arrhenius equation example
|
The Arrhenius Equation Practice
The Arrhenius Equation Practice Directions: Complete the following problems and show all of your work! Put a box around your final answers to each problem |
|
The Arrhenius Equations Questions
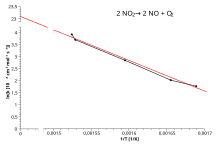
The decomposition of N2O5 at 308K has a rate constant of 1 35x10-4 s¹ The Arrhenius constant for this reaction is 4 79x1013 s¹ Calculate the activation |
|
Reaction Rates and Temperature; Arrhenius Theory
Arrhenius Equation Example ◇ If a reaction has an activation energy of 50 kJ/ mol then how much should the rate of the reaction accelerate if the |
|
Arrhenius equation
(a) Zero activation energy = Fraction of effective collision (f) will be very large = Very fast reaction (Instantaneous reaction) (b) Low activation energies = |
What is the basic Arrhenius equation?
Using this straight line, which is referred to as an Arrhenius plot, Arrhenius created the equation: ln(k) = (-Ea/R)(1/T)+ln(A), where ln(k) is the y variable, 1/T is the x variable, -Ea/R is the slope, and ln(A) is the y-intercept.
How do you write the Arrhenius equation?
Note: The Arrhenius equation is sometimes expressed as k = Ae-E/RT where k is the rate of chemical reaction, A is a constant depending on the chemicals involved, E is the activation energy, R is the universal gas constant, and T is the temperature.
Determining Activation Energy.
Notice that when the Arrhenius equation is rearranged as above it is a linear equation with the form y = mx + b; y is ln(k), x is 1/T, and m is -Ea/R.
The activation energy for the reaction can be determined by finding the slope of the line.
What is Arrhenius theory in chemical kinetics?
The Arrhenius law predicts that the motion of the structural units (atoms, molecules, ions, etc.) should slow down at a slower rate through the glass transition than is experimentally observed.
In other words, the structural units slow down at a faster rate than is predicted by the Arrhenius law.
|
Chemistry 146 Lecture Problems Arrhenius Equation
7/3/01. Chemistry 146 Lecture Problems. Arrhenius Equation. The Arrhenius Equation k Ae. E a. R T. . From Example 21.8 in Kask and Rawn. |
|
Notes and Example Calculations - Energetics - OCR (A) Chemistry A
Use the Arrhenius equation to calculate the pre-exponential factor at 290K if the activation energy was 55.5 kJmol?-1?. Step 1:? Decide which form of the |
|
Reaction Rates and Temperature; Arrhenius Theory Arrhenius
Arrhenius Equation Example. ? If a reaction has an activation energy of 50 kJ/ mol |
|
Reaction Rates and Temperature; Arrhenius Theory Arrhenius Theory
Arrhenius Equation Example. ? If a reaction has an activation energy of 50 kJ/ mol |
|
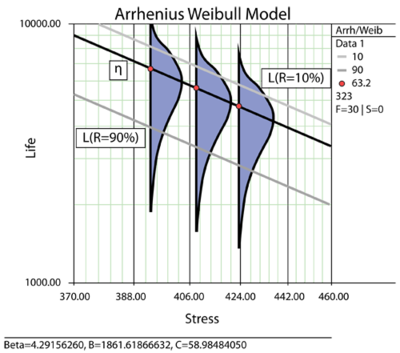
Calculating Reliability using FIT & MTTF: Arrhenius HTOL Model
Jan 9 2012 applicable equations |
|
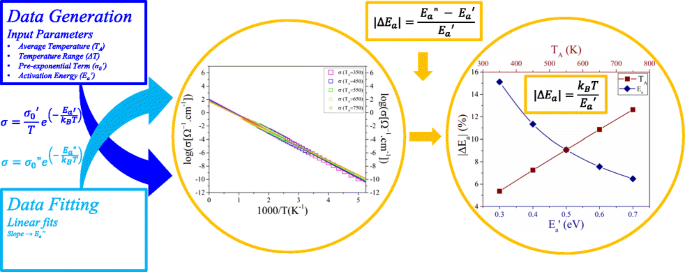
Evaluating the Simple Arrhenius Equation for the Temperature
Jul 18 2020 Figure 2: Apparent activation energies vary significantly between developmental stages. A) Two examples of developmental time intervals in D. |
|
Evaluating the Simple Arrhenius Equation for the Temperature
Jul 18 2020 The famous Arrhenius equation is well motivated to describe the temperature ... Figure 2A |
|
Lecture 22: The Arrhenius Equation and reaction mechanisms. As
For example in doing kinetics |
|
A Theoretical Justification for the Application of the Arrhenius
Although the Arrhenius equation has been widely and successfully applied to innu- homogeneous processes are (with few exceptions; see for example |
|
USPTO - July 2015 Update Appendix 1: Examples
Jul 1 2015 examples shows how claims should be analyzed under the 2014 IEG. ... The Arrhenius equation has long been used to calculate the cure time in ... |
|
Chemistry 146 Lecture Problems Arrhenius Equation
3 juil 2001 · The Arrhenius Equation k Ae E a R T From Example 21 8 in Kask and Rawn Calculating the activation energy from the rate constant at two |
|
Reaction Rates and Temperature; Arrhenius Theory Arrhenius Theory
is the activation energy R is the ideal-gas constant constant increases according to the Arrhenius Equation Rate (M s-1) = k Arrhenius Equation, Example |
|
The Arrhenius Equation Answers - Science Skool
The Arrhenius Equations Questions 35420* s spog Calculate the activation energy of this reaction (R=8 31 JK-moll) table, for example ① Isok A= k A= 4 59 |
|
Evaluating the Simple Arrhenius Equation for the - bioRxiv
18 juil 2020 · The famous Arrhenius equation is well motivated to describe the temperature ( example seen in fly Ea = 87 kJ/mol, Ea = 54 kJ/mol, p-value of |
|
AP* Chemistry Arrhenius Made Easy
the activation energy, which must be overcome to produce a chemical reaction Graphing Calculator Tutorial (Not intended to insult your intelligence ) |
|
Evaluating the Simple Arrhenius Equation for the Temperature
18 juil 2020 · Arrhenius Equation, Temperature Dependence, Xenopus laevis, Drosophila ( example seen in fly Ea = 87 kJ/mol, Ea = 54 kJ/mol, p-value of |
|
The development of the Arrhenius equation - ntc see result
k = A~-EIRT (1) where R is the gas constant and E is the activation energy The of a numher of reactions in solution, and in a paper published in 1885 (2 5) he |
|
The Arrhenius Equation and reaction mechanisms As we wrap up
For example, in doing kinetics, a couple important pieces of information are the rate constant, k, and the amount of starting material in a reaction, [Ao] Wouldn't it |