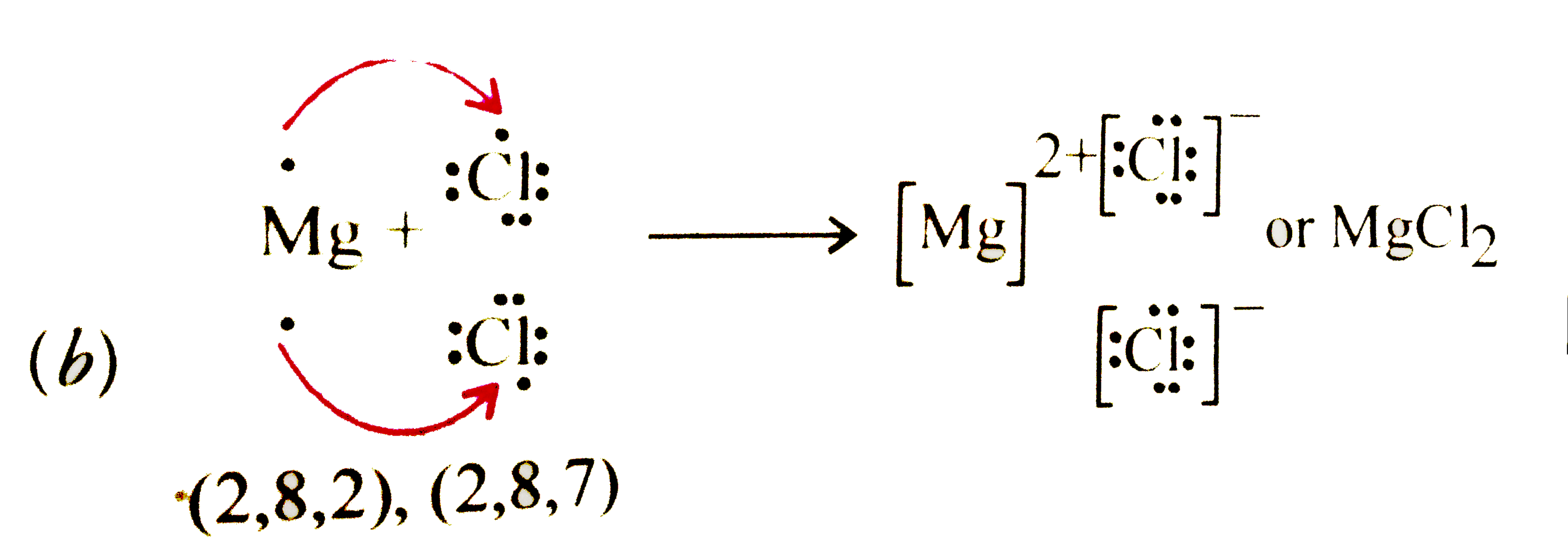electrorefining of copper reaction
What is electrolytic refining of copper for Class 12?
The electrolytic refining method is used to refine metals like copper using the process of electrolysis. In this article, we will learn about electrolytic refining of copper for class 12. What is refining? Electrolytic refining of copper is used to purify the metal using electrolysis.
What metals can be purified by electrorefining?
Electrorefining can be used to purify a number of metals including copper, nickel, cobalt, lead and tin. During the electrorefining stage of copper production, a series of impure copper anodes and thin starter sheet cathodes (such as thin copper foil) are suspended in a tank. The electrolyte containing the copper ions flows through tank.
What Is Electrolytic Refining?
Electrolytic refiningis a process of refining a metal (mainly copper) by the process of electrolysis. As far as the mechanism of the process is concerned, during electrolysis, a large chunk or slab of impure metal is used as the anode, with a thin strip of pure metal as the cathode. In this setup, an electrolyte (metal salt aqueous solution), depen
Electrolytic Refining of Metals
The below-given table outlines the methods used to refine five metals. It is necessary to choose the electrolyte and other conditions so that both anodic dissolution and metal deposition proceed with high efficiency while none of the impurity metals can move from the anode to the cathode. Clearly, there must be no passivation of the anode, and the
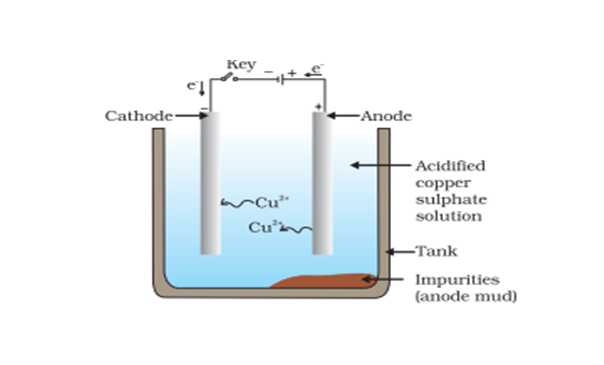
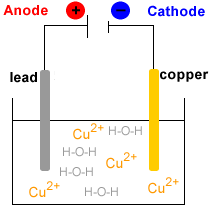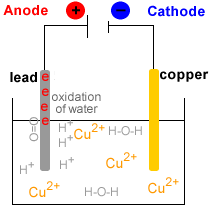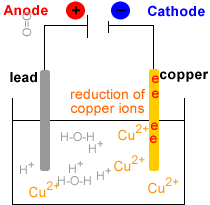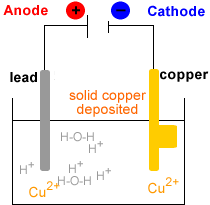
Electrolytic Refining of Copper
Here, we will take an example of electrolytic copper refining to understand the process more clearly. Copper is usually mined from coal, known as blister copper. It is about 98 to 99 per cent pure. However, the electro-refining process can easily make it 99.95% pure, which makes it a good product to be used in electrical components. A block of impu
Refining Process Against Impurities
Grinding and refining play a crucial role throughout metallurgy. Every metal obtained from its ore is typically unclean in nature. Refining is a way to remove impurities and produce high-purity metals. Impurities are extracted by different methods from raw metal depending on material properties and impurities. Some different techniques apart from e

Electrolytic Refining of Copper

Electrolytic Refining of Copper Class 10 ICSE Chemistry Electrolysis Lecture 6C.4

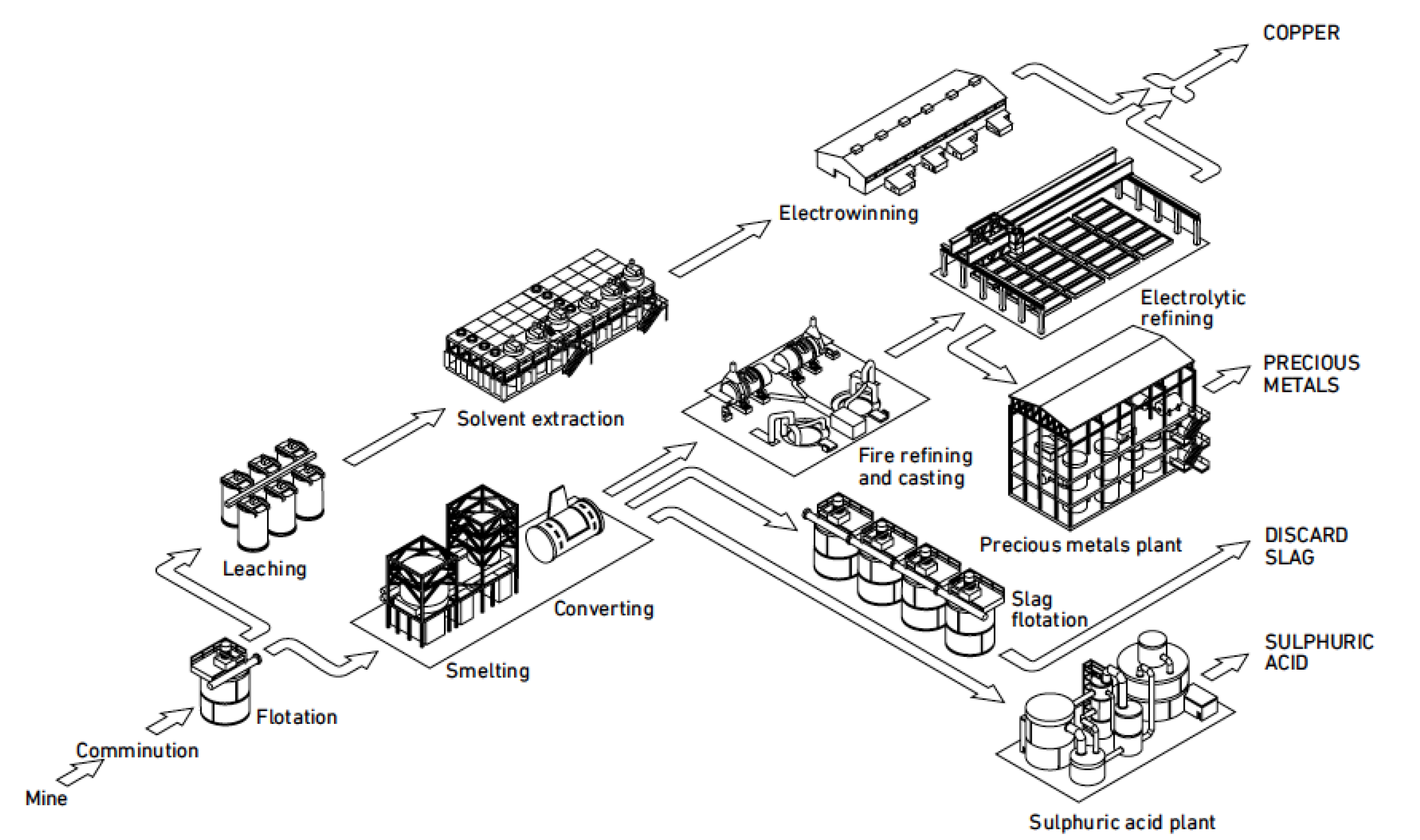
Copper electrowinning for copper refineries
|
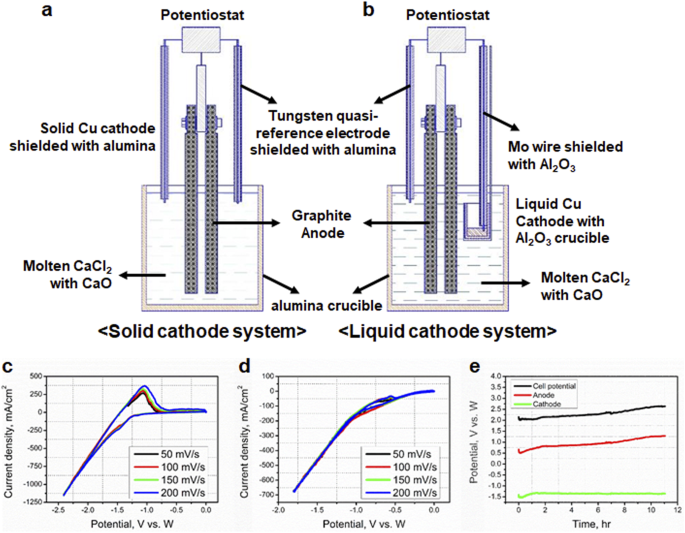
Alternate Anode Reaction for Copper Electrowinning
The conventional copper electrowinning process uses the water hydrolysis reaction as the anodic source of electrons. However this reaction generates acid and |
|
Electrorefining of Copper
i.e. the electrode reactions are at the anode: M ? Mn+ + ne- and at the cathode: Mn+ + ne- ? M. Electrorefining is a much more common process than |
|
Application of Electron Microscopy to the Electrorefining of Copper
6 janv. 1988 BSE image of the anode-electrolyte reaction interface (Kidd Creek). a- copper b-. Cu2Se (grey) + Cu-Pb oxide (bright) + Cu20 cluster(dark) |
|
Copper electrowinning: theoretical and practical design
The fundamental concepts lie in reaction kinetics mass transfer phenomenon |
|
EXTRACTION OF GOLD SILVER AND COPPER FROM THE
main anodic reaction which means the dissolution of the impure copper anodes [6]. The copper electrorefining process can be described by the reactions 1 |
|
S02 Electrowinning in Copper HydrometaIlurgy for Energy
Conventional copper electrowinning involves the decomposition of water at the anode and has a cell potential of approximately 2 V. If the anode reaction is |
|
A mineralogical study of the deportment and reaction of silver during
Reaction of Silver during Copper Electrorefining. T.T. CHEN and J.E. DUTRIZAC. The majority (>95 pct) of the silver in copper anodes occurs in metastable |
|
MODIFIED TITANIUM WHITE FROM MINERAL PROCESSING
Active nitrate ion reduction reactions catalyzed by copper ions are suggested In electrorefining of copper this requires new techniques and new means. |
|
Isolation of Copper from a 5-Cent Coin. An Example of Electrorefining
late copper from a 5-cent coin using electrolysis (an example of the process of electrorefining). reaction for copper is favored (8 9):. |
|
Modelling a copper electrowinning cell based on reactive
dic reaction was copper electrodeposition and the anodic reaction was ferrous to ferric ion Limitations of conventional copper electrowinning cells. |
|
Electrorefining of Copper - ct - ufrgs
i e the electrode reactions are, at the anode: M → Mn+ + ne- and at the cathode: Mn+ + ne- → M Electrorefining is a much more common process than |
|
Fundamental studies of copper anode passivation during - CORE
In copper electrorefining, such de- posits tend to potential-time (E-t) responses in about 1 hour copper electrorefining system, especially with regard to ca- |
|
S02 Electrowinning in Copper HydrometaIlurgy for Energy
Conventional copper electrowinning involves the decomposition of water at the anode and has a cell potential of approximately 2 V If the anode reaction is |
|
Copper electrowinning - SAIMM
Assuming and adequate supply of reactants to the reaction surface (the electrode ) the rate of reaction is described by the Butler- Volmer Equation If the applied |
|
SOLUTION PURIFICATION OF COPPER ELECTROREFINING
Typical copper electrorefining electrolyte contains tens of grams of copper, some cycling of electrodeposition and spontaneous redox replacement reactions |











![PDF] EXTRACTION OF GOLD SILVER AND COPPER FROM THE COPPER PDF] EXTRACTION OF GOLD SILVER AND COPPER FROM THE COPPER](https://ars.els-cdn.com/content/image/3-s2.0-B9780120887965500168-f13-08-9780120887965.gif)