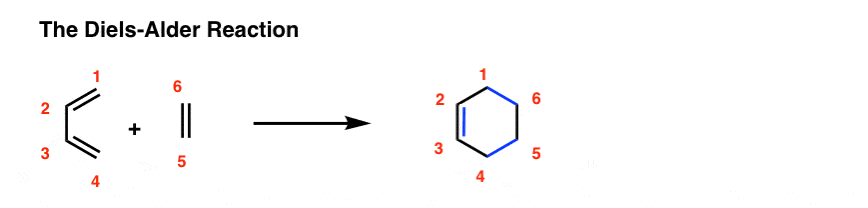
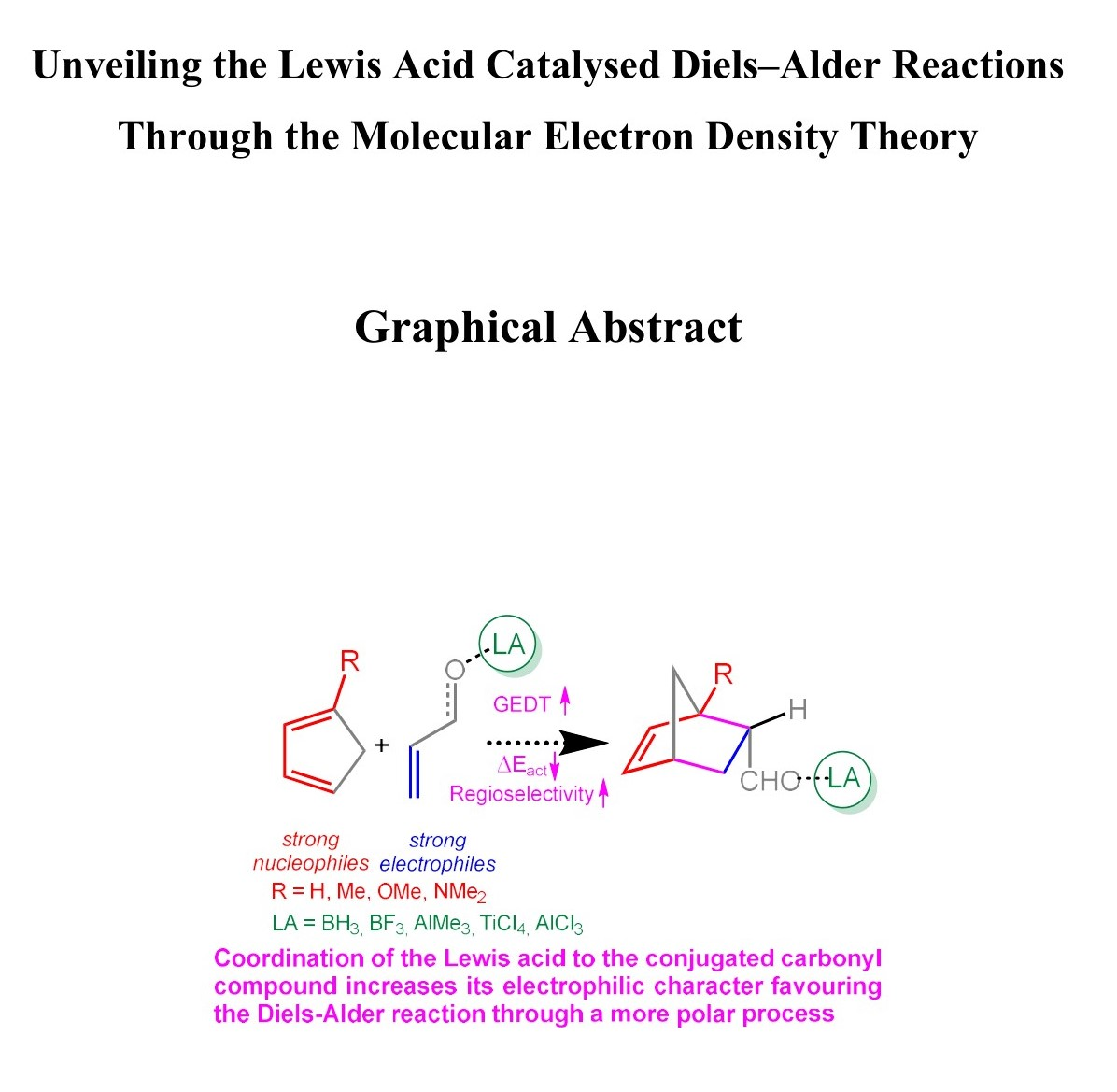
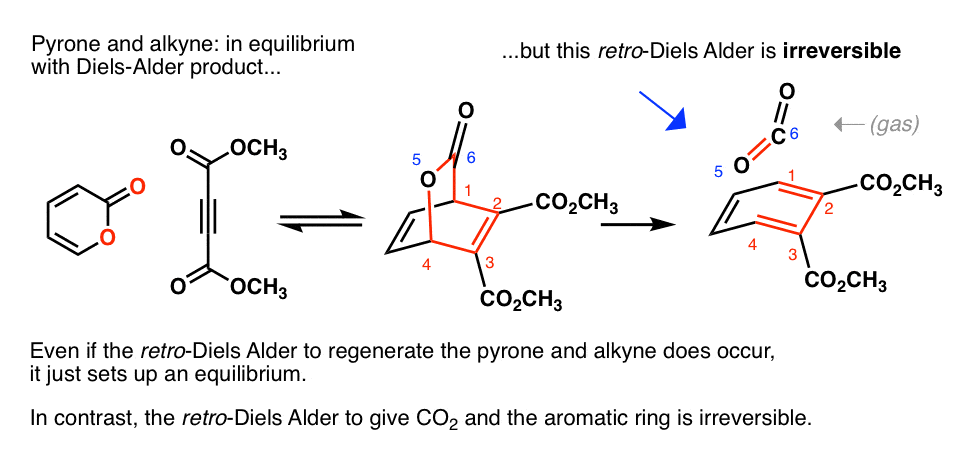
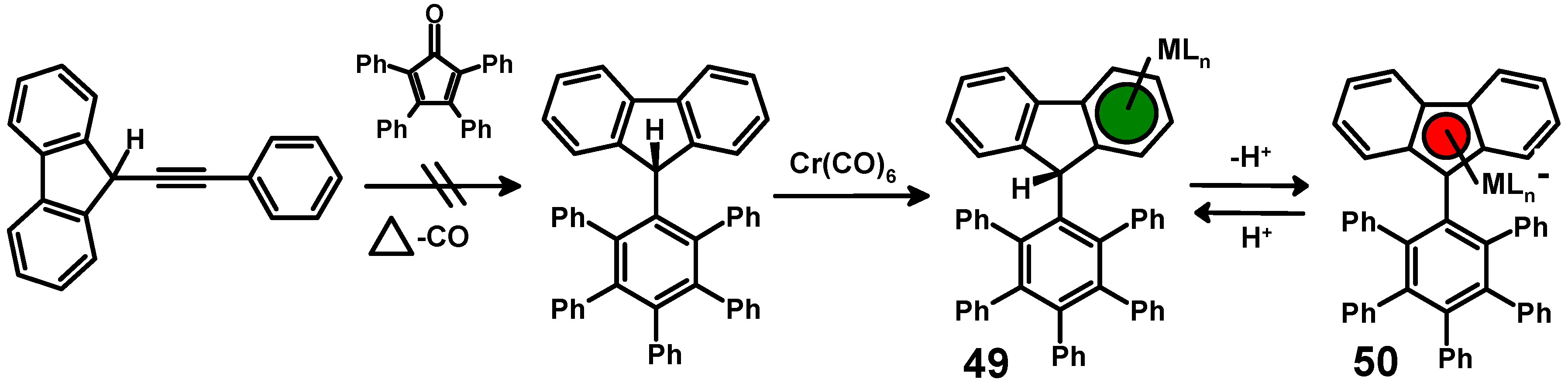
diels alder reaction practice problems
|
Microsoft Word
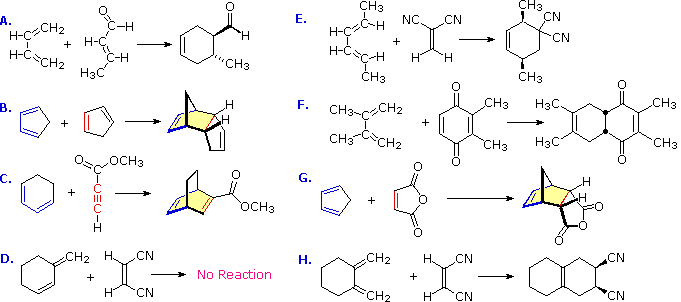
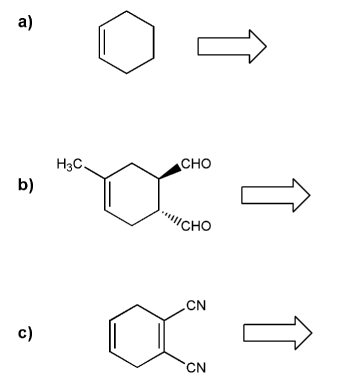
1 Predict the product of the following Diels-Alder reactions; under kinetic control Include the stereochemistry where appropriate 2 Furan and maleimide shown below react to produce and adduct via a Diels-Alder reaction At 25°C the isomer produced is the endo product however at 90°C the exo isomer predominates |
|
Practice Problems on Diels-Alder
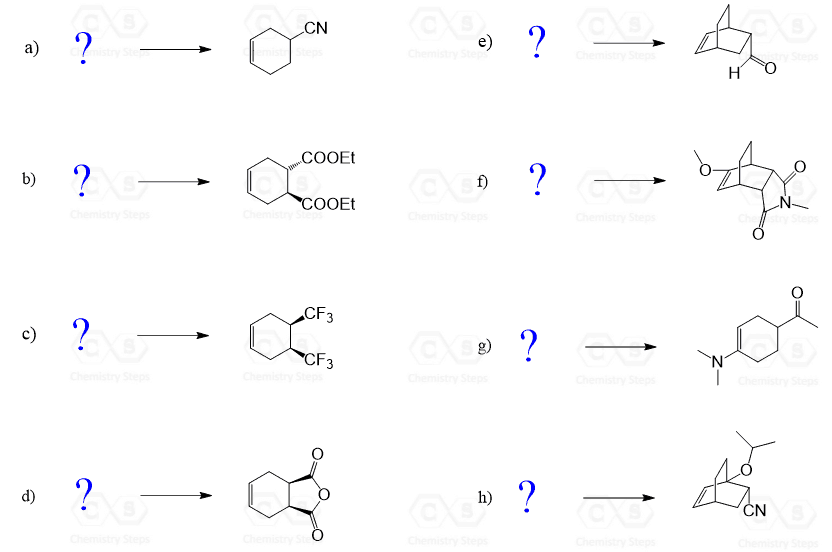
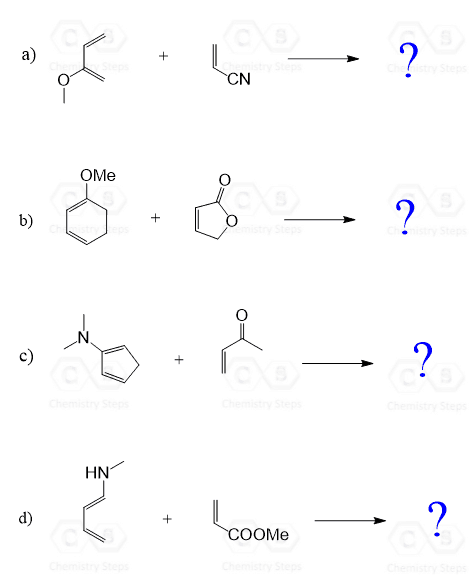
Practice Problems on Diels-Alder Reactions 1 Predict the product of the following Diels-Alder reactions; under kinetic control Include the stereochemistry where appropriate (a) + COOH (b) + O (c) H + C C O CH 3 O O CH 3 H CN (c) + H CN 2 Furan and maleimide shown below react to produce and adduct via a Diels-Alder reaction |
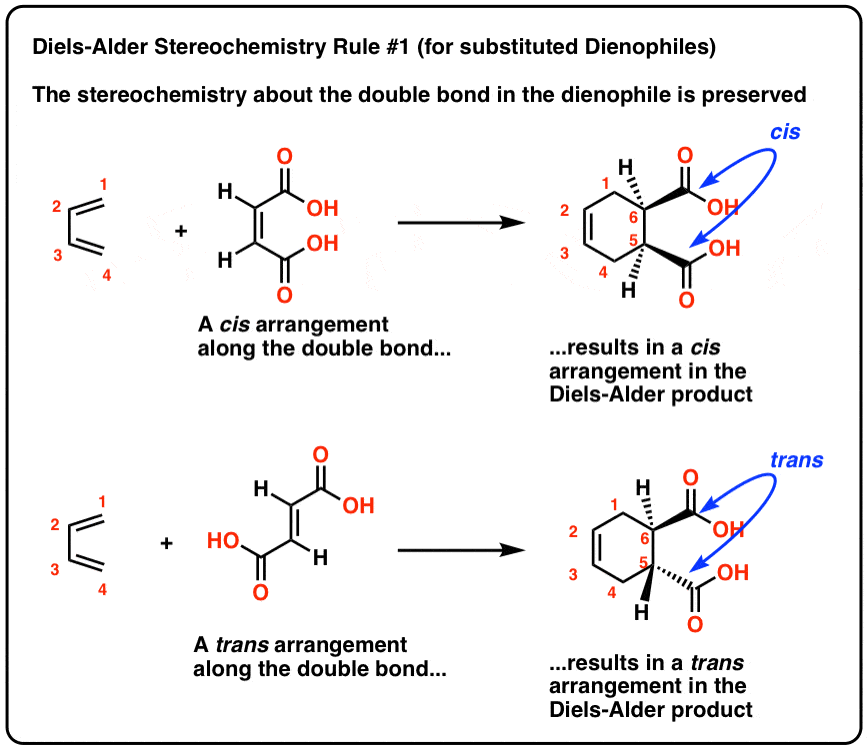
Are Diels-Alder reactions stereospecific?
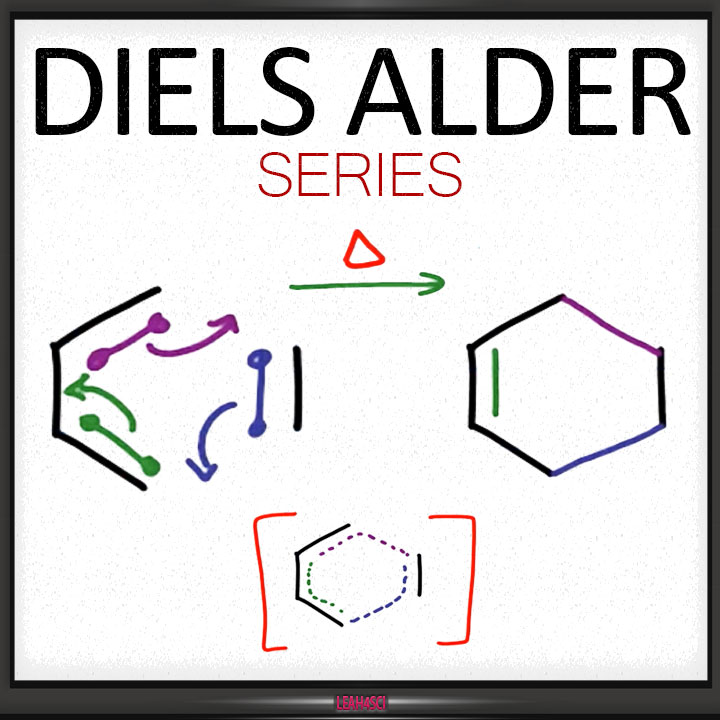
Secondly, Diels-Alder reactions are stereospecific.This means that the substituents attached to the both the diene and the dienophile retain their stereochemistry throughout the reaction.For example, if the functional groups on the dienophile are trans to each other in the reactants, they should remain trans to each other products.
Does a Diels Alder reaction require heat?
Although heat is not required in Diels-Alder reactions, heating up the reaction will improve yield. But again heat is not required for the reaction to go through. To go into more detail, the alkene that reacts with the diene is commonly reffered to as the dienophile .Although this reaction occurs readily, it doesn’t give a very good yield.
How do you predict a Diels-Alder reaction?
1. Predict the product of the following Diels-Alder reactions; under kinetic control. Include the stereochemistry where appropriate. 2. Furan and maleimide, shown below, react to produce and adduct via a Diels-Alder reaction. At 25°C the isomer produced is the endo product, however at 90°C the exo isomer predominates.
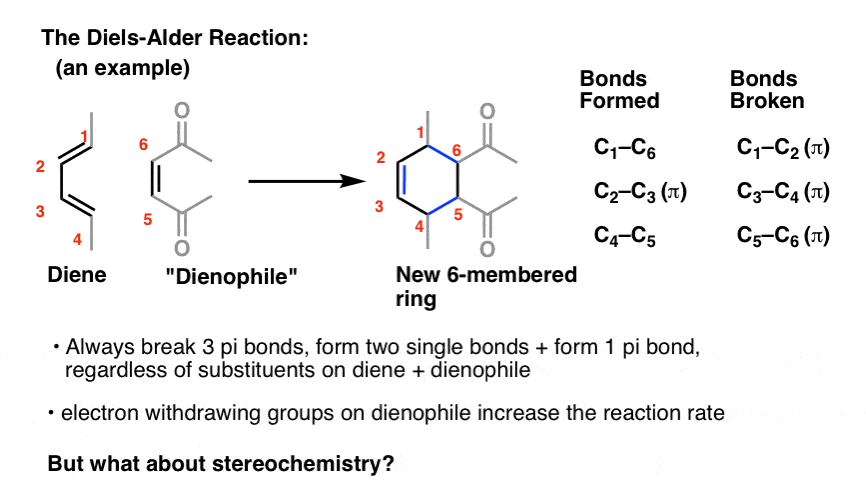
Which is the preferred outcome of the Diels-Alder reaction under kinetic control?
The endo product is the preferred outcome of the Diels-Alder reaction under kinetic control. The transition state to the endo product has lower activation energy than transition state of the exo product. c) Is your answer to question 4(b) dependent on a kinetically or thermodynamically controlled reaction? Yes.
How Do I Figure This Out For Any Diels-Alder reaction?
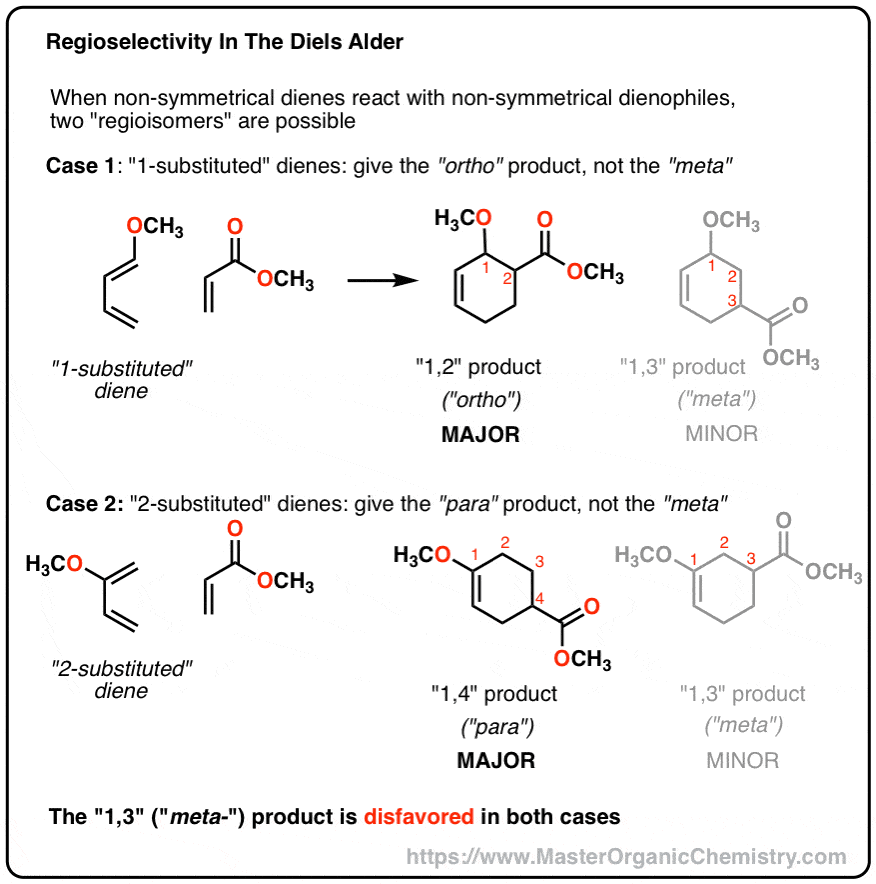
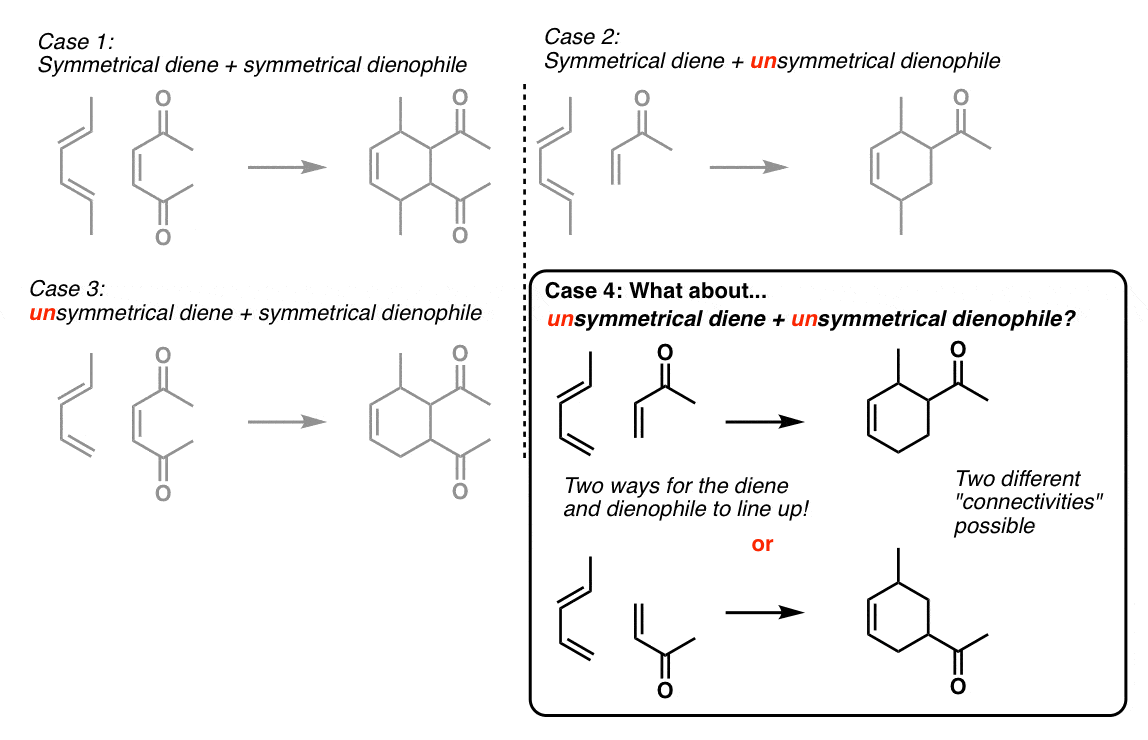
This regioselectivity is a result of the electron distribution in the diene and the dienophile. The most electron-rich carbon of the diene reacts with the mot electron-deficient carbon of the dienophile. You have two ways to determine the proper alignmentof the diene and the dienophile. The first one is to draw the resonance structures of the diene
Is There A Shorter route?
You can also predict the major regioisomer of a Diels-Alde reaction without drawing the resonance forms. Simply place the molecules next to each other and draw the curved arrowsconnecting the first two carbons of the diene and the dienophile; The correct alignment is the one that supports theelectron flow from the electron-donating diene substituen
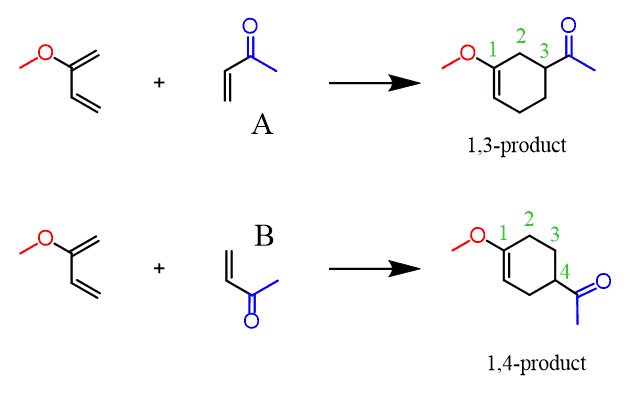
What If The CH3O- Is on The Other Carbon?
Let’s also look at an example where the electron-donating group of the diene is on carbon number 1 (1-substituted diene): This time, we can first try the electron-flow method: Making a short summary, we can see that depending on the structure of the diene, the 1,2 or 1,4-product is obtained. The 1,3-product is never favored. chemistrysteps.com
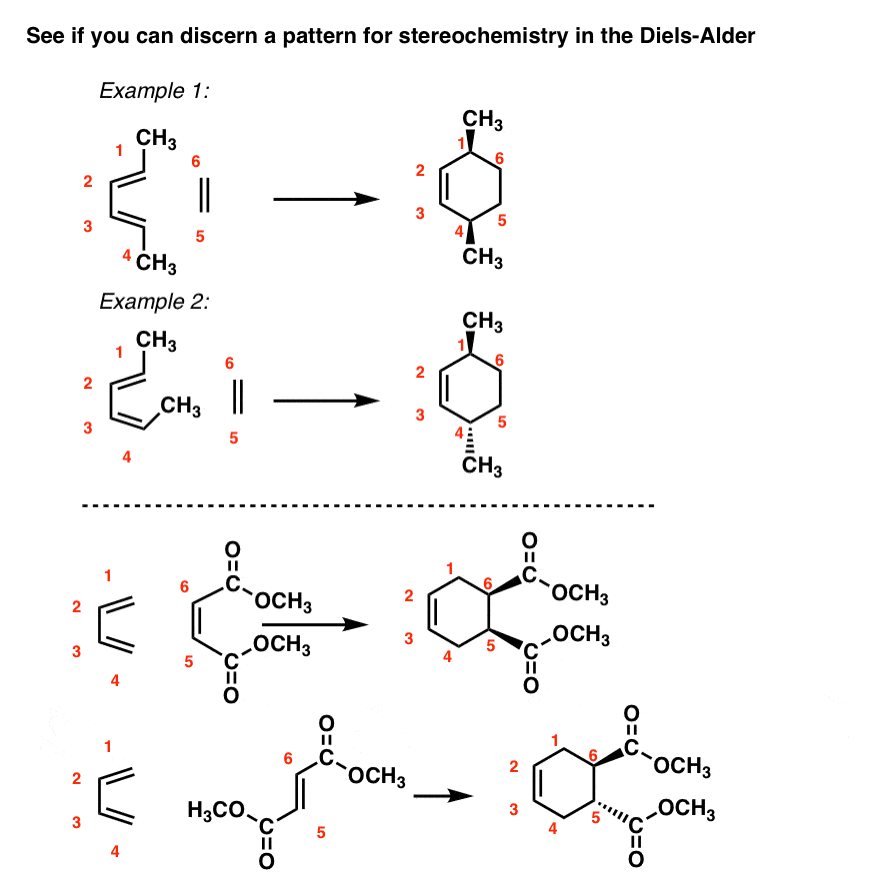
Stereochemistry of The Unsymmetrical Diels-Alder Reaction
In reactions of both 1-substituted diene and 2-substituted diene, there is a formation of a stereogenic center(s) which we ignored so far to avoid additional complications. And depending on what your instructor asks, you may not need this at all. However, we will address this as well. Let’s start with the 2-substituted diene since this one is a bit

Diels Alder Reaction: Organic Chemistry PRACTICE PROBLEMS

Diels-Alder reaction Organic chemistry Khan Academy

Practice Problem: Diels-Alder Reactions
|
Practice Problems on Diels-Alder Reactions – Answers 1. Predict
Furan and maleimide shown below |
|
Practice Problems on Diels-Alder Reactions 1. Predict the product of
Furan and maleimide shown below |
|
Test 4 Extra Practice Conjugation-Allylic-Diels-Alder
Extra Practice Problems: Conjugated Systems Dienes |
|
PRACTICE PROBLEMS – UNIT 12
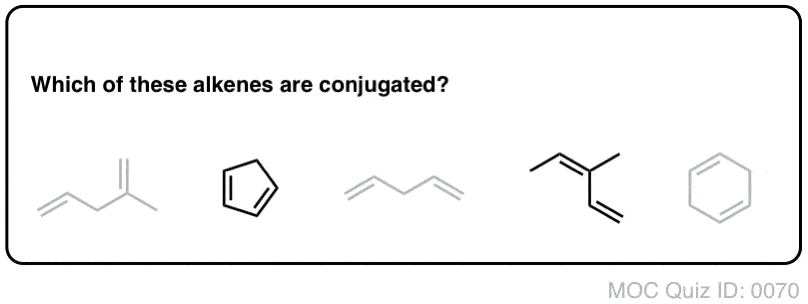
PRACTICE PROBLEMS – UNIT 12. 12A. Identify groups of conjugated atoms. Predict the products of the Diels-Alder reaction including stereochemistry. |
|
Untitled
PRACTICE PROBLEMS. -. UNIT 12. 12A. Identify groups of conjugated atoms. O. C. = conjugated. 12A.1 Determine if the alkenes in the following molecules are |
|
Retro Diels-Alder worksheet
A common question for retro DA reactions is shown below. Here are a few more practice problems on the retro Diels-Alder (DA) reactions. |
|
Practice Set Answer Keys Organic Chemistry I Table of Contents
Test 4 PS2: Test 4 Conjugation-Allylic-Diels-Alder Practice Draw arrows for each of the steps in the following reactions. |
|
Answers To Chapter 4 Problems.
Diels–Alder reaction. The C=C bond of ketenes is pretty electron-rich due to overlap with the lone pairs on O: H2C=C=O¨ ? H2C. |
|
CHEM 242-601 Practice for Quiz 2 (problems from old quizzes) 1
(12) The following Diels-Alder reaction between furan and thiomaleic anhydride gives rise to the exo adduct (A). Subsequent oxidation of A with |
|
Organic Chemistry II_Week 8
In these sessions I will provide practice problems and be available for specific Key Words: Diels-Alder Electrocyclic Reactions |
|
Practice Problems on Diels-Alder Reactions - Chemwiscedu
Furan and maleimide, shown below, react to produce and adduct via a Diels- Alder reaction At 25°C the isomer produced is the endo product, however at 90° C |
|
Practice Problems on Diels-Alder Reactions 1 Predict the product of
Furan and maleimide, shown below, react to produce and adduct via a Diels- Alder reaction At 25°C the isomer produced is the endo product, however at 90° C |
|
PRACTICE PROBLEMS – UNIT 12
Predict the products of the Diels-Alder reaction including stereochemistry 12C 1 Predict the major product, if any, indicate stereochemistry where appropriate a) |
|
Diels – Alder Reaction 1,4-CycloAddition Reaction of Dienes
What's Going With Respect to a Diels-Alder 1,4 Cycloaddition Reaction? X X Examples of Dienes and Dienophiles Solomons and Fryhle, problem 14 16,17 |
|
Test 4 Extra Practice Conjugation-Allylic-Diels-Alder
Extra Practice Problems: Conjugated Systems, Dienes, Allylic Systems and the Diels-Alder Reaction 1 Rank the heats of hydrogenation for the following, |
|
CHEM 242-601 Practice for Quiz 2 (problems from old quizzes) 1
(12) The following Diels-Alder reaction between furan and thiomaleic anhydride gives rise to the exo adduct (A) Subsequent oxidation of A with |
|
Problem set 8-27-10
27 août 2010 · The Diels-Alder Reaction James Partridge Diels Alder quotes • “Thus it appears to us that the possibility of synthesis problems ” 5 |
|
34 - MSU chemistry
PROBLEM 1 Predict the structure of the product of this Diels-‐Alder reaction with a ketone in it might present a problem, but the ten-‐membered ring containing problem Revision of synthesis (chapters 24 and 28) with some cycloaddition |
|
316 DielsAlder practice keycdx
Diels-Alder Practice Problems: Answer Key tBu tBu prohibits the required s- cis conformation so no Diels-Alder reaction attempt to rotate this pi bond |
|
Practice Questions : Chem 226 / Exam 3 MULTIPLE CHOICE
Practice Questions : Chem 226 / Exam 3 MULTIPLE CHOICE 13) Which of the following dienes is the most reactive in a Diels-Alder reaction? A) I B) II C) III |