fundamental analytical chemistry calculations
|
Chapter 1 fundamental calculations in analytical chemistry
Fundamental Calculations in Analytical Chemistry. 3 in order to avoid working with very large or very small numbers being advisable to obtain a number |
|
Chapter 4: Calculations Used in Analytical Chemistry
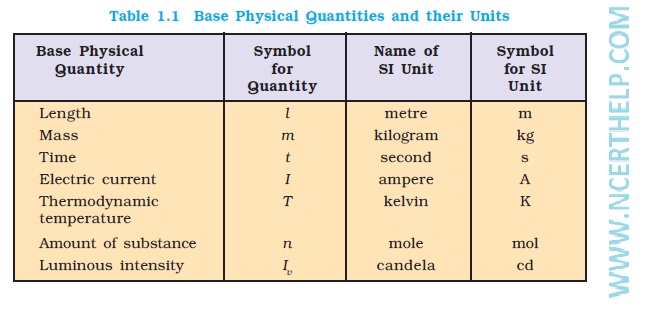
?The International System of Units (SI) is based on 7 fundamental base units. ?Numerous other useful units such as volts |
| Chapter 25: Fundamentals of Analytical Chemistry |
|
Calculations used in Analytical Chemistry
measurement all over the world. Page 4. SI units. SI is based on seven fundamental base units. |
|
Analytical Chemistry 2.1 Solutions Manual
Chapter 2 Basic Tools of Analytical Chemistry. Chapter 2 tion B to calculate the concentration of solution A; thus. Solution A: 0.100 M. 250.0 mL. |
|
CHAPTER 4 Calculations Used in Analytical Chemistry
Calculations Used in Analytical Chemistry Analytical molarity is the total number of moles of a solute ... chemical state |
|
Basic Tools of Analytical Chemistry
In calculating a result the answer can never be more certain than the least certain measurement in the analysis. Rounding answers to the correct number of |
|
Chapter 2
Basic Tools of. Analytical Chemistry. Chapter Overview. 2A Measurements in Analytical Chemistry. 2B Concentration. 2C Stoichiometric Calculations. |
|
Chapter 11 Solving Equilibrium Problems for Complex Systems
Source: slideplayer.com/Fundamentals of Analytical Chemistry F.J. Holler |
|
COURSE SYLLABUS for CHEM 21 : ANALYTICAL CHEMISTRY 1
analytical chemistry stoichiometric calculations with the general Learn the basic analytical concepts and theories with focus on data. |
|
Chapter 1 fundamental calculations in analytical chemistry
OBJECTIVES AND SCOPE This chapter summarizes the units and the expressions of concen- trations employed more often in the calculations associated to the |
|
Chapter 4: Calculations Used in Analytical Chemistry
In this chapter we describe several methods used to compute the results of a quantitative analysis • SI system of units and the distinction between mass |
|
Calculations used in Analytical Chemistry
An empirical formula gives the simplest whole number ratio of atoms of each element present in a molecule or chemical compound Eg CH is empirical formula of |
|
CHAPTER 4 Calculations Used in Analytical Chemistry
CHAPTER 4 Calculations Used in Analytical Chemistry In this chapter we describe several methods used to compute the results of a quantitative analysis |
|
(PDF) Fundamentals of Analytical Chemistry- 9th Edition Yasin ?enol
Fundamentals of Analytical Chemistry- 9th Edition Download Free PDF Calculations of pH and of titration curves are also described |
|
Solutions Manual Fundamentals of Analytical Chemistry 9th Edition
Solutions Manual Fundamentals of Analytical Chemistry 9th Edition (a) SQRT returns the square root of a number or result of a calculation |
|
CALCULATIONS OF ANALYTICAL CHEMISTRY HandoutsEt
Quantitative Chemical Analysis to Calculations of Analytical Chem- are understood than to give practice in fundamental mathematical operations |
|
Fundamentals of Analytical Chemistry - PersianGig
Student Solutions Manual Prepared by Calculations Used in Analytical Chemistry Fundamentals of Analytical Chemistry: 9th ed Chapter 3 Chapter 3 |
|
Analytical Chemistry
(2) “Fundamental of Analytical Chemistry” by Doglas A Skooge Donald M West Example (2):-Calculate the number of moles in 500 mg Na2WO4 Solution: |
|
Analytical Chemistry 21 Solutions Manual
Solutions Manual to Analytical Chemistry 2 1 by David Harvey (Summer 2016) to use it to complete these most basic of statistical calculations |
|
Chapter 4: Calculations Used in Analytical Chemistry - İYTE
Chapter 4: Calculations Used in Analytical Chemistry ➢The International System of Units (SI) is based on 7 fundamental base units ➢Numerous other useful |
|
CHAPTER 4 Calculations Used in Analytical Chemistry
Calculations Used in Analytical Chemistry In this chapter, we describe several methods 4A-5 Calculating the amount of a substance in moles or millimoles |
|
Analytical Chemistry 21 Solutions Manual
Chapter 2 Basic Tools of Analytical Chemistry Chapter 2 1 (b) To calculate the mass and volume of solution we begin with the molarity calculated in part (a) |
|
Basic Tools of Analytical Chemistry
Chapter 2 Basic Tools of Analytical Chemistry Chapter Overview 2A Measurements in Analytical Chemistry 2B Concentration 2C Stoichiometric Calculations |
|
Errata Student Solution Manual for Fundamentals of Analytical
Fundamentals of Analytical Chemistry: 9 th ed Chapter 3 Chapter 3 3-1 (a) SQRT returns the square root of a number or result of a calculation (b) AVERAGE |
|
Modern Analytical Chemistry - Focus User Management
In a quantitative analysis, we measure a signal and calculate the amount of the basic tools of analytical chemistry, including significant figures, units, and stoi- |
|
45 Quantitative Chemical Analysis
Describe the fundamental aspects of titrations and gravimetric analysis • Perform stoichiometric calculations using typical titration and gravimetric data |
|
Chapter 25: Fundamentals of Analytical Chemistry
Therefore, basic concepts of chemical measurement discussed in Sections 1 8– 1 13 must be mastered before studying analytical chemistry Calculating and |
|
Skoog - Fundamentals of Analytical Chemistry 8e - UCI Chemistry
of Analytical Chemistry to Solve a Problem Chapter 6 Random Errors in Chemical Analysis 105 Feature 6-2 Calculating the Areas under the Gaussian |













![PDF REVIEW] Analytical Chemistry: A Practical Guide FREE REGISTER PDF REVIEW] Analytical Chemistry: A Practical Guide FREE REGISTER](https://d1whtlypfis84e.cloudfront.net/guides/wp-content/uploads/2019/06/19095224/NCERT-Solutions-for-Class-11-Chemistry.png)



