dilute solution
What is the difference between dilution and concentration?
Dilution is the addition of solvent, which decreases the concentration of the solute in the solution. Concentration is the removal of solvent, which increases the concentration of the solute in the solution. (Do not confuse the two uses of the word concentration here!) In both dilution and concentration, the amount of solute stays the same.
What does it mean to dilute a solution?
To dilute a solution means to add more solvent without the addition of more solute. The resulting solution is thoroughly mixed so as to ensure that all parts of the solution are identical. The same direct relationship applies to gases and vapors diluted in air for example.
How do you dilute and concentrate a solution?
Learn how to dilute and concentrate solutions. Often, a worker will need to change the concentration of a solution by changing the amount of solvent. Dilution is the addition of solvent, which decreases the concentration of the solute in the solution.
How do you calculate dilutions in chemistry?
Preparing dilutions is a common activity in the chemistry lab and elsewhere. Once you understand the above relationship, the calculations are simple. Suppose that you have 100.mL 100. mL of a 2.0M 2.0 M solution of HCl HCl. You dilute the solution by adding enough water to make the solution volume 500.mL 500. mL.
Overview
0 energy points A common method of making a solution of a given concentration involves taking a more concentration solution and adding water until the desired concentration is reached. This process is known as dilution. We can relate the concentrations and volumes before and after a dilution using the following equation: M₁V₁ = M₂V₂ where M₁ and V₁ represent the molarity and volume of the initial concentrated solution and M₂ and V₂ represent the molarity and volume of the final diluted solution. Created by Sal Khan. khanacademy.org
Want to join the conversation?
Log in khanacademy.org

What is Dilute Solution? Chemistry

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations

Dilute Solutions One Shot #BounceBack Series Unacademy Atoms Sakshi Vora
|
15 Theory of Dilute Solutions
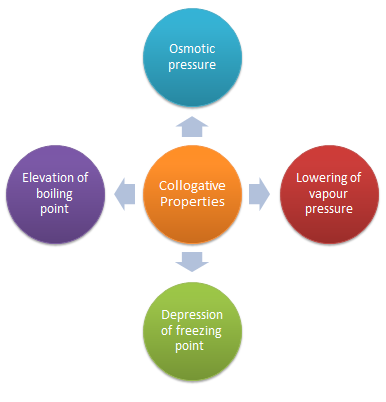
COLLIGATIVE PROPERTIES Dilute solutions containing non-volatile solute exhibit the following properties : (1) Lowering of the Vapour Pressure |
|
Lecture 17 Dilute Solutions (Ch 5 ) - Rutgers Physics
To get the quantitative results we need the equation for the Gibbs free energy of a dilute solution in terms of the number of solvent and solute molecules |
|
Thermodynamics of dilute solutions
liquid binary dilute solution at temperature T and pressure p in which an amount n2 of a (non-dissociating) solute 2 (of |
|
Dilute solution theory of polymer crystal growth: Fractionation effects
crystal growth from dilute solution for a monodisperse polymer In a second paper [18] paper II we applied the theory to polyethylene (PE) crystallized |
|
15 Theory of Dilute Solutions
9 oct 2020 · The solid is referred to as the solute and the liquid as the solvent COLLIGATIVE PROPERTIES Dilute solutions containing non-volatile solute |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
material to be diluted) + 4 unit volumes of the solvent medium (hence 1 + 4 = 5 = dilution factor) Example 1: To dilute a streptavidin solution 1:300 |
|
Preparation and Dilution of Solutions
B Solution dilution 1) Volume to volume dilutions : ? This type of dilutions describes the ratio of the solute to the final volume of the dilute solution |
|
Unit -2 THEORY OF DILUTE SOLUTIONS - PUE
2) What is dilute solution? [1] A: It is a solution in which solute concentration is very less of all the components in the solution 10) Define molarity and give the |
|
Thermodynamics of dilute solutions
solution" and "ideal dilute solution," and by the confusion arising from the use of liquid, binary dilute solution, at temperature T and pressure p, in which an |
|
Diluting a % solution - Mathcentre
Q1 How much water should you add to 50mL of a 10 v/v solution to reduce it in strength to a 0 5 v/v solution? Q2 How much water should you add to 1 2L of |
|
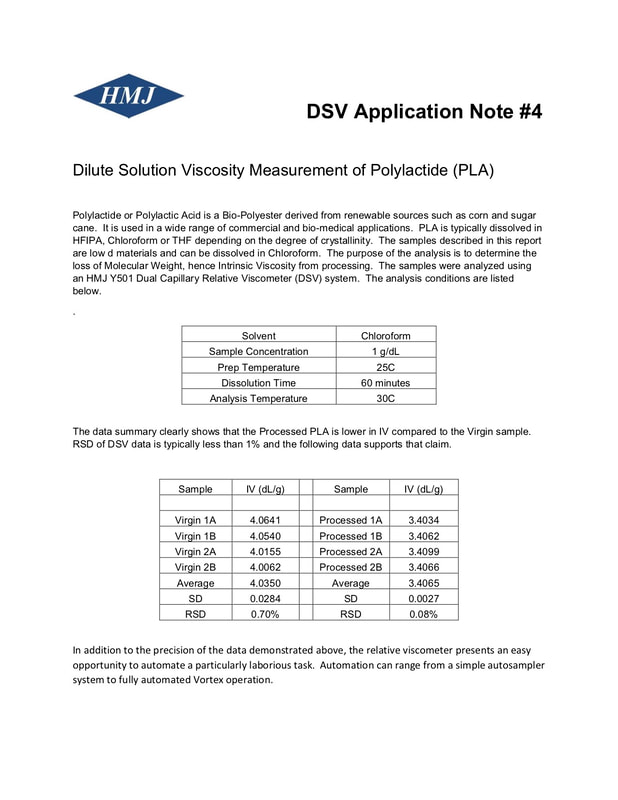
DILUTE SOLUTION PROPERTIES OF - ScienceDirect
Abstract--Viscosities and osmotic pressures of dilute solutions of fractions of polyacrylonitrile and of copolymers of acrylonitrile and methyl acrylate, and of |









/volumetric-flask-containing-solution-of-potassium-dichromate--vi---k2cr2o7--and-other-flasks-of-transition-metal-salts--dry-chemicals-and-solutions--compounds-with-transition-metals-are-often-colored-702545755-5a579379eb4d520037b7a397.jpg)