gibbs free energy
|
Standard Thermodynamic Values
Gibbs Free Energy. (kJ/mol). (NH4)2O. (l). -430.70096. 267.52496. -267.10656. (NH4)2SiF6. (s hexagonal). -2681.69296. 280.24432. -2365.54992. (NH4)2SO4. |
|
Physics 3700 Free Energy. Thermodynamic Identities. Phase
Here we introduce two: the Helmholtz free energy and the Gibbs free energy. Just as the enthalpy can be used to characterize heat the free. |
|
Increasing temperature increases disorder because the entropy
The difference between the energy and enthalpy changes in expanding an ideal gas. (b) What is the Gibbs free energy change? (a). ?S = ? dqrev. |
|
Appendix A: Tables Showing Gibbs Free Energy as a Function of
Free Energy as a Function of Temperature of Formation Reactions. Table A.1 Gibbs free energy of methane formation at differ- ent temperatures (Adapted from |
|
Gibbs Free Energy and Chemical Equilibrium
22-Jan-2013 A criterion for equilibrium is that the total free energy. (Gibbs free energy G r. ) of the reaction is at a minimum:. |
|
Droplet Formation Gibbs Free Energy of a droplet as
We let the Gibbs free energy per unit mass of the vapor be GV and of the droplet Gd. Next we make use of a standard 'trick' in lots of areas of physics and |
|
Lecture 8: Free energy
calculation con rms that for a monatomic ideal gas the chemical potential is the Gibbs free energy per molecule. Note that we found that G = N for the ideal gas |
|
Free Energy Calculation in MD Simulation
Helmoholtz free energy. A = U – TS + ? ? i. N i. dA = w rev. (reversible const N V T) eq (22.9) McQuarrie & Simon. ? Gibbs free energy. |
|
Why passive diffusion happens – free energy considerations
Gibbs free energy G |
|
Gibbs Free Energy of Protein-Protein Interactions reflects tumor stage
13-Jul-2015 thermodynamic measure (known as Gibbs free energy) of protein-protein interaction networks for several cancer types and 5-year overall ... |
|
Thermodynamic Laws & Gibbs Free Energy - SOEST Hawaii
17 jan 2012 · G -- Gibbs free energy – H -- enthalpy Page 2 1/17/12 2 Intensive properties Internal energy of system is increased by gaining heat |
|
Gibbs free energy
27 oct 2011 · In thermodynamics the Gibbs free energy (IUPAC recommended name: Gibbs energy or Gibbs function; also known as free enthalpy[1] to |
|
Gibbs Free Energy
Gibbs free energy is a measure of chemical energy All chemical systems tend naturally toward states of minimum Gibbs free energy |
|
Lecture 8: Free energy
At constant pressure and constant temperature Gibbs free energy has its minimum value at equilibrium A powerful function of the Gibbs free energy is that it |
|
Chapter 14 (and 154): Entropy and Free Energy
Chapter 14 (and 15 4): Entropy and Free Energy Key topics: Spontaneous processes Entropy S Gibbs free energy G Begin with a review of some key ideas |
|
Chemistry 130 Gibbs Free Energy
Gibbs Free Energy Dr John F C Turner the change in internal energy U which involves heat transferring in or out of the system only |
|
Gibbs Energy and Thermodynamics - Shivaji College
Every chemical reaction involves a change in free energy called delta G (?G) • To calculate ?G subtract the amount of energy |
|
Chemical Thermodynamics SYBSc Concept of Gibbs free energy
8) The Gibb's free energy change ?G is related to enthalpy change ?H Equation (11) is called Gibbs – Helmholtz equation The term |
|
185: FREE ENERGY - Esalq
To understand the relationship between Gibbs free energy and work One of the major goals of chemical thermodynamics is to establish criteria for predicting |
|
Enthalpy Entropy and Free Energy
energy; entropy (?S); Gibb's free energy (?G); spontaneous; state function Refers to all of the energy contained within a chemical system |
|
A method of estimating the Gibbs free energies of - Horizon IRD
solubility products and Gibbs free energies of formation for ( 1 ) poorly crystallized, hydrated clay minerals and (2 11 for poorly crystallized, dehydrated clay |
|
Gibbs free energy of formation of hydrated and - Horizon IRD
GIBBS FREE ENERGY OF FORMATION OF HYDRATED AND DEHYDRATED CLAY MINERALS TARDY Y Institut Francais de Recherche Scientifique pour le |
|
Standard Gibbs Free Energy, Enthalpy, and Entropy Changes as a
The standard Gibbs free energy change AGZ,,, number (nn) of moles of H+ produced, number (nMg) of moles of Mg2+ produced, standard enthalpy change |
|
Tables Showing Gibbs Free Energy as a Function of Temperature of
Table A 1 Gibbs free energy of methane formation at differ- ent temperatures ( Adapted from David 2012) C + 2H2 CH4 T (K) ΔG (kJ/mol) 298 15 А50 53 300 |
|
Gibbs Free Energy
Gibbs Free Energy Equation • ΔG = ΔH -TΔS • If ΔG is negative (< 0) the reaction is favorable or spontaneous • If ΔG is positive (> 0) the reaction is unfavorable |


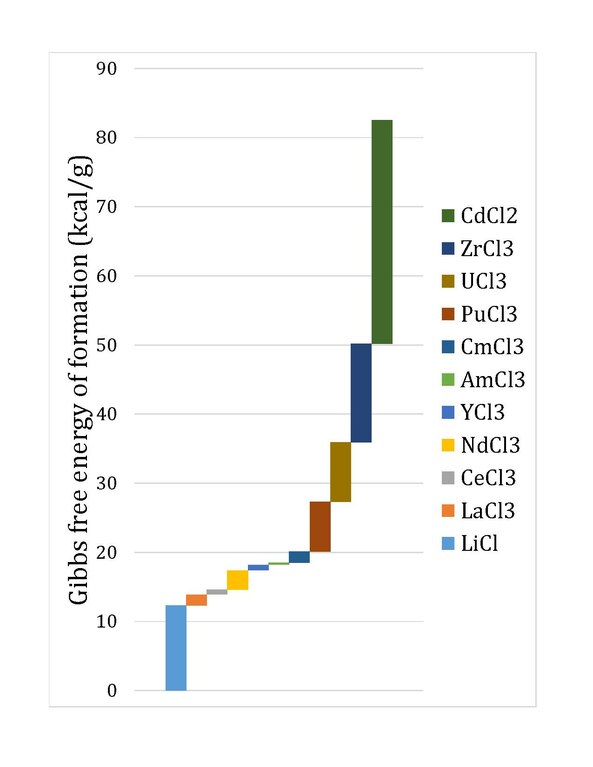
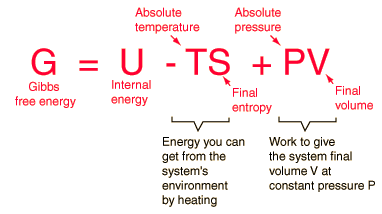














![PDF] Standard Gibbs free energy enthalpy and entropy changes as PDF] Standard Gibbs free energy enthalpy and entropy changes as](http://image.slidesharecdn.com/tang05-entropyandgibbsfreeenergy-120209095951-phpapp01/95/tang-05-entropy-and-gibbs-free-energy-13-638.jpg?cb\u003d1422605471)




