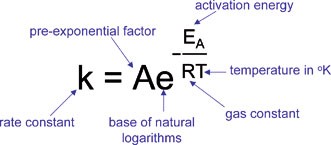
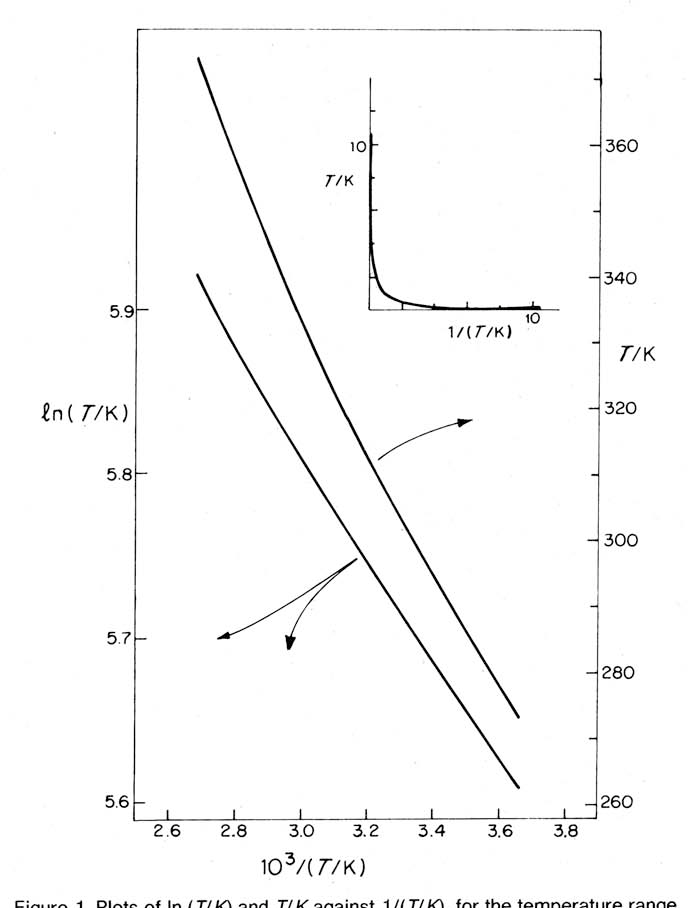
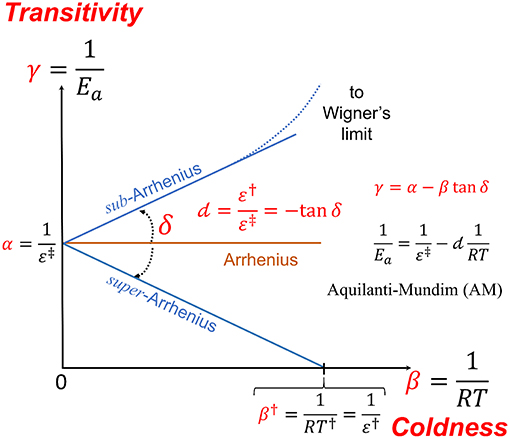
arrhenius equation ln
|
Chemical Kinetics (Note -2) The Arrhenius Equation
Since ln(A) is a constant the equation corresponds to that of a straight line (y = mx + c) whose slope (m) is -Ea/R When the logarithm of the rate constant ( |
|
Chemistry 146 Lecture Problems Arrhenius Equation
3 juil 2001 · The Arrhenius Equation k Ae E a R T Calculate the change in the rate constant at different temperatures (for a known activation energy) |
|
The Arrhenius equation indicates the relationship between the
The Arrhenius equation indicates the relationship between the reaction temperature Therefore Equation 4 can be expressed as follows in Equation 6 The |
|
Arrhenius Equation /
Arrhenius Equation (Fall-2015) Arrhenius Equation Arrhenius equation happens to be one of the most important equations in physical chemistry The equation |
|
Arrhenius equation
Calculate the energy of activation in calories (R = 1 987 cal degree-1 mol-1) Solution: or or = |
Can the Arrhenius equation be written as LNK?
Often the equation is written in logarithmic form lnk=lnA – E a/RT A graph of lnk against 1/T is a straight line with a gradient –E a/R and an intercept on the lnk axis of lnA.
It is named after Svante Arrhenius.What is ln in Arrhenius equation?
Using this straight line, which is referred to as an Arrhenius plot, Arrhenius created the equation: ln(k) = (-Ea/R)(1/T)+ln(A), where ln(k) is the y variable, 1/T is the x variable, -Ea/R is the slope, and ln(A) is the y-intercept.
The Arrhenius law predicts that the motion of the structural units (atoms, molecules, ions, etc.) should slow down at a slower rate through the glass transition than is experimentally observed.
In other words, the structural units slow down at a faster rate than is predicted by the Arrhenius law.
|
Chemical Kinetics (Note -2) The Arrhenius Equation: The Arrhenius
Arrhenius Plot. When logarithms are taken on both sides of the equation the Arrhenius equation can be written as follows: ln k = ln(Ae-Ea/RT). |
|
Loi de vitesse dune réaction chimique
L'équation d'Arrhenius permet de déterminer l'énergie d'activation d'une Pour x = 0 on a y = b = Ln(A) et la pente de la droite |
|
A Study on the Modified Arrhenius Equation Using the Oxygen
14 janv. 2019 In general reactions the linear Arrhenius equation is used to ... generally expressed as Equation (2). ?. dP dt. = kP |
|
Modified Arrhenius Equation in Materials Science Chemistry and
26 nov. 2021 According to the Arrhenius equation the reaction rates at very low temperatures are very small ... singer equation into the form: 2 ln ln. |
|
Polymers/Organic Materials Aging Overview
ln(k) = ln(A) – Ea/RT. Empirical equation. Arrhenius equation: Gillen K. T.; Celina |
|
17 3. Rate and equilibrium constants and free energies. (a) Energy
because the Arrhenius equation makes ln (k) linear in (1/T) while the Eyring equation does not. At some temperature T in the middle of the range studied we |
|
Determination of the joint uncertainty domain of rate parameters:
Calculation of the variance of ln k from uncertainty factor f. (assuming 3? deviation between Matrix-vector form of the linearized Arrhenius equation:. |
|
The Arrhenius Equation Revisited
The Arrhenius equation has been widely used as a model of the temperature effect on the rate The linearity of the Arrhenius plot that is |
|
Test of the formal basis of Arrhenius law with heat capacities
7 mai 2018 ing that the Arrhenius equation should be grounded on ... entropy equation. dS. dU. = 1. T. (11) yields. kB h? ln. |
|
Arrhenius Equation
ln k = -Ea/RT + ln A …………..(II). Therefore from the Arrhenius equation |
|
01 - Loi dArrhenius - Chimie - PCSI
On peut donc conclure que les points sont bien alignés : Les résultats de cette expérience suivent la loi d'Arrhenius La loi ln = ln − ( |
|
Arrhenius Equation
Arrhenius Equation The rate Arrhenius equation Van't Hoff ln( k1/k)=Ea/R*( 1/T-1/T1) for a reaction taking place at T and T1, temperatures References: 1 |
|
The Arrhenius Equation and reaction mechanisms As we wrap up
Answer: The ln [C] plot which corresponds to the first order integrated rate law equation So it must be true that this is the data for a first order reaction and I can |
|
The Arrhenius equation indicates the relationship between the
The Arrhenius equation indicates the relationship between the reaction ln ln One straight line is obtained by plotting lnk against 1/T after measuring the |
|
ER Cinétique Chimique — Loi dArrhénius
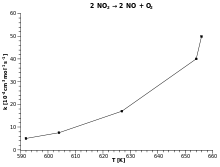
ER Cinétique Chimique — Loi d'Arrhénius La réaction 2N2O5 240 10−5 ln(k) −10,97 −9,62 −7,20 −6,03 • La loi d'Arrhénius s'écrit : k = A exp ( − ℰа |
|
Chemistry 146 Lecture Problems Arrhenius Equation
3 juil 2001 · This has the form y=mx + b where x axis is 1/T y axis is ln(k) y intercept is ln(A) slope is -Ea/R Calcuate the activation energy and the |
|
(a) Energy relationships, temperature dependence For an
because the Arrhenius equation makes ln (k) linear in (1/T) while the Eyring equation does not At some temperature T in the middle of the range studied we |
|
31-1 WEEK 11 Lecture, 31 Rate and Temperature Arrhenius ln k 1/T
What does it mean if a plot of Rn( ) plotted against 1/T gives a negative slope? What sort of algebraic equation does this imply? Arrhenius supplied in the answer |
|
Reaction Rates and Temperature; Arrhenius Theory Energy profile
Arrhenius Equation ◇ Arrhenius noted that reaction rates could be understood to depend on ln(k 2/k 1) = - (Ea/R)[(1/T2) - (1/T1)] Arrhenius Eqn , Graphical |