how do colligative properties differ in electrolytes and in non electrolytes
|
(Nonelectrolytes)
water is homogeneous since it does not differ significantly from a solution of sucrose. The colligative properties of solutions of nonelectrolytes. |
|
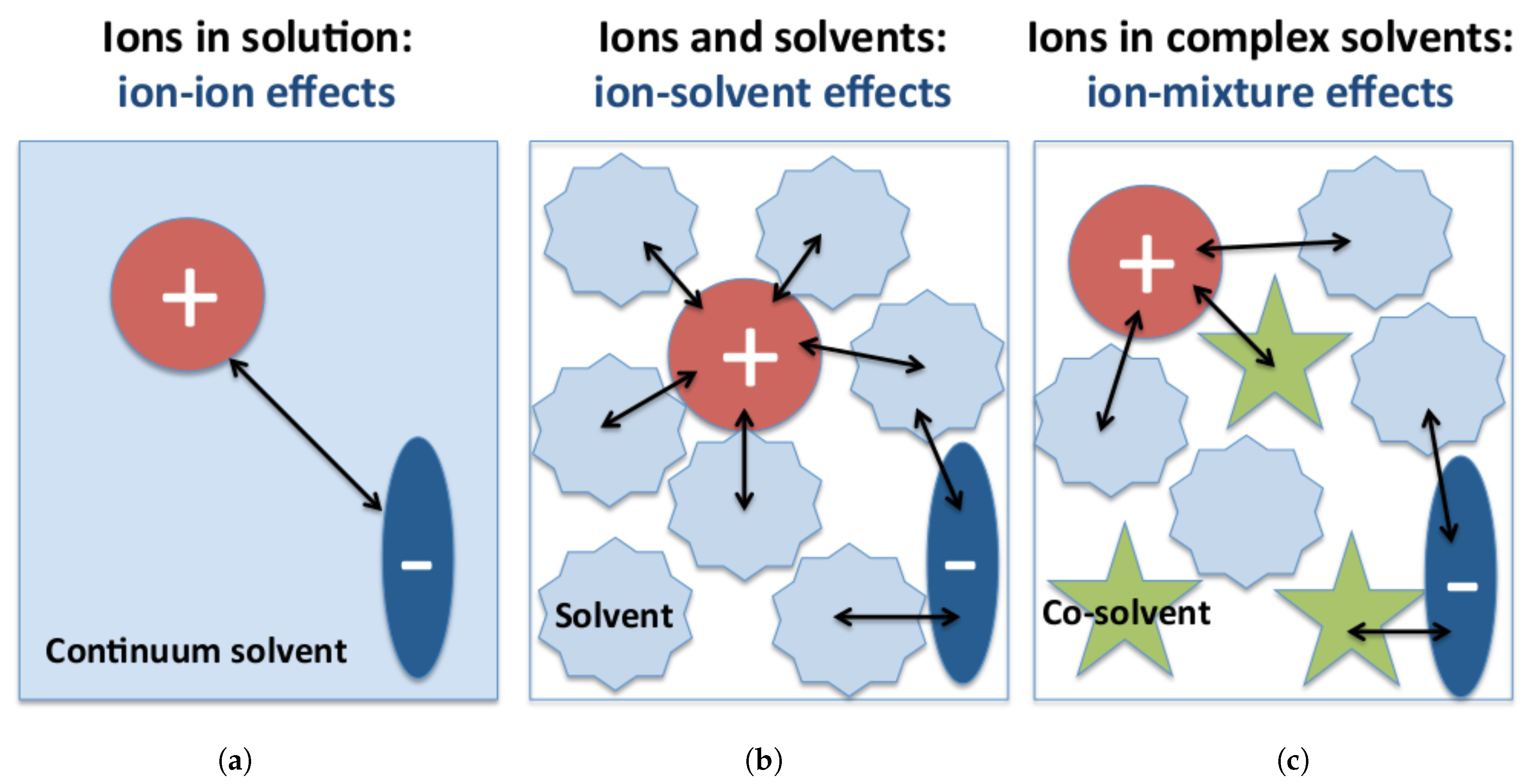
Ionic Strength In dilute solutions of nonelectrolytes activities and
weak electrolytes that are present alone in solution the differences between the ionic concentration terms and activities are usually disregarded in |
|
Physical Chemistry Laboratory
colligative properties of electrolyte solutions supported the Arrhenius theory for the freezing point depression of a solution of a non-electrolyte as a. |
|
THE INTERPRETATION OF THE COLLIGATIVE PROPERTIES OF
Acetates tartrates and ammonium salts do not interfere but decrease tance to the study of weak electrolytes and of non-electrolytes. Moreover |
|
13.5 Colligative Properties of Solutions
Weak electrolytes – dissociate partially (weak acids and bases). • Nonelectrolytes – do not dissociate (many organic compounds). |
|
Physical Chemistry Laboratory
Colligative properties can be described reasonably well by a simple equation for solutions of non-electrolytes. The “abnormal” colligative properties of |
|
CHAPTER 13
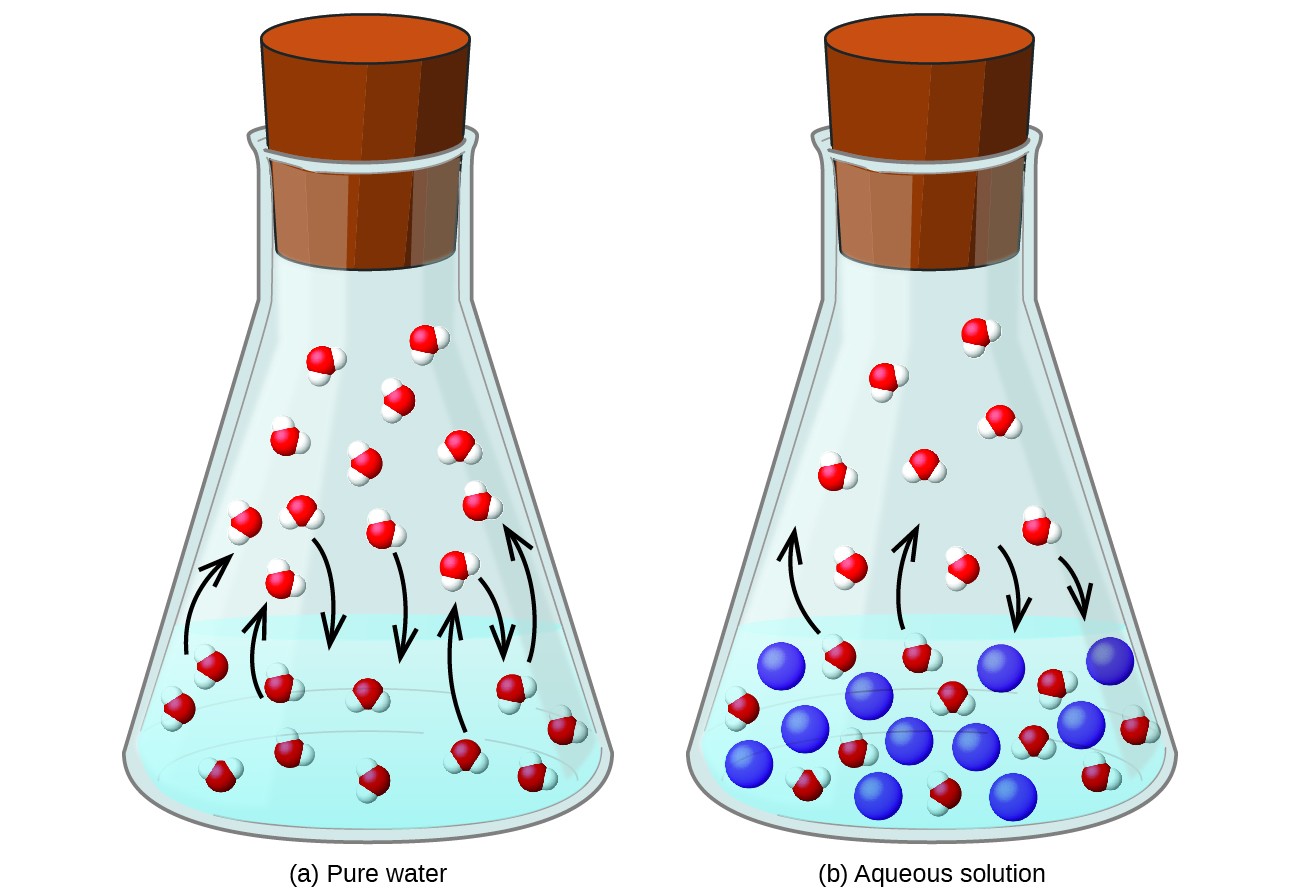
Ions in Aqueous Solutions and Colligative Properties They do not include any ions or substances that do ... Strong and weak electrolytes differ. |
|
Developing multiple representations teaching materials assisted by
chemical learning that have relationship between one concept and another are the electrolyte-non-electrolyte solution and the colligative properties of the |
|
Nonelectrolytes
16 oct. 2018 Nonelectrolytes are substances that do not yield ions ... Colligative properties will be discussed soon later. |
|
Solution of Electrolyte
5 nov. 2018 Colligative Properties of Electrolytic Solutions and Concentrated ... nonelectrolytes could be expressed satisfactorily by the equation ? =. |
|
(Nonelectrolytes)
The colligative properties of solutions are osmotic pressure vapor pressure lowering freezing point depression and boiling point elevation The values of the |
|
Colligative Properties of Non Electrolytes PDF Solution - Scribd
Avis 10 |
|
Colligative properties of solutions
1 fév 2021 · The colligative properties of electrolytes is characterized by slightly different approach than the one used for the colligative properties of |
|
1615: Electrolytes and Colligative Properties - Chemistry LibreTexts
8 août 2022 · Electrolytes and Colligative Properties Ionic compounds are electrolytes and dissociate into two or more ions as they dissolve |
|
114: Colligative Properties - Chemistry LibreTexts
18 jui 2020 · These observations suggest that the ions of sodium chloride (and other strong electrolytes) are not completely dissociated in solution To |
|
126 Colligative Properties of Electrolyte Solutions - UCF Pressbooks
These observations suggest that the ions of sodium chloride (and other strong electrolytes) are not completely dissociated in solution To account for this and |
|
Differentiate the colligative properties of electrolyte and non
Free Properties Of Solutions Lab Report Answers Pdf Pdf Webweb lab summary On the contrary non-electrolytes are those chemical compounds whose aqueous |
|
Properties Of Solutions Electrolytes And Nonelectrolytes Lab Report
Non- ElectrolytesElectrolytes and Colligative Properties Ionic compounds are electrolytes and dissociate into two or more ions as they dissolve |
|
How does the colligative properties of electrolyte solutions differ
How does the colligative properties of electrolyte solutions differ from those for non Postée : 26 avr 2023 |
How are colligative properties different from non Colligative?
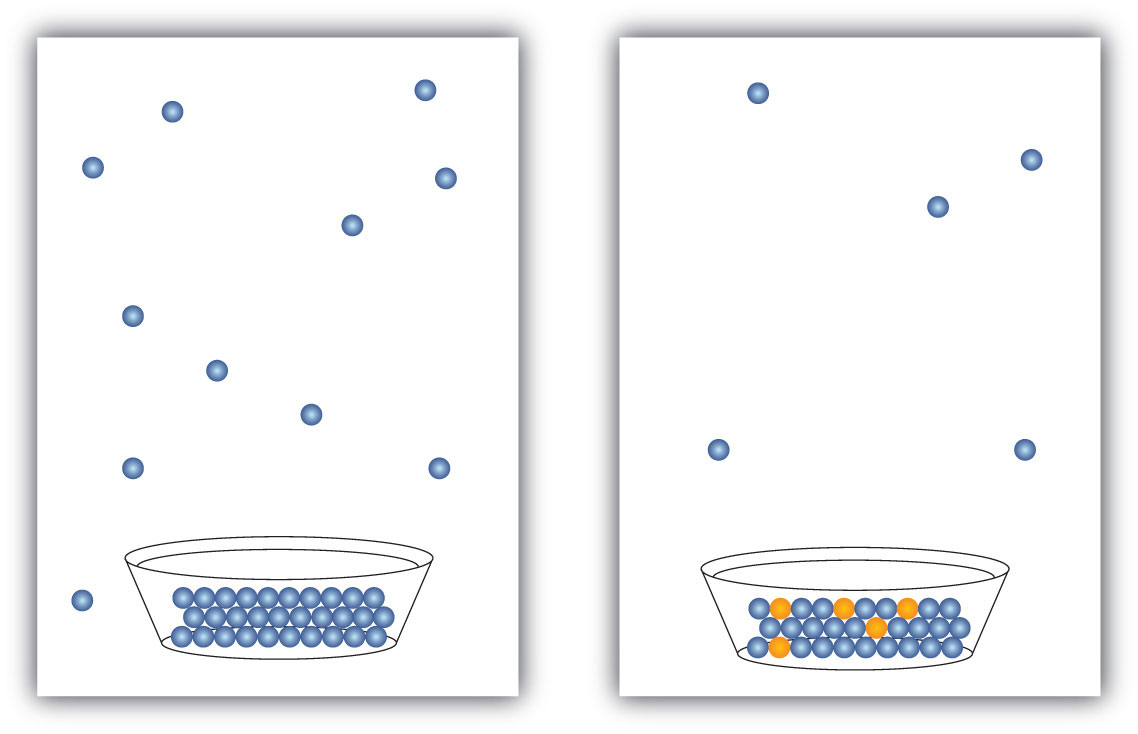
Colligative properties depend only on the number of dissolved particles of solute in solution and not on their identity. Non-colligative properties depend only on the identities of the dissolved species and the solvent.Why are colligative properties of electrolytes greater than non electrolyte solutions?
Explanation: This is because electrolyte dissociates in aqueous solution to give ions. So the number of particles are more and hence it has high colligative effect than non-electrolyte (which does not dissociate).What is the difference between an electrolyte and a non electrolyte?
Electrolytes conduct electric current when in solution or melted. Nonelectrolytes do not conduct electric current when in solution or melted. Some electrolytes play important roles in the body.- The colligative properties of solutions are osmotic pressure, vapor pressure lowering, freezing point depression, and boiling point elevation.
|
CHAPTER 13
Ions in Aqueous Solutions and Colligative Properties 419 SECTION 1 compounds are strong electrolytes Electrolytes Strong and weak electrolytes differ |
|
Colligative - Chandidas Mahavidyalaya
Electrolytes 9 Osmotic Pressure in the solution –Colligative properties do not depend on The only differences are the size of the effect which is reflected in |
|
Electrolyte Solutions: Thermodynamics, Crystallization - CORE
18 déc 2017 · Another basic difference between electrolyte and non-electrolyte solutions is the As it will be shown here, these colligative properties are not |
|
Colligative Properties - Crestwood Local Schools
Ionic compounds are usually strong electrolytes • Weak electrolytes provide few The sugar sucrose does not ionize at all in solution contributes to the colligative properties of the solvent Their physical properties are quite different from |
|
Properties Of Solutions Electrolytes And Non Electrolytes - Unhaggle
And Nonelectrolytes Colligative Properties of Electrolyte Solutions 13 9: Solutions of Electrolytes - Chemistry Solutions Electrolytes AndElectrolytic Properties Introduction to Difference Between Colligative Properties of Electrolytes per 1 L |
|
Chempor2001pdf - Biblioteca Digital do IPB
A Simple Apparatus for the Measurement of Colligative Properties in Aqueous enough insight for the big differences when compared to the non-electrolyte |
|
Chapter 13 Properties of Solutions Classification of Matter
Use colligative properties of solutions to calculate molar masses of solutes ➢ Explain the difference between a solution and a colloid ➢ Explain how solutions of electrolytes show greater changes than those of nonelectrolytes ∆ Tb = iK b |