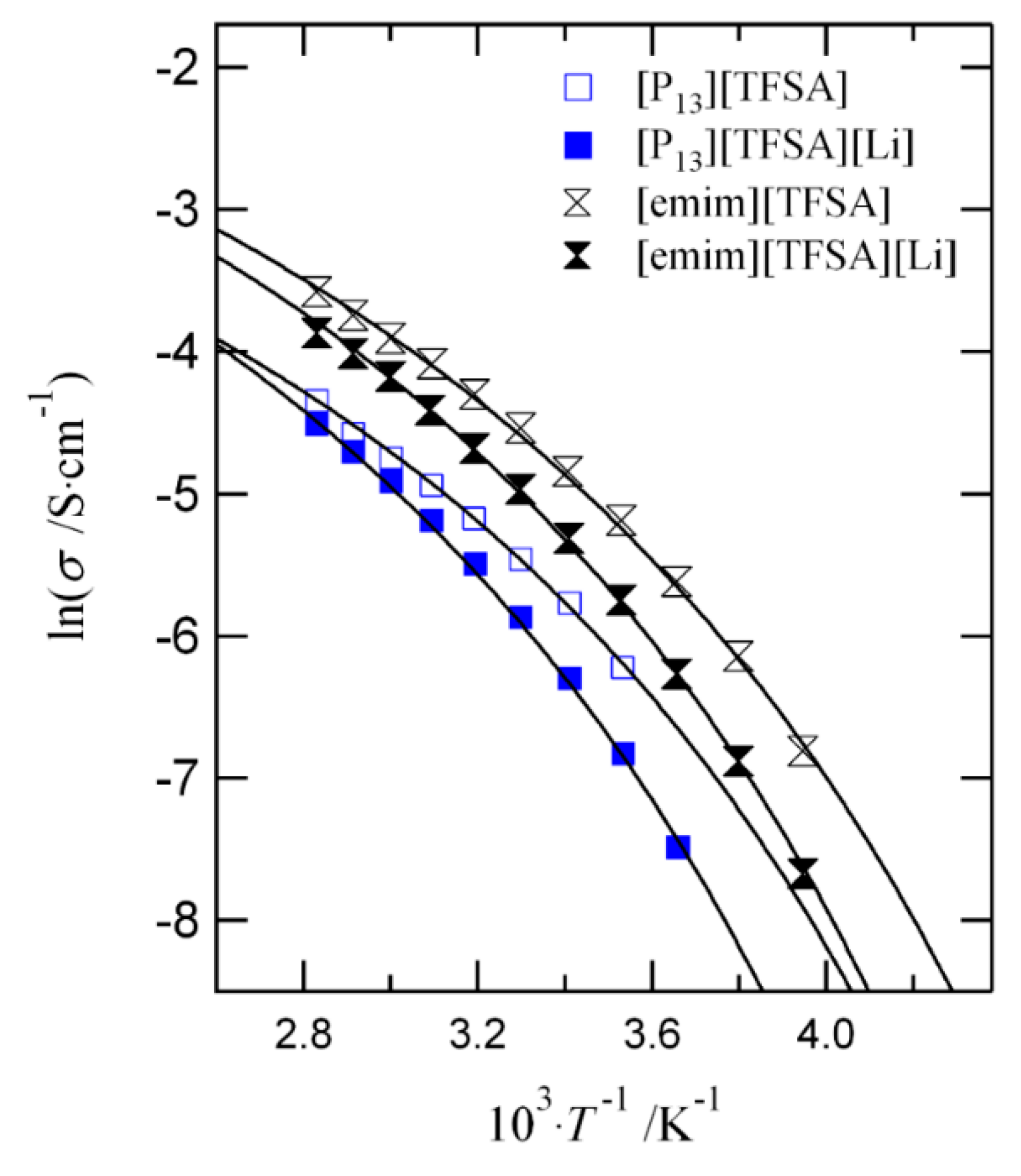
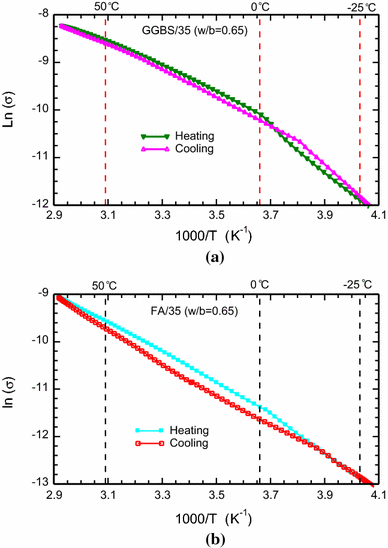
arrhenius equation conductivity
|
An Arrhenius Argument to Explain Electrical Conductivity Maxima
A density-dependent Arrhenius equation fit the experimental conductivities splendidly and could also explain the results of two simulations under “off-diagonal |
|
S1 Arrhenius equation
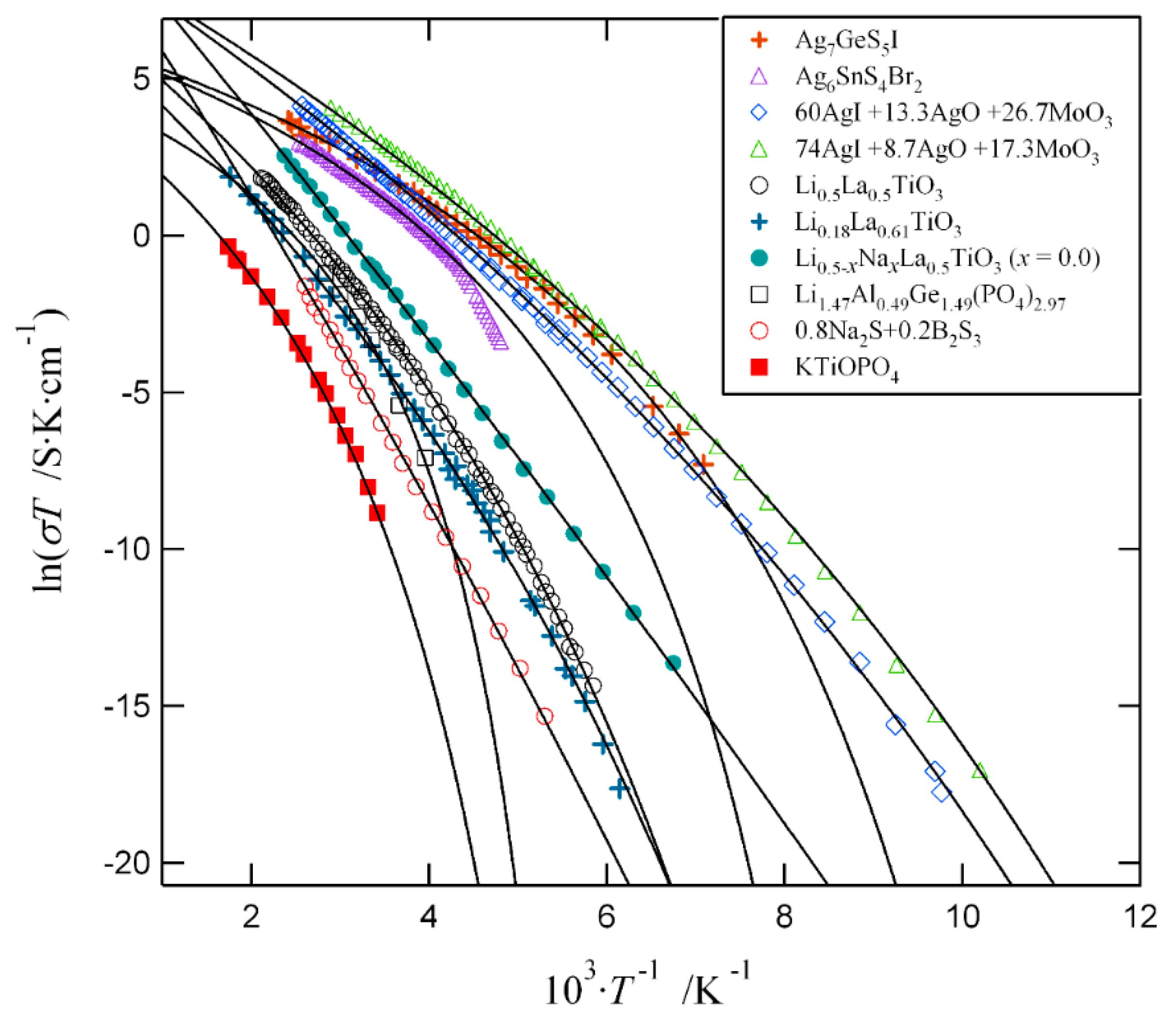
S1 Arrhenius equation Describes the conductivity for a thermally activated process1: (S1) = 0 (‒ ∆ ) σ – conductivity σ0 – |
|
Variable Range Hopping and Thermal Activation Conduction of Y
Arrhenius equation σ(T)=σ0exp[-(Ea/kT)] which shows the thermal activation conduction The activation energy Ea increases from 0 47 meV for ZnO film to |
What does the Arrhenius equation tell us?
The Arrhenius equation can be used to determine the effect of a change of temperature on the rate constant, and consequently on the rate of the reaction.
What is the Arrhenius equation for the diffusion coefficient?
Temperature dependence of this escape rate or diffusion coefficient is given by Van't Hoff–Arrhenius–Kramers formula [3], [4], D ( T ) = D 0 e ( − E a ∕ k B T ) This equation was first proposed by Van't Hoff and Arrhenius and later derived by Kramers by solving the Fokker–Planck equation for Brownian motion in phase
What is K in Arrhenius equation?
k is the rate constant (frequency of collisions resulting in a reaction), T is the absolute temperature, A is the pre-exponential factor or Arrhenius factor or frequency factor.
Arrhenius originally considered A to be a temperature-independent constant for each chemical reaction.5) the average value of the activation energy for conduction is calculated to be 1.15 eV which leads, using eq. [l], to a calculated "band gap" of 2.3 & 0.1 eV.
|
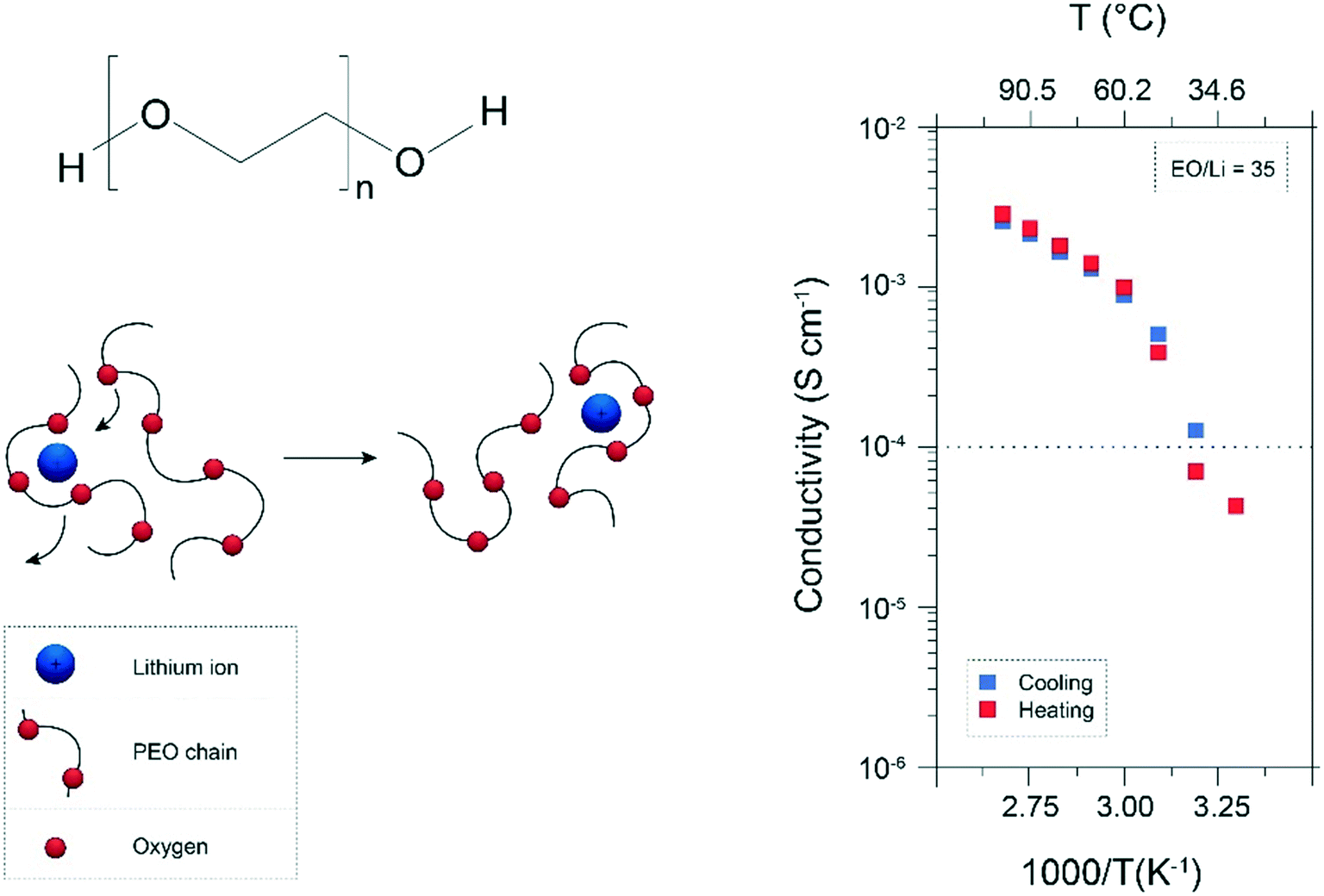
Temperature-dependent ionic conductivity and transport properties
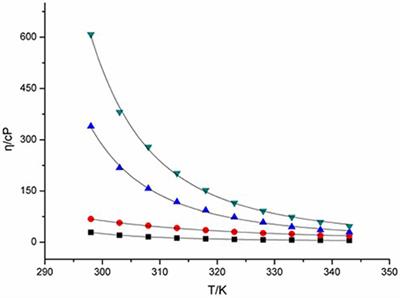
ionic conductivity and mobility of the carrier ions, doping the higher concentration of salts tion of the Tg with activation energy and fragility were studied using |
|
Ionic conductors
µ = µ0 exp (-Ea/kT) - Arrhenius equation Temperature dependence of conductivity σ = (σ0 /T) exp(-Ea/kT) – Contribution from mobility and defect formation |
|
CHAPTER-6 ELECTRICAL CONDUCTIVITY - ResearchGate
of electrical conductivity with temperature is explained by the equation Where Ea and Eb are the activation energy for the intrinsic and extrinsic conduction |
|
Ionic Conductivity and Solid Electrolytes
commonly expressed by an Arrhenius equation: (2) •Where Q is the activation energy , Do is a pre- exponential factor that depends on the vibrational frequency |