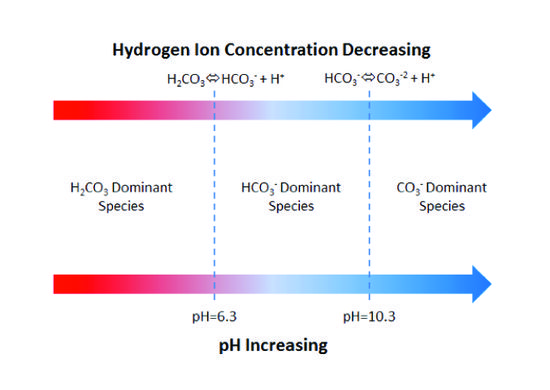
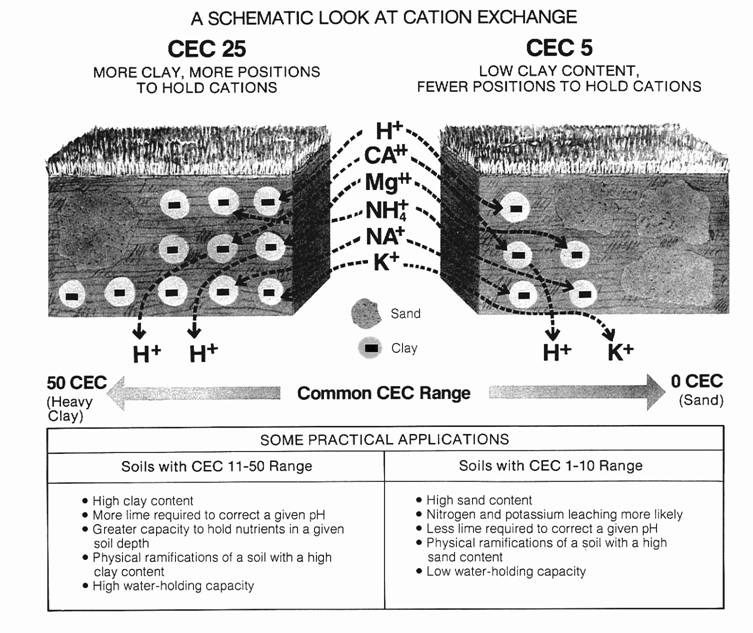
how to calculate buffer capacity
|
Buffer Calculations
buffer's capacity to neutralize a strong acid or a strong base. The Standard Equilibrium Approach to Calculating a Buffer's pH. |
|
Lime Requirement by Measurement of the Lime Buffer Capacity
This pH buffering capacity is calculated from the rise in pH measured. 30-45 minutes after adding an aliquot of Ca(OH)2. What is the Lime Buffer Capacity? |
|
TYTU? PREZENTACJI
Buffering Capacity. Page 9. How to calculate buffer capacity? Example 3. For 100 mL of buffer was added 1 mL of 0.9 M HCl calculate the buffer capacity:. |
|
Buffer Capacity
Buffer capacity is measured as the amount of acid/base it takes the change In this equation H = 10-pH |
|
Calculation of the Buffering Capacity of Bicarbonate in the Rumen
The pH under different pressures at equilibrium and the buffering capacity are easily calculated for in vitro and in vivo systems. Key Words: Rumen pH |
|
Seahorse XF Buffer Factor Protocol Quick Reference Guide
dependent on the buffer capacity of the medium and the properties of the measurement chamber. Determining and reporting the actual amount of protons. |
|
The ion?association model and the buffer capacity of the carbon
Aug 1 1973 model of Berner has been extended to calculate the contributions of the various ion-pair formation reactions to the buffer capacity of the ... |
|
Chapter 1.2: Visualization of Buffer Capacity with 3-D Topos: Buffer
Dec 14 2020 for teaching with BufCap TOPOS |
|
A buffer is a solution that resists changes in pH upon the addition of
Buffering capacity refers to the amount of added acid or added base that can be neutralized by a buffer. Calculate the pH of the following 3 solutions. |
|
Buffering In Muscles Activity
Although we are interested in muscles' ability to buffer acid this experiment investigates their Calculate the buffering capacity of each of the three. |
|
PH buffers Buffer capacity and buffering range
The term "buffer capacity" (?) quantifies the change in pH of the solution caused by the addition of a strong acid or base It is calculated in relation to |
|
Buffer Capacity
Buffer capacity is measured as the amount of acid/base it takes the change In this equation H = 10-pH Ka and Kw are the acid equilibrium constant and |
|
Buffer Capacity
To determine the maximum buffer capacity of a number of buffer solutions ? To establish the relationship between buffer capacity and buffer concentration |
|
Buffer Index and Buffer Capacity for a Simple Buffer Solution
According to IUPAC (6) the definition of buffer index or buffer capacity is as follows: “The capacity of a solution to resist changes in pH on the addition of |
|
Buffer Calculations - DePauw University
buffer's capacity to neutralize a strong acid or a strong base The Standard Equilibrium Approach to Calculating a Buffer's pH |
|
Section 191 Acid-Base Buffer Solutions
Buffer Calculations: Two types we must be able to handle: (A) Buffer Capacity is how much added acid or base a buffer solution can “mop up” |
|
Buffer capacity
Consequently at pH ? 8 the buffer capacity of the carbonate system in a more complex aqueous system such as seawater the charge balance equation is |
|
Calculating the pH of a buffer Buffer capacity pH of blood
Buffering Capacity Page 9 How to calculate buffer capacity? Example 3 For 100 mL of buffer was added 1 mL of 0 9 M HCl calculate the buffer capacity: |
|
Calculation of the Buffering Capacity of Bicarbonate in the Rumen
The addition of NaHCO3 to buffer solutions and ruminal fluid under high pressure of CO2 increased pH as predicted The buffering capacity of ruminal fluid |
|
(PDF) Understanding Deriving and Computing Buffer Capacity
Unfortunately however derivation and systematic calculation of buffer capacity is a topic that seems to be neglected in the undergraduate analytical |
|
Buffer Calculations - DePauw University
which simplifies to pH = pKa + log (moles CH3COO–)o (moles CH3COOH)o and when we add a strong base the buffer capacity is defined by the equation (moles weak base)o + moles OH– (moles weak acid)o − moles OH– = 10 Example 3 = 7 20 + log 0 1435 0 1837 = 7 09 |
|
Buffer Preparation and Capacity
The buffering capacity of a solution is determined using the Henderson-Hasselbalch Equation, and the closer the ratio of acid to conjugate base is to one the higher buffering capacity A buffer also can consist of a weak base and its conjugate acid as well |
|
PH buffers Buffer capacity and buffering range
Based on the Henderson-Hasselbalch equation, it can be seen that the pH of the buffer solution is equal to the pKa value of the acid, when the ratio of |
|
LEC 0616 Titration curves and buffering capacity - PHYWE System
2 Determine the titration curve of an ampholyte (glycine) 3 Determine the buffering capacity of various aqueous acetic acid/sodium acetate mixtures at different |
|
DETERMINATION OF THE BUFFERING CAPACITY OF - CORE
possible to estimate the buffering capacity of meat from data based on titrations made with different dilutions A mean value for buffering capacity valid in pH |
|
BUFFERS AND BUFFER CAPACITY
Graphically the Henderson-Hasselbalch equation plotted as the acid : conjugated base ratio vs pH of buffer actually constitutes the titration curve of the weak acid |
|
Buffers and Titration
perform titrations of that buffer with acid or base to find the 3) Hydrolyze BSA with trypsin and calculate the Highest buffering capacity obtained when [A-] = |
|
Calculation of the Buffering Capacity of Bicarbonate - Natural Soda
We describe a model to calculate the buffering capacity of bicarbonate in the rumen The addition of NaHCO3 results in the release of CO2 from solution and |