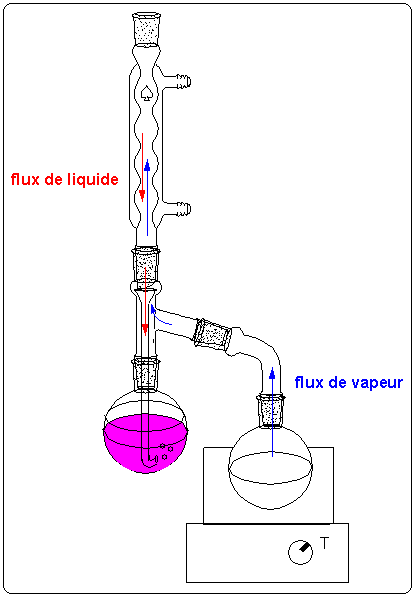
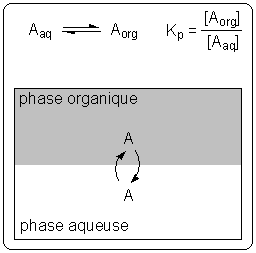
Extraction liquide-liquide. * Deux solvants sont dits miscibles s`ils
What is liquid-liquid extraction (LLE)?
Liquid–liquid extraction (LLE), also known as solvent extraction, has long been an effective method of separating compounds having different solubilities in two immiscible liquids (1). The two liquids are typically water, perhaps with some additives, and a nonpolar organic solvent such as isooctane.
What is solvent extraction?
The term solvent extraction can also refer to the separation of a substance from a mixture by preferentially dissolving that substance in a suitable solvent. In that case, a soluble compound is separated from an insoluble compound or a complex matrix. [not verified in body]
What solvent does not dissolve well in water?
The other solvent is a liquid that does not dissolve very well in water, such as diethyl ether (this is the most common type of ether, and it is often called simply "ether"). If you look closely at a mixture of ether and water, you will see two layers because the two compounds do not dissolve very well in each other.
How do you measure the success of a liquid–liquid extraction column?
Success of liquid–liquid extraction is measured through separation factors and decontamination factors. The best way to understand the success of an extraction column is through the liquid–liquid equilibrium (LLE) data set.

Extraction par solvant 1ère Lycée Physique-chimie Méthode 💡

Solvent extraction or separation