halogenoalkane reactions
|
Classifying Halogenoalkanes Reactions of Halogenoalkanes
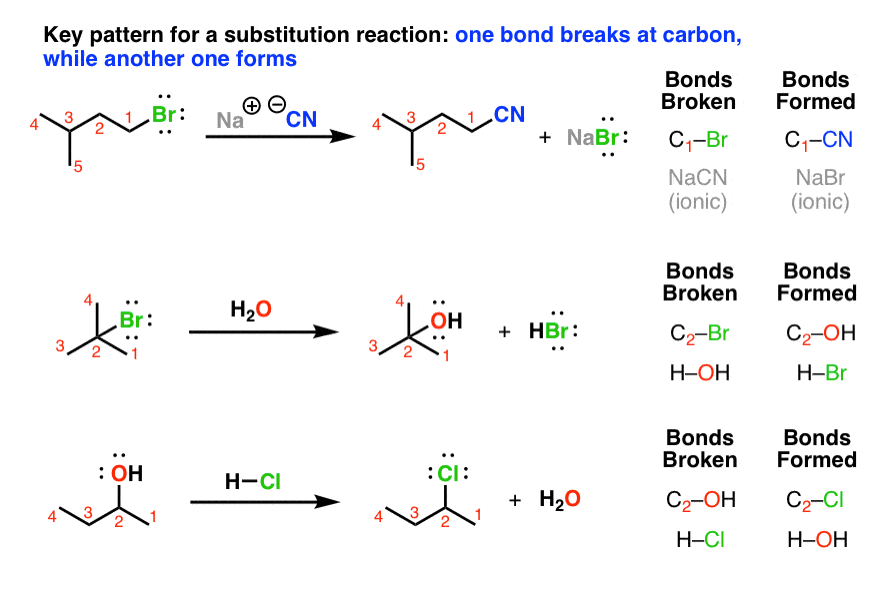
10 avr. 2018 The rate of these substitution reactions depends on the strength of the C-X bond ... case a halogenoalkane) by a reaction with water. |
|
Classifying Halogenoalkanes Reactions of Halogenoalkanes
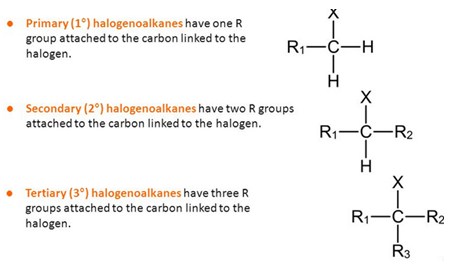
Tertiary halogenoalkane. Three carbons attached to the carbon atom adjoining the halogen. Reactions of Halogenoalkanes. Nucleophilic substitution reactions. |
|
Chemrevise
3 nov. 2018 The rate of these substitution reactions depends on the strength of the C-X bond ... case a halogenoalkane) by a reaction with water. |
|
Subject: Chemistry Topic: Halogenoalkanes 3.3.3 Year Group: 12
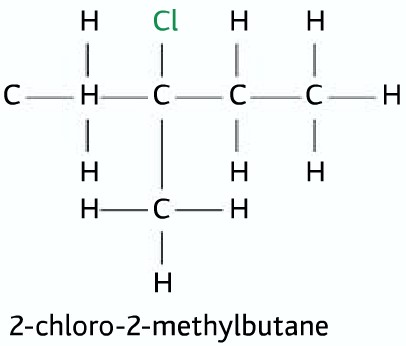
A small amount of ethanol is added to help dissolve the halogenoalkane as they are mostly insoluble in water. Key Vocabulary. 1 Substitution A reaction whereby |
|
Chemrevise
Reactions of Halogenoalkanes. 1. Nucleophilic substitution reactions. Nucleophile: electron pair donator e.g. :OH- :NH3 |
|
Mechanisms 1) Free radical substitution – Alkane
3) Nucleophilic substitution of halogenoalkanes a) With aqueous hydroxide OH- Hydrolysis – forming alcohols. • This reaction converts a halogenoalkane to |
|
Core practical 4: Investigate the hydrolysis of halogenoalkanes
Write an equation for the reaction of 1-bromobutane with water. 2. In these reactions a precipitate forms. Identify the precipitate formed when the. |
|
Chemrevise
Reactions of Halogenoalkanes. 1. Nucleophilic substitution reactions. Nucleophile: electron pair donator e.g. :OH- :NH3 |
|
C?+– F?- > C?+ – Cl?- > C?+– Br?-
6 nov. 2018 halogenoalkanes in a substitution reaction. Hydrolysis is defined as the ... Aqueous silver nitrate is added to a halogenoalkane and the. |
|
27 Halogenoalkanes
Synthesis of halogenoalkanes. In looking at the chemistry of alkanes and alkenes we described the forma- tion of halogenoalkanes in a number of reactions |
|
Classifying Halogenoalkanes Reactions of - chemrevise
10 avr 2018 · Tertiary halogenoalkane Three carbons attached to the carbon atom adjoining the halogen Reactions of Halogenoalkanes |
|
HALOALKANES (HALOGENOALKANES) - Knockhardy
NUCLEOPHILIC SUBSTITUTION REACTIONS Theory • halogens have a greater electronegativity than carbon • a dipole is induced in the C-X bond and it becomes |
|
Detailed Notes - Topic 33 Halogenoalkanes - AQA Chemistry A-level
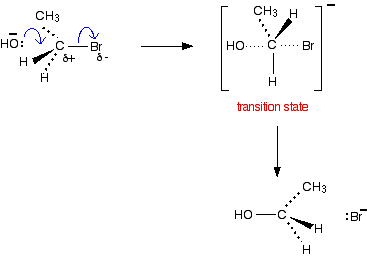
This is the reaction mechanism that shows how nucleophiles attack halogenoalkanes It can be used to produce alcohols or aminesfrom halogenoalkanes Mechanism |
|
33 The Halogenoalkanes
Reactions of the halogenoalkanes: Reactivity The polar bond makes the halogenoalkanes more reactive The C is deficient of electrons ?+ Nucleophiles |
|
Haloalkanes
A haloalkane (An alkyl halide) In this chapter we study two characteristic reactions of haloalkanes: nucleophilic substi- tution and b elimination |
|
HALOGENO COMPOUNDS - Savita Pall and Chemistry
Reactions of halogenoalkanes are nucleophilic substitution reactions Examples of nucleophiles: OH-1 NH3 CN-1 OR-1 (all Lewis ) |
|
7: Reactions of Haloalkanes Alcohols and Amines Nucleophilic
The haloalkane ionizes (reaction (7)) to form the t-butyl carbocation and a bromide ion as we showed earlier in the general SN1 mechanism (reactions (3) and (4)) |
|
Chapter 7 Haloalkanes - SDSU Chemistry
Table of Contents 1 Alkyl Halides (Haloalkane) 2 Nucleophilic Substitution Reactions (S N X X=1 or 2) 3 Nucleophiles (Acid-Base Chemistry pka) |
|
LES REACTIONS EN CHIMIE ORGANIQUE
V 2 LES REACTIONS DE SUBSTITUTION Dans ce type de réaction un agent nucléophile réagit avec un halogénoalcane afin de remplacer l'atome d'halogène |
|
Alkanes and Halogenoalkanes Science Skool!
Organic reaction mechanisms help chemists to understand how the reactions of organic compounds occur The following conversions illustrate a number of different |
|
LES REACTIONS EN CHIMIE ORGANIQUE
Dans ce type de réaction un agent nucléophile réagit avec un halogénoalcane afin de remplacer l'atome d'halogène La liaison C – X du substrat subit une |
|
Alkanes and Halogenoalkanes Science Skool!
7 (b) An alternative reaction can occur between this haloalkane and potassium hydroxide as shown by the following equation Name and outline a mechanism for |
|
33 Halogenoalkanes - chemrevise
Nucleophile: electron pair donator e g :OH-, :NH3, CN- Halogenoalkanes undergo either substitution or elimination reactions Substitution: swapping a halogen |
|
Classifying Halogenoalkanes Reactions of - chemrevise
10 avr 2018 · The rate of these substitution reactions depends on the strength of the C-X case a halogenoalkane) by a reaction with water CH3CH2X + |
|
27 Halogenoalkanes
Secondary halogenoalkanes can undergo SN2 reactions, but the reaction is generally slow because of steric hindrance Tertiary halogenoalkanes do not undergo |
|
Module 2 Alcohols, halogenoalkanes and analysis - Pearson
When halogenoalkanes react with an aqueous solution of hot hydroxide ions, a nucleophilic substitution reaction occurs The product of this reaction is an alcohol |
|
Questions on the reactions of halogenoalkanes with - Chemguide
HALOGENOALKANES: REACTIONS WITH HYDROXIDE IONS 50/50 mixture of ethanol and water, and a substitution reaction occurs (i) What do you |
|
Answers HALOGENOALKANES: REACTIONS WITH - Chemguide
Chemguide – answers HALOGENOALKANES: REACTIONS WITH SILVER NITRATE SOLUTION 1 a) You get a substitution reaction in which the halogen |
|
33 The Halogenoalkanes
This makes the halogenoalkanes more reactive Reactions of the halogenoalkanes: Reactivity This reaction converts a halogenoalkane to an alkyl nitrile |
|
Mechanisms 1) Free radical substitution – Alkane
3) Nucleophilic substitution of halogenoalkanes a) With aqueous hydroxide, OH- Hydrolysis – forming alcohols • This reaction converts a halogenoalkane to an |
|
Halogenoalkane Questions 1 Some reactions of halogenoalkanes
Some reactions of halogenoalkanes are show in the diagram below: a) Name The reaction to form compound A is called nucleophilic substitution Explain |
|
Halogenoalkanes and Alcohols - Physics & Maths Tutor
A nucleophile is a species (molecule or negative ion) which can donate an electron pair in a chemical reaction Halogenoalkanes undergo substitution reactions |