how to calculate dilution factor from concentration
|
Dilutions Help
CALCULATING THE CONCENTRATION. To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor . CALCULATING THE |
|
Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor this by |
|
Appendix 8: Dilution Factor and Effluent Limitation Calculations for
A DF for sites that discharge to freshwater receiving waters in Massachusetts is calculated using the equation below. Alternate calculation methods for DFs may |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working. (source solution |
|
Appendix V: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: Dilution Factor projected downstream concentration of a parameter calculated in accordance with C. |
|
Preparation and Sequencing of RNA Libraries on the Ion PGM
17-May-2012 After determining the molar concentration of the library calculate the Template Dilution Factor to dilute the library for preparation of ... |
|
ICH guideline M10 on bioanalytical method validation and study
25-Jul-2022 At least 5 replicates per dilution factor should be tested in one run to determine if concentrations are accurately and precisely measured ... |
|
Calculating Nucleic Acid or Protein Concentration Using the GloMax
07-Aug-2009 Multiply this number by the DNA or RNA constant from Table 1 c. Multiply by the sample dilution factor. 3. Estimate nucleic acid purity a. |
|
Appendix V: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: QS + above and the discharge concentration are greater than the WQC calculated for that parameter in. |
|
Thermo Fisher Scientific
measurements and then expressing these concentrations as a ratio. The ratio Record the dilution factor which will be required in the calculations. 3 ... |
|
Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor . CALCULATING THE UNDILUTED CONCENTRATION. Often we wish |
|
Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor |
|
Laboratory Math II: Solutions and Dilutions
The concentration of a solution is how much of the solute You will also want to determine your dilution factor. The dilution factor is the factor by. |
|
1. dilutions and concentrations. liquid and solid samples
of this 1:10 dilution 1 ml corresponds to the sample. properly apply the dilution or concentration factors to determine the microbial density of a ... |
|
Appendix VI: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: downstream concentration of a parameter calculated in accordance with C.1 below |
|
Groundwater risk assessments for infiltration systems: calculations
Calculate the dilution factor (DF) for groundwater below the drainage field: 1. Calculate DF taking background concentration into account. |
|
Determination of Dilution Factors for Discharge of Aluminum
flow-ratio DF for streams defined at low flow (7Q10) |
|
JT - Dilution Factor
Dilution Factor: reading gave a concentration of 24.0 ppm for this diluted sample what is the concentration of lead in the original sample? Solution:. |
|
Appendix V: Dilution Factor and Effluent Limitation Calculations for
Calculate WQBEL: The freshwater WQBEL is calculated by rearranging the above mass balance equation to solve for the effluent concentration (Cd) by setting the |
|
Preparation of Solutions Preparation of Solutions
Oct 17 2016 To make a dilute solution without calculating concentrations use a dilution factor. • Divide the final volume by the initial volume. |
|
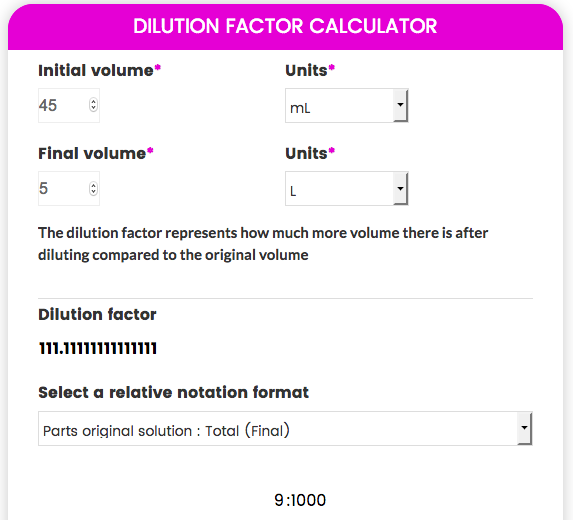
Dilution Factor:
Dilution factor is defined as: total volume of solution per aliquot volume Where total volume of solution is: 10 0 + 240 0 = 250 0 mL (volumetric flask ) 25 |
|
Formula 3 Serial Dilutions
A)Calculate the concentration of the three diluted solutions? ? First: find the D F: Dilution factor (D F) = final volume / aliquot volume |
|
1 dilutions and concentrations liquid and solid samples
We must know not only how to dilute or concentrate a sample; we must also understand how to properly apply the dilution or concentration factors to determine |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working (source solution |
|
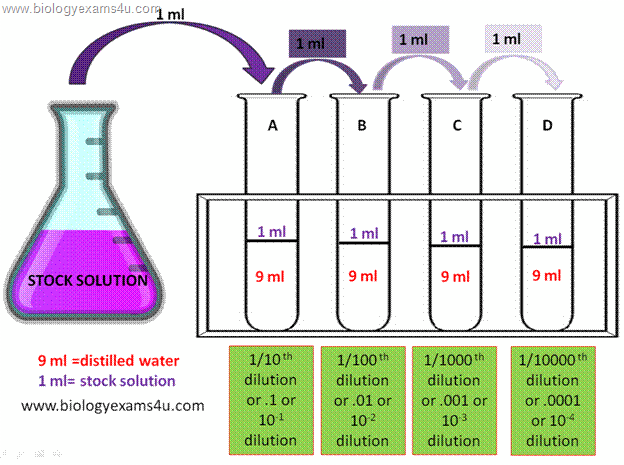
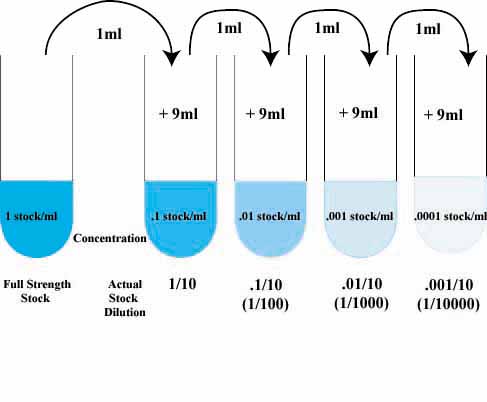
Dilution-Notespdf - SJU WordPress Sites
Serial dilutions are successive dilutions of a sample The final dilution factor is the multiplicative product of all dilutions 1 mL 1 mL |
|
Laboratory Math II: Solutions and Dilutions
The concentration of a solution is how much of the solute You will also want to determine your dilution factor The dilution factor is the factor by |
|
Dilutions - Weber State University
Since dilutions are simply changes in the ratio of solute to solvent a simple ratio of concentration to volume can be used for almost all calculations |
|
Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor |
|
Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor CALCULATING THE UNDILUTED CONCENTRATION Often we wish |
|
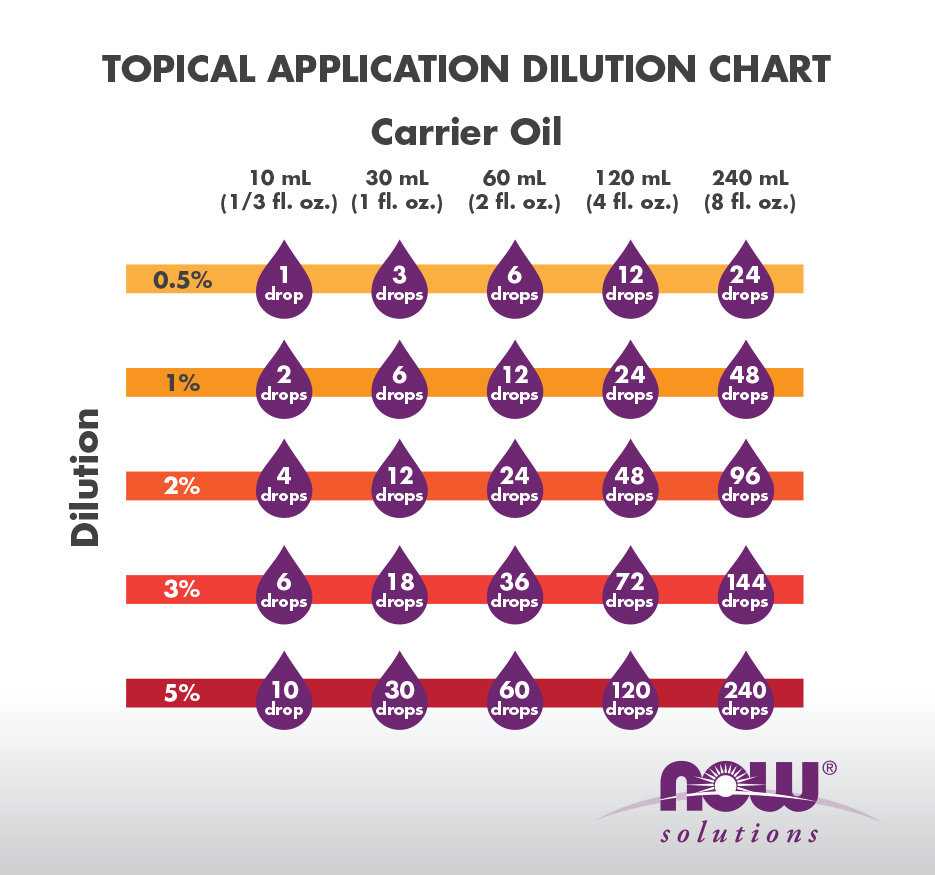
How to Make Simple Solutions and Dilutions
l Simplc Dilution (Dilution Factor Nlethod based on ratios) per volume and can lrc simply calculated as the concentration t tolune needed = nass of |
How do you find the dilution factor based on concentration?
The dilution factor is the inverse of the concentration factor. For example, if you take 1 part of a sample and add 9 parts of water (solvent), then you have made a 1:10 dilution; this has a concentration of 1/10th (0.1) of the original and a dilution factor of 10.How do you calculate the dilution factor?
The formula for dilution factor (or DF for short) is as follows: DF = (final volume of cells + stain)/(initial volume of cells). For example, If you mix your sample 1:1 with AO/PI, you'll need to add 20 uL AO/PI to 20 uL cells, for a total of 40 uL. So, DF = ( 40 uL)/(20uL cells) = 2.What is the dilution factor if you add 0.1 ml aliquot of specimen to 9.9 ml of diluent?
What is the dilution factor if you add 0.1 ml aliquot of a specimen to 9.9 ml of diluent? The dilution factor is equal to the final volume divided by the aliquot volume: 10 mL/0.1 mL = 1:100 dilution.- What is the dilution factor when 0.2 ml is added to 3.8 ml diluent? 4.0/0.2 = 1:20 dilution.
|
Dilution Factor:
Dilution factor is defined as: total volume of solution per aliquot volume Where total volume of solution is: 10 0 + 240 0 = 250 0 mL (volumetric flask ) Note: For multiple dilutions the dilution factor is the product of the dilution factors for each individual dilution |
|
Dilutions Occasionally a solution is too concentrated to be used as it
use in calculating the concentration of solutions in a series is to multiply the original dilution factor, and so on until the final concentration is known Example: A |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
Slide 17 Serial Dilutions: Things to Consider ▫ Start with your final volume and concentration and your initial concentration ▫ Determine your dilution factor |
|
Lab Math Solutions, Dilutions, Concentrations and Molarity - APHL
25 avr 2016 · ✓The dilution factor is the total number of unit volumes in which your Example: Experiment requires a final concentration of 0 1 M Tris, in a |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
Example 2: A 1X solution of SSC can be prepared from a 20X SSC stock solution a serial dilution the total dilution factor at any point is the product of the individual dilution V2 = total volume needed at the new concentration = 200 l = 0 2 ml |
|
Dilution (and Concentration) Theory
Dilution and concentration theory sounds ominous but you use dilutions and colony count x dilution factor (the inverse of the dilutions) = CFU/ml of original |
|
How to Make Simple Solutions and Dilutions
l Simplc Dilution (Dilution Factor Nlethod based on ratios) A sinple per volume and can lrc simply calculated as the concentration t tolune needed = nass of |
|
Calculating Concentrations and Mixing Reagents
A dilution ratio is used to describe a simple dilution, in which a unit volume of a solute is combined with a desired volume of solvent C Dilution Factor Although |


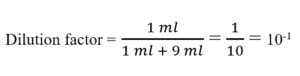






















![Pharmacy Calculations [1] - [PDF Document] Pharmacy Calculations [1] - [PDF Document]](https://study.com/cimages/videopreview/5.67_101788.jpg)