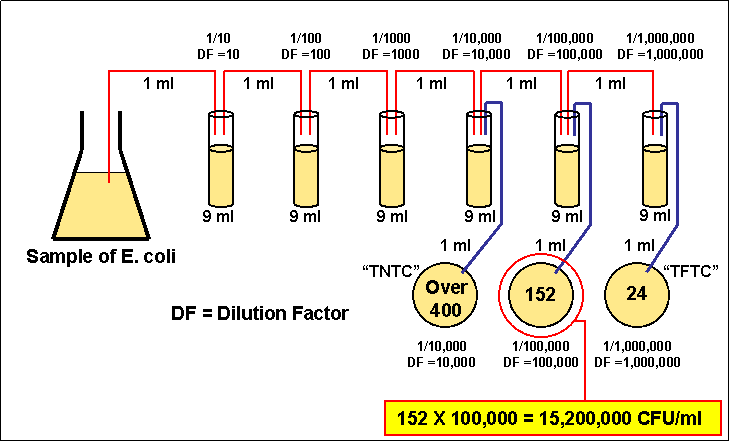
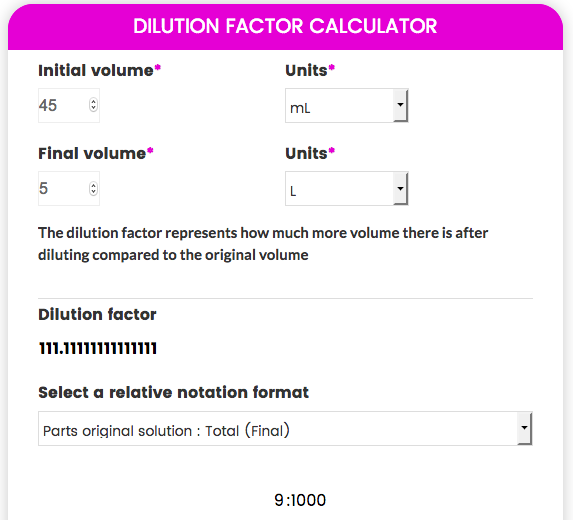
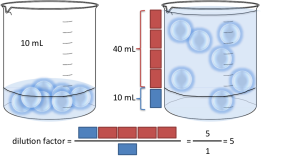
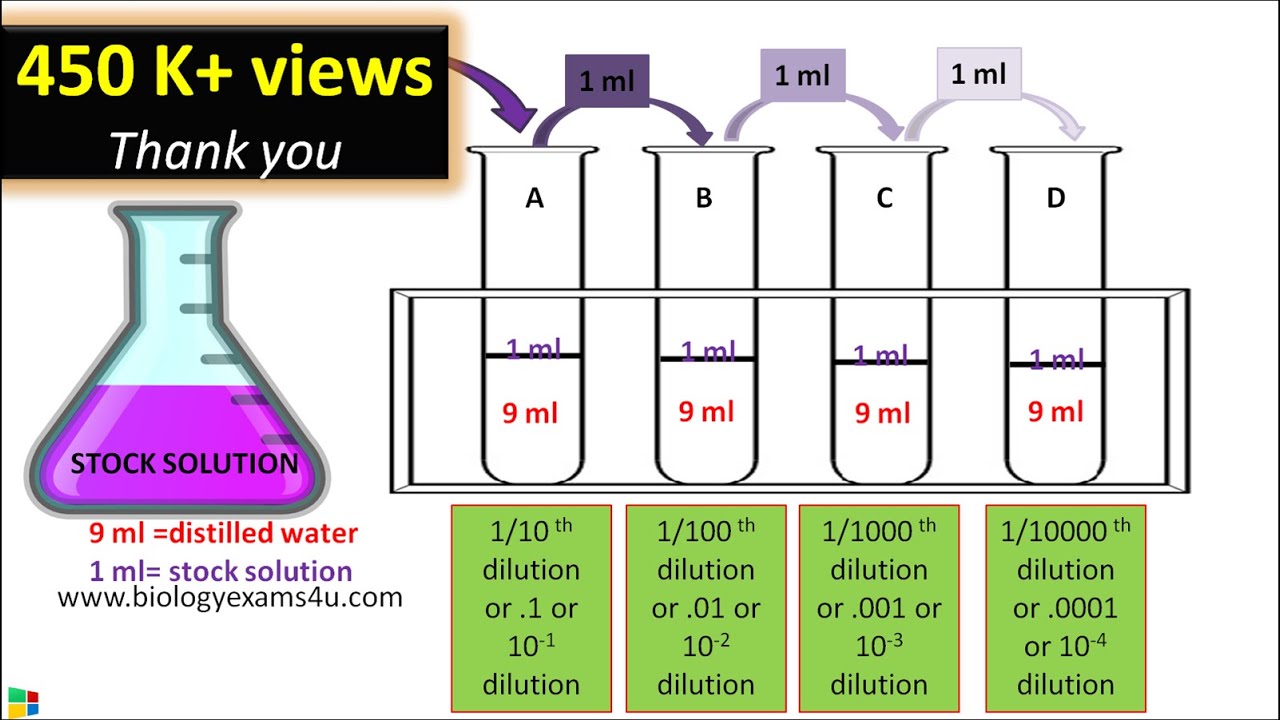
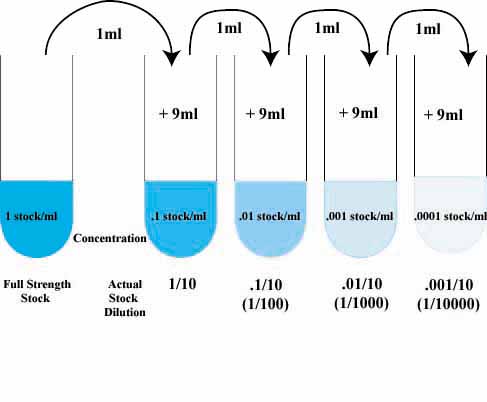
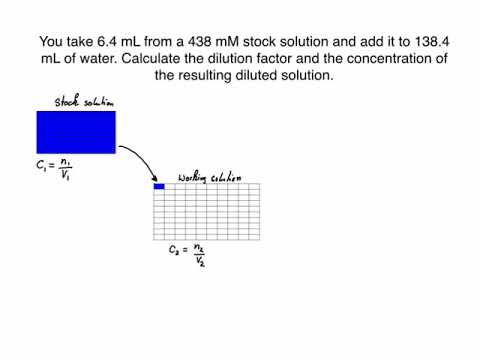
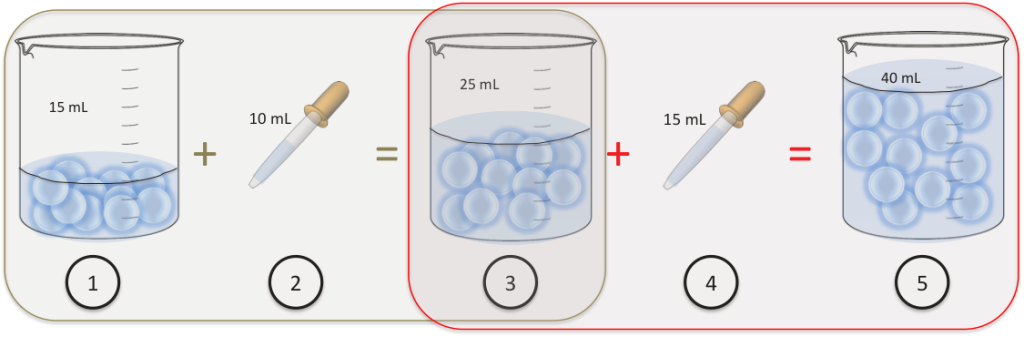
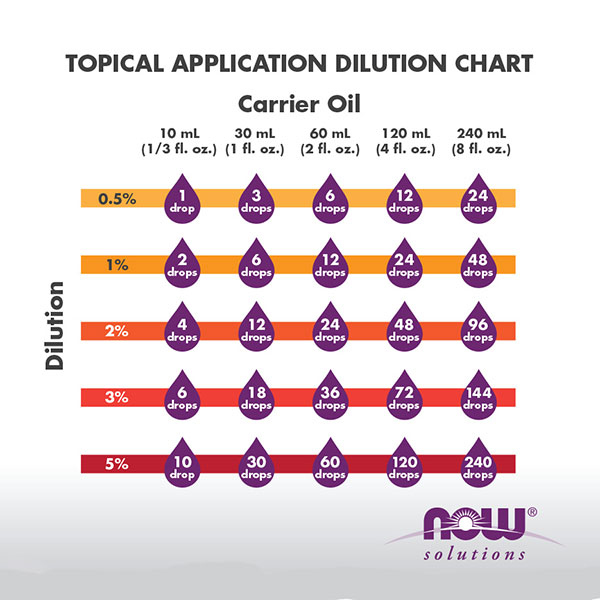
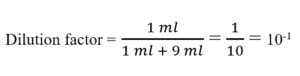dilution factor formula
What is a dilution factor of 100?
You have diluted the sample by a factor of 100. The dilution factor is often used as the denominator of a fraction. For example, a DF of 100 means a 1:100 dilution. How would you make 500 mL of a 1:250 dilution? Pipet 2.00 mL of your stock solution into a 500 mL volumetric flask.
What is the dilution factor equation?
The dilution factor equation describes the relationship between the volume and concentration of the starting solution (sometimes called a stock solution) and the volume and concentration of the dilution you are preparing. It is written as:
What is a 1 20 dilution factor?
A 1:20 dilution factor means that for each unit of the stock solution, there are 19 units of dilutant, resulting in a total volume of 20 units. Note that a 1:20 quotient can represent either an S:D (stock to diluant) or S:T (stock to total solution) ratio. In the context of a dilution factor, it specifically refers to an S:T ratio.
How many units are in a 1 5 dilution?
In a 1:5 dilution, for example, 1 unit volume of solute (the item to be diluted) is combined with (roughly) 4 unit volumes of the solvent to produce five units of total volume. It’s worth noting that some solutions and combinations have a smaller volume than their constituents. What is Dilution? What is the Dilution Factor? What is Dilution?

How to Calculate Dilution Factor

Calculating Dilution Factor

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations
|
JT - Dilution Factor
Dilution Factor: A water sample was tested (with dithizone in methylene chloride to make a color solution) for lead content but was diluted prior to |
|
Laboratory Math II: Solutions and Dilutions
If molarity or normality the molecular or formula dilution factor would be the sum of 100 microliters plus 900 microliters divided by 100 microliters. |
|
Attachment B: Example Engineering and Dilution Factor Calculations
equations based on the use of the receiving water as the applicant's public water supply. Massachusetts: Equation used to calculate the dilution factor at |
|
Brownstock Washing Cost Optimization
Oct 28 2019 Minimal operating cost occurs at minimal wash water usage. Dilution Factor and Washing Efficiency. The dilution factor is the washing parameter ... |
|
Appendix VI: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: Dilution Factor = Use the equations and appropriate factors for each parameter as specified in NH. |
|
Appendix V: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: Use the equations and appropriate factors for each parameter as specified in EPA's. |
|
CRRT Formulas
CRRT DOSE DILUTION FACTOR: When using Pre-Filter Replacement Fluid and/or Pre-Blood-Pump. (PBP) Fluid the CRRT dose is diluted and |
|
Elemental Analysis Manual - Section 3.4 Version 3.1 (December 2021)
calculations formulas do not constitute a “deviation” from the method. Dilution factor (DF)—factor by which the concentration in a diluted analytical ... |
|
Appendix VII: Dilution Factor Calculations for Massachusetts and
Note: The dilution factor in New Hampshire is calculated using two different equations based on the use of the receiving water as the applicant's public |
|
Appendix 8: Dilution Factor and Effluent Limitation Calculations for
I. Dilution Factor The equation used to calculate the dilution factor is: ... Use the equations and appropriate factors as specified in 314 CMR. |
|
Attachment B: Example Engineering and Dilution Factor Calculations
equations based on the use of the receiving water as the applicant's public water supply Massachusetts: Equation used to calculate the dilution factor at the |
|
How to Make Simple Solutions and Dilutions
l Simplc Dilution (Dilution Factor Nlethod based on ratios) A sinple The formula belorv is a quick approach to calculating such dilutions V = yolume, C |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
normality, you will need to know the molecular or formula weight of the substance equal to the stock concentration divided by our dilution factor of ten to the |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
a serial dilution the total dilution factor at any point is the product of the individual Molar (1 0 M) solution is equivalent to 1 formula weight (FW) (g/mole) of |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor, and so on until the final concentration is known Example: A 5M A quick way to do problems of this sort is to use the famous formula: C1 V1 = C2 |
|
261_Labs_Making Solutions and Dilutions - Penguin Prof Pages
The source of dilution material for each step comes from the diluted material of the previous In a serial dilution the total dilution factor at any point is the product of |
|
Dilutions - Weber State University
Making dilutions is one of the most common practices in chemistry Concentrated solutions As seen, this calculation is simply the final solution volume divided |