h3c functional group
|
Functional Groups
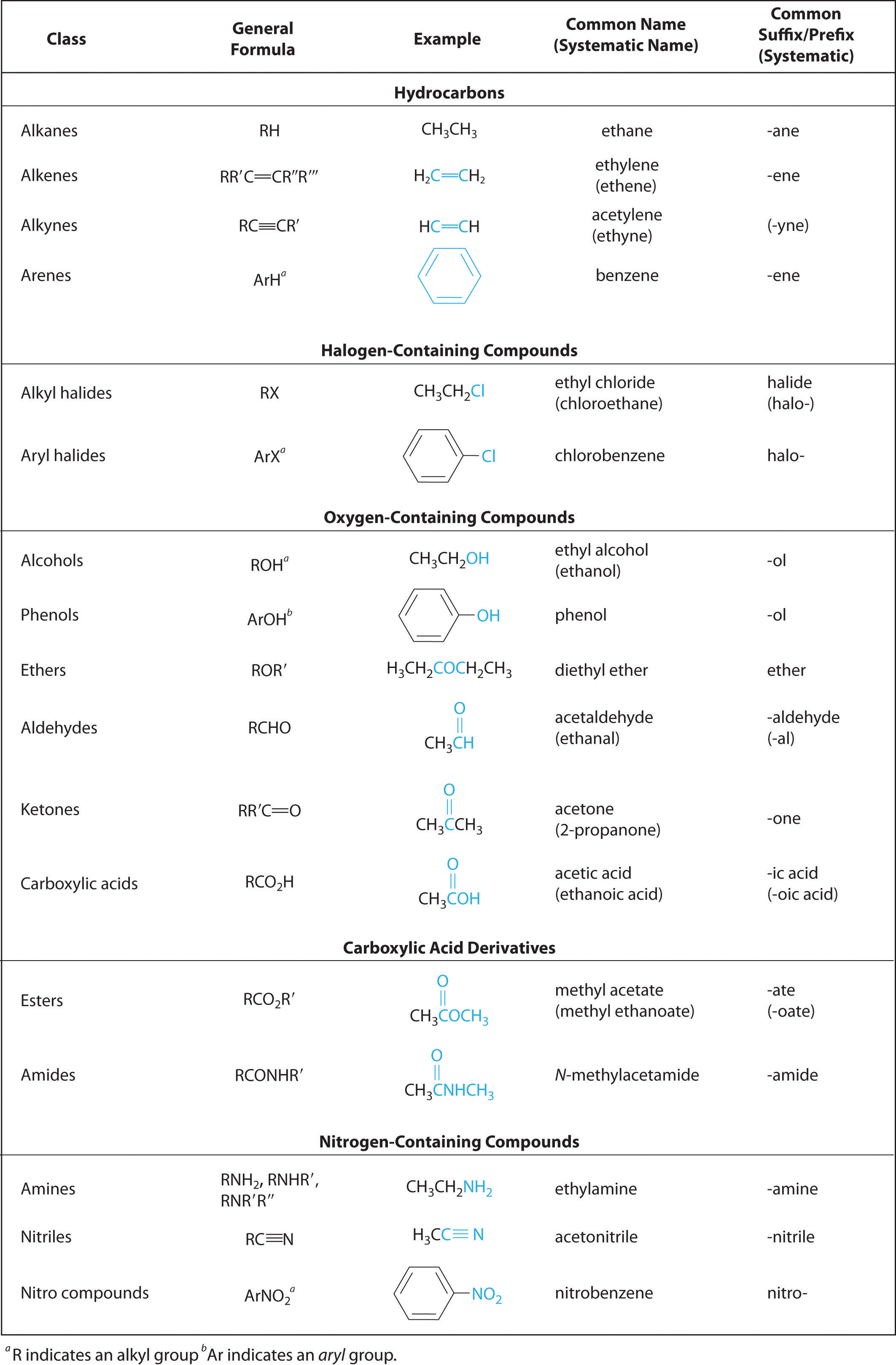
A functional group is a defined grouping of atoms in an organic molecule. Functional Group –. General Structure. Example. C. H. O. Aldehyde. H3C. |
|
H3C CH3 C C C C H CH3 H3C
Functional Groups to Know and Love. Family Name. Functional group structure. Simple example. Alkane. Contains only C-H and C-C single bonds. H3C CH3. |
|
Classification of Organic Compounds: Functional Groups Chemistry
H3C. CH2OH. H3C. OCH3. H3C. CH2Br. C alkane alkene alkyne alcohol ether alkyl halide. Classification of Organic Compounds: Functional Groups. |
|
Classification of Organic Compounds: Functional Groups Chemistry
H3C. CH2OH. H3C. OCH3. H3C. CH2Br. C alkane alkene alkyne alcohol ether alkyl halide. Classification of Organic Compounds: Functional Groups. |
|
Generic Functional Group Pattern Specific Example Using Suffix
z: ilesclassesÌÌ Functional Groups.doc. Generic Functional Group Pattern H3C. C. O. O carboxylic acid suffix: -oic acid prefix: #-carboxy. |
|
Handout: Families of Organic Compounds FAMILY FUNCT GROUP
FUNCT GROUP. EXAMPLE. SUFFIX. Hydrocarbons Alkane or. (only single bonds) H3C Cl. C O H. H3C OH. C O C. H3C O CH3. C N. H3C NH2. |
|
Functional group Example pKa Conjugate Base Alkane H3C CH3
Functional group. Example. pKa. Conjugate Base. Alkane. H3C. CH3. ~50. H3C. CH2. Alkene. H. ~43. Amine. ~35. Sulfoxide. H3C. S. CH3. O. 31. H3C. |
|
Systematic Nomenclature (IUPAC System)
Suffix- functional groups. Naming Alkanes H3C. CH2. CH3 branch pts. at carbons 3 6 |
|
1 Chapter 14: Organometallic Compounds - Reagents with carbon
(H3C)2Cu- Li+. Dimethylcopper lithium Reactive functional groups: ... The solvent or alkyl halides can not contain functional groups. |
|
Chapter 17: Alcohols and Phenols
H3C. _. 1) 2 H3CMgBr. 2) H3O+. CH3. HO CH3. OCH2CH3. O CH3. _. CH3. O. H3CMgBr the H3O+. MgX. Some functional groups are incompatible with Grignard reagents. |
|
Classification of Organic Compounds: Functional Groups - CSUN
Classification of Organic Compounds: Functional Groups H Chemistry 235 H3C OCH3 O aldehyde ketone carboxylic acid acid chloride ester H3C |
|
Functional Groups
A functional group is a defined grouping of atoms in an organic molecule Functional Group – General Structure Example C H O Aldehyde H3C |
|
H3C CH3
Functional Groups to Know and Love Family Name Functional group structure Simple example Alkane Contains only C-H and C-C single bonds H3C CH3 |
|
Handout: Families of Organic Compounds
Fundamentals of General Organic Biol Chem Handout: Families of Organic Compounds FAMILY FUNCT GROUP EXAMPLE SUFFIX Hydrocarbons Alkane |
|
Functional Groups
functional group: an atom or group of atoms (with specific connectivity) exhibiting identical chemical reactivity regardless of the molecule containing it |
|
10 Functional Groups of Organic Compounds
Important information about organic compounds and their functional groups can deprotonated forms (? acceptor effect of the groups) SH H3C SH H3C NO2 |
|
NOMENCLATURE AND STRUCTURE OF ORGANIC COMPOUNDS
methane; C2H6 or H3C-CH3 ethane; C3H8 or H3C-CH2-CH3 propane; C4H10 or H3C-CH2 Organic compounds are classified by the functional groups they contain |
|
3/08 1 Unit 4 Chemistry of Carbon Organic Chemistry studies the
Carboxyl group with H replaced by C Methylmethanoate H3C no number since the functional group is always on the end or first carbon Structure |
|
Nitrogen-Containing Functional Groups
Nitrogen-Containing Functional Groups Amines RNH2 primary (1°) R2NH secondary (2°) R3N tertiary (3°) CH3CH2NH2 ethylamine PhCH2NH2 benzylamine |
|
Hydrocarbons and Functional groups ANSWER KEY
Problem Set 2: Hydrocarbons and Functional groups ANSWER KEY Chemistry 260 Organic Chemistry H3C C CH3 Cl CH3 1-chlorobutane 2-chlorobutane |
What is H3C in chemistry?
H3C. isobutylene. (2-methylpropene)What is the structure of H3C?
Straight chain hydrocarbons are named according to the number of carbon atoms: CH4, methane; C2H6 or H3C-CH3, ethane; C3H8 or H3C-CH2-CH3, propane; C4H10 or H3C-CH2- CH2-CH3, butane; C5H12 or CH3CH2CH2CH2CH3, pentane; C6H14 or CH3(CH2)4CH3, hexane; C7H16, heptane; C8H18, octane; C9H20, nonane; C10H22, CH3(CH2)8CH3,What is H3C CH3 in chemistry?
Answer and Explanation: The functional group ( CH3?O? ) ( C H 3 ? O ? ) is termed as methoxy group or methoxide ion.
|
Functional Groups
A functional group is a defined grouping of atoms in an organic molecule Functional Group – General Structure Example C H O Aldehyde H3C C H O |
|
H3C CH3 CCCCH CH3 H3C HCCCCHHCCCCCCCCCCCCHH
Functional Groups to Know and Love Family Name Functional group structure Simple example Alkane Contains only C-H and C-C single bonds H3C CH3 |
|
Classification of Organic Compounds: Functional Groups - CSUN
H3C CH2OH H3C OCH3 H3C CH2Br C alkane alkene alkyne alcohol ether alkyl halide Classification of Organic Compounds: Functional Groups H |
|
Generic Functional Group Pattern Specific Example Using Suffix
Other patterns with special names based on the number of attached hydrogen atoms R H3C R H2 C R X H2 C |
|
Handout: Families of Organic Compounds FAMILY FUNCT GROUP
FAMILY FUNCT GROUP EXAMPLE SUFFIX methyl acetate –ate Amide acetamide –amide C C C H H H H H3C CH3 C C H2C CH2 C C HC CH C |
|
An Introduction to Functional Groups in Organic Chemistry What are
H3C C NH2 CH3 H N CH3 CH3 alcohols thiols amines Ethers ▫ The ether functional group is described as an sp3 oxygen atom bonded to two carbon |
|
Functional Groups
Functional Groups functional group: an atom, or group of atoms (with specific connectivity), exhibiting identical chemical reactivity regardless of the molecule |
|
Unit One Part 2: naming and functional groups
Identify some common functional groups found in organic molecules 1 O CH3 OH hept amide Step 2: suffix Step 4: functional groups (prefix) H3C N H O |
|
Organic molecules with functional groups containing - NUI Galway
the same functional group (FG) C,H carbon (*) bonded to the functional group H3C H carboxylic acid aldehyde 1° alcohol red red ox ox 2° alcohol ketone |
|
10 Functional Groups of Organic Compounds - UZH Chemistry
Important information about organic compounds and their functional groups can pKa = 4 8 phenols ArOH 10 ± 3 (approx ) pKa = 10 0 H3C S OH O O H3C |