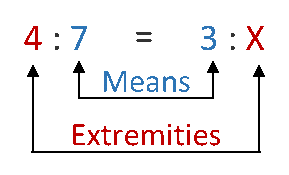
dilution calculator weight percent
When should a dilution equation be used?
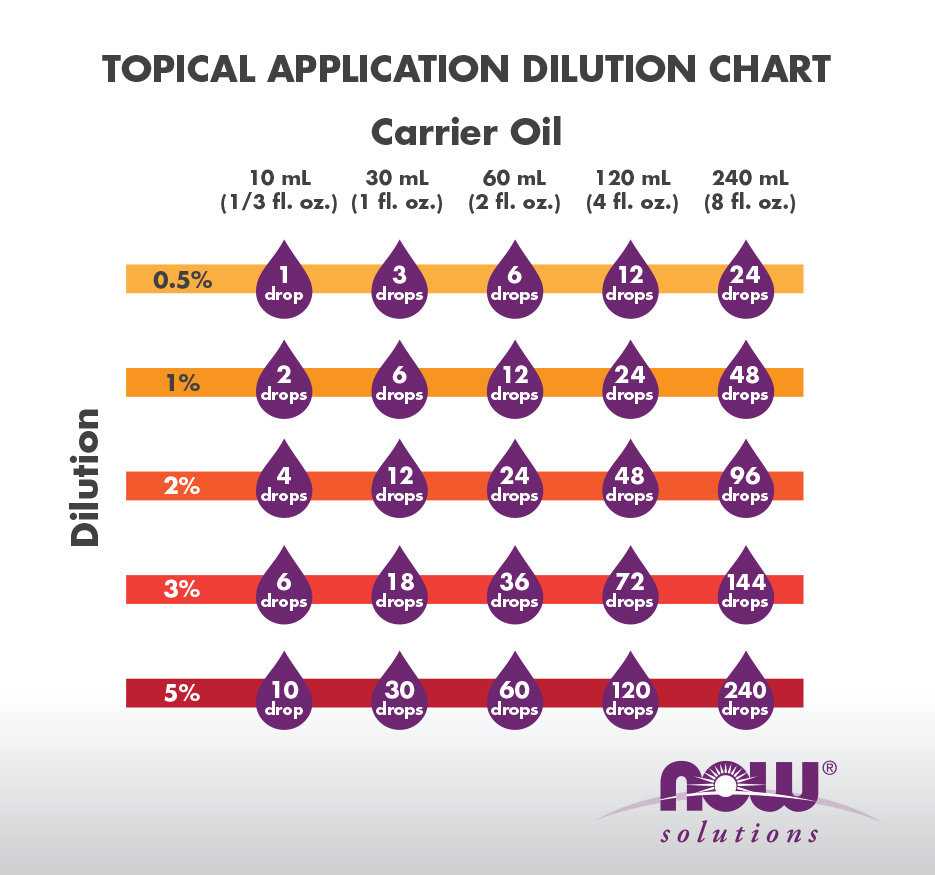
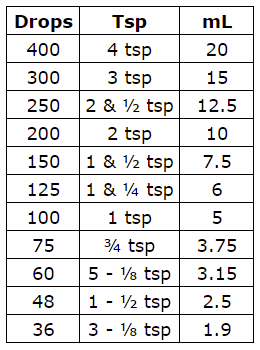
It should be used when talking about dissolving a liquid chemical into a liquid diluent. For example a 10% v/v solution of HCl means 10 mL of HCl were added to 90 mL of water to obtain a solution whose final volume is 100 mL. The dilution equation works even when you don't have a molarity associated with the stock.
What is the difference between dilution and solute concentration?
The solute concentration describes the amount of solute dispersed in a given quantity of the solvent. Dilution is a process whereby the concentration of the solute in a stock solution is reduced by the addition of more solvent. Diluting a solution resulting from a previous dilution by adding the same amount of solvent is known as a serial dilution.

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations

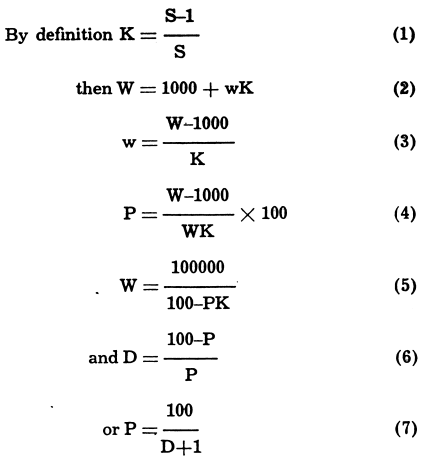
How to Calculate Dilution Factor

How To Dilute Detailing Products (10:1 4:1)
|
Medical Calculations
Medical Calculations. Dilutions and Solutions (Cont.) When calculating the percent strength of a solution (w/v) use the following formula: mass (g) |
|
1160 PHARMACEUTICAL CALCULATIONS IN PRESCRIPTION
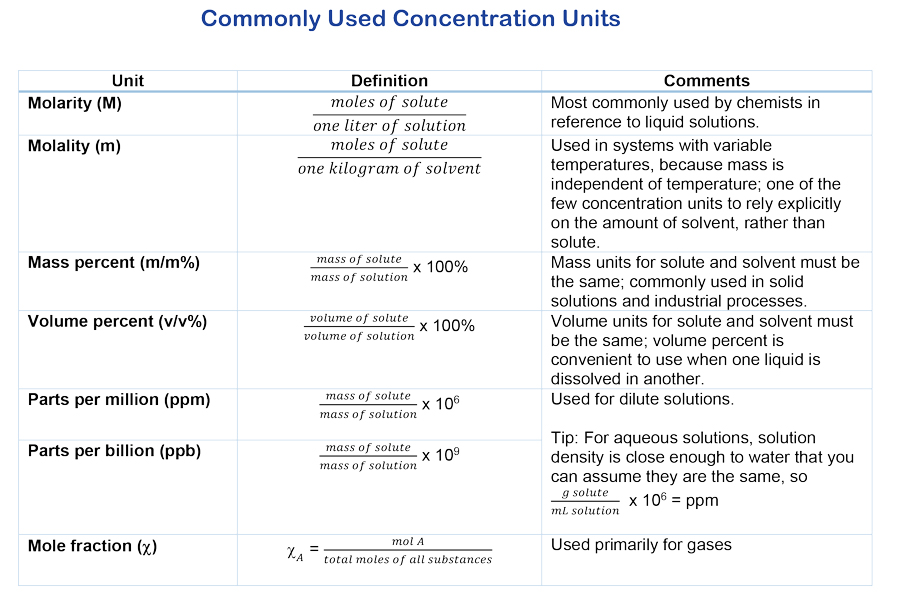
formula weight of the ingredient including waters of hydration for hydrous Weight percent (w/w) = (Weight of solute × 100%)/Weight of solution. |
|
This is the table of contents and a sample chapter from WSO Water
Equivalent Weights and Normality. 7. Dilution Calculations The first step in calculating percent by weight of an element in a compound. |
|
Clinical Mathematics for Anesthetists
Calculate desired rate setting for IV infusion pumps given weight |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
When working with a dry chemical it is mixed as dry mass (g) per volume where #g/100 ml = percent concentration. A 10% NaCl solution is equal to 10 g dissolved |
|
(2) Preparation and Dilution of Solutions
W/V% ? Weight/Volume Percentage Concentration. ?In order to calculate the volume of the stock solution required for a given preparation the density. |
|
PESTICIDE RATE AND DOSAGE CALCULATIONS
always expressed as percent by weight. Application rates are usually expressed HOW TO CALCULATE PESTICIDE DILUTIONS AND DOSAGES FOR LARGE AREAS ... |
|
Calculating Liquid Chemical Dilutions
Example No. 1 - Dry Powder-weight Dilutions. Assume desired concentration of citric acid solution is 15 percent (%). Concentration of stock dry powder. |
|
Conserve O Gram Volume 6 Issue 4: Making Percent Solutions of
while submerged in a strongly alkaline solution. percent is measured as the ratio of the weight ... To use non-metric quantities first calculate. |
|
Pharmaceutical calculation
16 juil. 2021 If an injection containing a medication 50 mg/10 mL |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
w/v = weight (of solute) per final solution volume 1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 unit volume of diluent (the material to be mixed as dry mass (g) per volume where #g/100 ml = percent concentration A 10 |
|
Percent Solution Concentration Calculator - dTV
Percent solutions can take the form of weight/volume (wt/vol or w/v ), weight/weight (wt/wt or w/w ), or Dilution calculator for percent solutions |
|
Dilution and Concentration
Determine the percent strength and ratio strength of a given product when the This relationship is generally true except for volume-in-volume and weight-in- |
|
Diluting a % solution - Mathcentre
solution to reduce it in strength to a 0 4 w/v solution? Q3 You have 80g of a 15 w/w concentration What weight of base should you add to reduce its strength |
|
Percentage Solution Calculations
23 fév 2018 · Percent solutions can take the form of weight/volume (wt/vol or w/v ) Dilution Calculator - Molarity, Percent - PhysiologyWeb |
|
Citric Acid Dilution Chart
Citric Acid Dilution Chart percent citric acid would be made from 436 grams (g) of dry powder citric acid and The weight of one ml of pure water is one gram |
|
Concentrations and Dilutions INTRODUCTION
as ointment, percent strength would represent the number of grams contained in Calculating weight/weight concentrations can be easily and accurately performed by Rx aluminum acetate solution 1:13 dilution 480 mL You have a box of |