dienophile (an alkene or alkyne), an electrocyclic reaction that involves the 4 π-electrons of the diene and 2 π-electrons of the dienophile. The driving force of the reaction is the formation of new σ-bonds, which are energetically more stable than the π-bonds.

Diels-Alder reaction Organic chemistry Khan Academy

Diels Alder Reaction

16.5a Introduction to Diels Alder Reactions
|
The Diels-Alder reaction
Diels-Alder (DA) reaction is incredibly valuable method for the synthesis of 6-rings. • It is not within the remit of this course to go into detail about |
|
Arylgermanes as Linkers for Solid Phase Synthesis
Woodward & Hoffmann. – Symmetry correlation diagrams. • Diels-Alder reactions. • [2+2]-cycloaddition reactions. • Electrocyclic ring-opening of cyclobutenes. |
|
The Transannular Diels-Alder Reaction: Background and Applications
What is a Transannular Diels-Alder Reaction? How does it compare with an. Intramolecular Diels-Alder Reaction? General Outline. Triene (or Diene) Geometry:. |
|
10_12_14.html.ppt [Read-Only]
The Diels-Alder Reaction. Synthetic method for preparing compounds containing a cyclohexene ring. Page 2. conjugated diene alkene. (dienophile) cyclohexene. |
|
Heterocyclic Chemistry
Synthesis and reactions of pyrazoles isoxazoles and isothiazoles as dienes in Diels-Alder reactions with highly reactive dienophiles. |
|
The Diels-Alder Reaction: an Update Introduction
Thus the original version of the Diels-Alder reaction. (Scheme 1) joins together a wide variety of conjugated dienes and alkenes with electron withdrawing |
|
6-MEMBERED RING FORMATION 150 Six Membered Rings 1
Diels-Alder Reaction. 2. o-Quinodimethanes. 3. Intramolecular ene reaction. 4. Cation olefin cyclizations. 5. Robinson annulation Diels Alder Reactions:. |
|
Diels–Alder Reaction (Part 2)
Diels-Alder reactions proceed with good dienophiles at low temperature (without the need for Lewis acid catalysis) with incredible regioselectivity. |
|
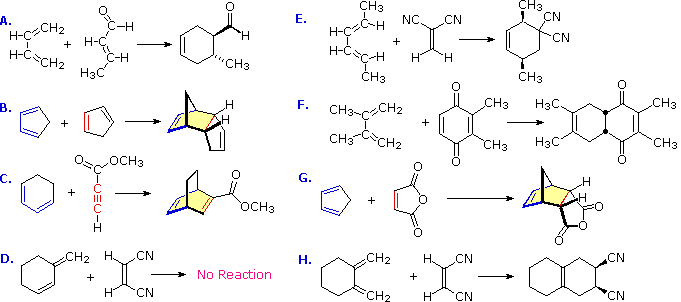
Using the Diels-Alder Reaction in the Synthesis of Biologically
08-May-2019 Diels-Alder reactions generally favor endo- substituted products due to stabilization of an endo-approach transition state by secondary orbital. |
|
Mass spectroscopy
47. Cycloalkenes usually show a distinct molecular ion peak. A unique mode of cleavage is a type of Retro Diels-Alder reaction. The fragmentation mode involves |
|
The Diels-Alder reaction
Diels-Alder (DA) reaction is incredibly valuable method for the synthesis of 6- rings • It is not within If you are a little rusty on the Diels-Alder reaction either re -read your lecture notes or These two examples form axially chiral compounds |
|
CYCLOADDITIONS IN ORGANIC SYNTHESIS
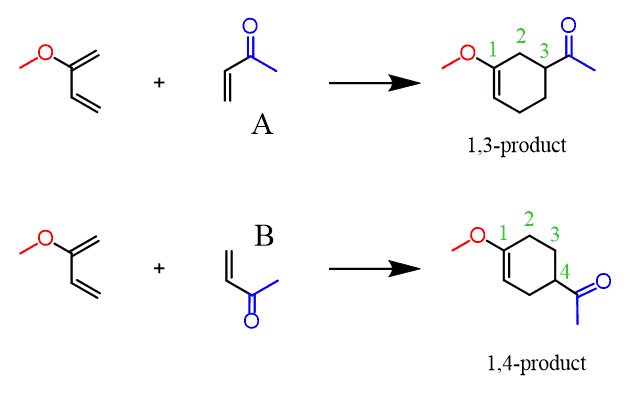
The Diels-Alder reaction is the best known of the cycloaddition reactions Although the Diels Alder reaction combines a diene and a dienophile to generate a Predicting the Regiochemical Outcome 23 Examples Generally, Terminally |
|
Asymmetric DielsAlder Reactions
These cycloadditions are proposed to proceed by a stepwise mechanism: Chiral Auxiliaries for Asymmetric Diels-Alder Reations – Applications in Synthesis: |
|
A Diels Alder reaction
At higher temperature, both of the barriers with significantly different energies can be “overcome”, but the endo „mechanism” will be reversible (Thermodynamic |
|
ASYMMETRIC CATALYSIS OF DIELS-ALDER REACTIONS
The Diels-Alder reaction of cyclopentadiene (1) with methacrolein (2), or of other (a) Outline a mechanism of the uncatalyzed Diels-Alder reaction between |
|
34 - MSU chemistry
PROBLEM 1 Predict the structure of the product of this Diels-‐Alder reaction CO2Me compound We should draw a mechanism for one of the reactions to see |
|
1 Asymmetric Catalysis of Diels–Alder Reaction - Wiley-VCH
OR OR O O CO2H O B O O R' Scheme 1 2 Selected examples for enantioselective DA reactions catalyzed by 1–3 1 2 Asymmetric Diels–Alder Reaction j3 |
|
Tema 2 Pericyclic Reactions
The organic reactions can be classified according to their mechanism 1 Ionic or polar For a review on the Diels-Alder reaction in total synthesis: Nicolaou |
|
Investigating Imidazolidinone Catalysts - Caltech THESIS
Figure 11 Iminium catalysis cycle for Diels–Alder reaction of cinnamaldehyde and cyclopentadiene Figure 12 Components of iminium reaction mechanism |