how to determine the rate law of a reaction
|
Differential and Integrated Rate Laws
Read along and you will find out. On more thing – I must insist: it is not possible to predict the rate law from the overall balanced chemical reaction; rate |
|
Experiment 5 Kinetics: The Oxidation of Iodide by Hydrogen
To determine the differential rate law for the reaction between iodide and hydrogen peroxide in an acidic environment. To determine the activation energy |
|
Sample Exercise 14.1 Calculating an Average Rate of Reaction
Sample Exercise 14.3 Relating Rates at Which Products Appear and. Reactants Disappear. Solution. Analyze We are given a balanced chemical equation and asked |
|
1 General Chemistry II Jasperse Kinetics. Extra Practice Problems
The orders cannot be determined without a chemical reaction. 18. For the rate law Rate = k[A][B]3/2 the order with respect to A is ______ |
| Rate of Reaction of Sodium Thiosulfate and Hydrochloric Acid |
|
Chemistry 11 IB Determining the Order of a Reaction 1 Planning B
28 avr. 2004 Although it wasn't possible to write out the actual rate law for this particular reaction (initial rates weren't found) we were able to ... |
|
Sample Exercise 14.1 Calculating an Average Rate of Reaction
Analyze: We are given a table of data that relates concentrations of reactants with initial rates of reaction and asked to determine (a) the rate law |
|
Concept Check 13
Consider the reaction Q + R ? S + T and the rate law for the reaction: Rate In this equation like chemical rate equations |
|
AP® CHEMISTRY 2010 SCORING GUIDELINES (Form B)
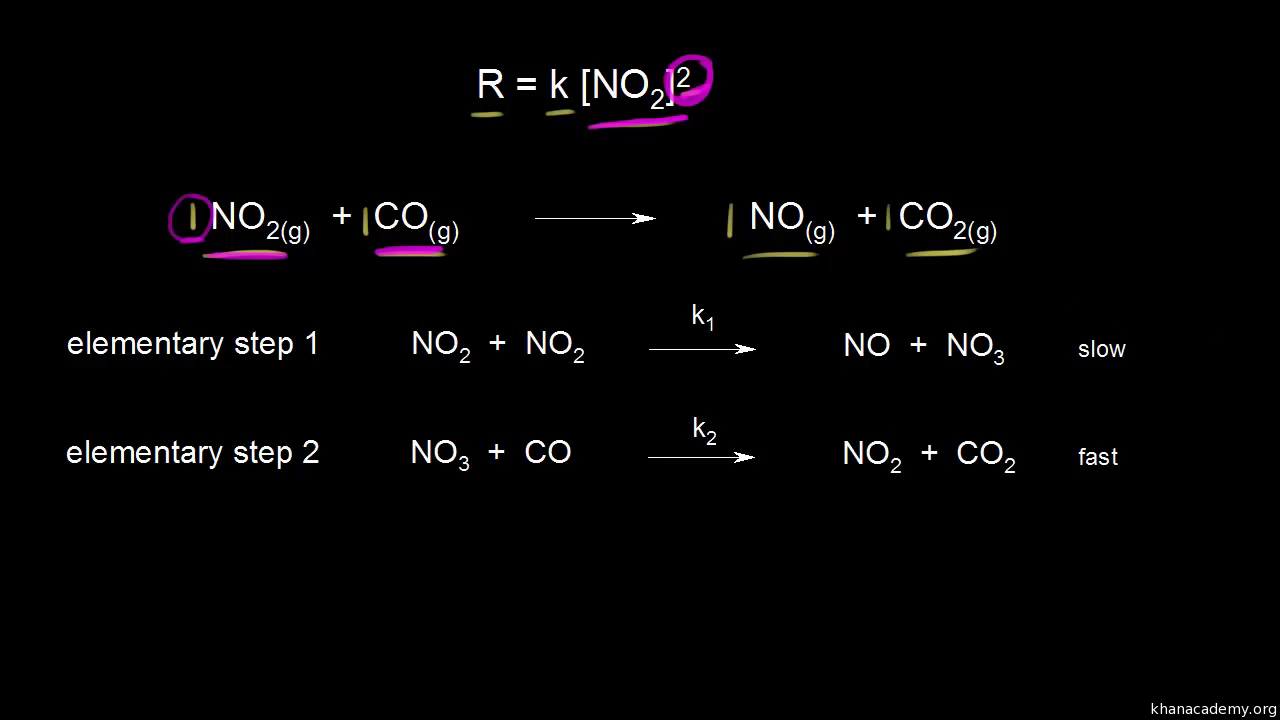
(h) Identify the slower step in the mechanism if the rate law for the reaction was determined to be rate = k [N2O]. Justify your answer. Step 1 is slower |
|
Exercise 12.5 - Reaction Mechanisms - Answers.pdf
balanced equation. They must also explain the experimentally determined rate law. The slowest step in the reaction mechanism will determine the overall rate |
|
Chapter 14 Chemical Kinetics - UNF
? Determine the rate law and rate constant for a reaction from a series of experiments given the measured rates for various concentrations of reactants ? Use |
|
Chemical Kinetics: Rates and Rate Laws
Rates of chemical reactions are usually defined by comparing the change in reactant or product concentration over time Consider the reaction N2 + 3 H2 2 NH3 |
|
Chemical Kinetics: Determining Rate Laws for Chemical Reactions
We can determine the rate of the crystal violet reaction with OH- by using a SPEC-20 to monitor the drop in absorbance of crystal violet in solution THE |
|
Differential and Integrated Rate Laws
Rate laws describe the progress of the reaction; they are mathematical These rate laws help us determine the overall mechanism of reaction (or process) |
|
RATE LAWS ORDER OF A REACTION
An expression which shows how the reaction rate is related to concentrations is called the rate law or rate equation The power (exponent) of concentration |
|
Chapter 14 Chemical Kinetics
Knowing the concentrations of reactants and the rate of a reaction with these concentrations we can determine the rate constant 5 4 x 10-7 M/s = k (0 0100M)( |
|
Chemical Kinetics
Catalysts speed up reactions by changing the mechanism of the reaction This equation is called the rate law and k is the rate constant |
|
Reaction Kinetics - Claire Vallance
Introduction 2 Rate of reaction 3 Rate laws 4 The units of the rate constant 5 Integrated rate laws 6 Half lives 7 Determining the rate law from |
|
Ch 3 Rate Laws and Stoichiometry Part 1
Rate Law – Find the rate as a function of concentration What is the reaction rate law for the reaction A + ½ B ? C if the reaction is elementary? |
|
Establishing the Rate Law
What's special about the initial rate? Once the reaction starts it is very difficult to know what the concentrations are at any given time unless you already |
- The rate law can be measured by the reaction rate as the initial concentration activity by keeping the concentration of one of the reactants constant and converting the other reactant.
|
Reaction Rates: Chemical Kinetics
For the general reaction: Rate Law Reaction Order aA + bB → cC + dD x and y are the reactant orders determined from experiment x and y are NOT the |
|
PDF chapter, Determining the rate law and obtaining - IOPscience
Kinetic data may be analysed to determine the reaction rate, orders with respect to each reactant, and the rate constant for the reaction As we shall see, these |
|
Sample Exercise 141 Calculating an Average Rate of Reaction
Sample Exercise 14 3 Relating Rates at Which Products Appear and Reactants Disappear Solution Analyze We are given a balanced chemical equation and |
|
Determination of Rate Laws
seuille's laws) are the nonlinear rate laws for chemical reactions The rate constant is The first is to establish, experimentally, the rate law which describes the |
|
Chapter 14 Chemical Kinetics
We can then use the rate constant to determine the reaction rate for any given set of concentrations of [NH4 +] and [NO2 -] A Reaction Order rate laws for most |
|
Ch 3 Rate Laws and Stoichiometry Part 1: Rate Laws
Rxn Order Rate Law: Algebraic equation that relates –rA to the concentrations of the reactants is called the “kinetic expression” or “rate law” |
|
13 • Chemical Kinetics
Determine the order of the reaction for each reactant b Write the rate equation for the reaction c Calculate the rate constant at 904 °C d Find |
|
Differential and Integrated Rate Laws
Read along and you will find out On more thing – I must insist: it is not possible to predict the rate law from the overall balanced chemical reaction; rate laws must |




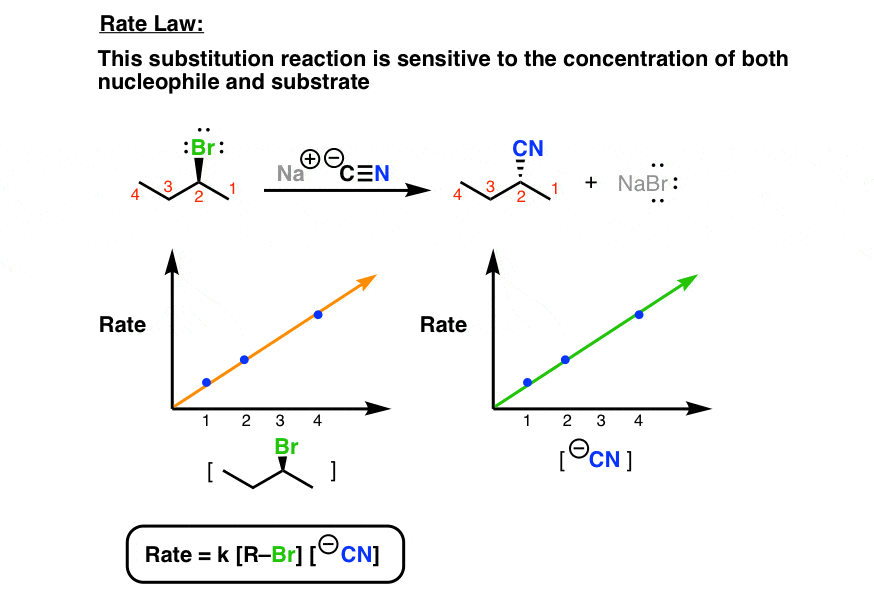
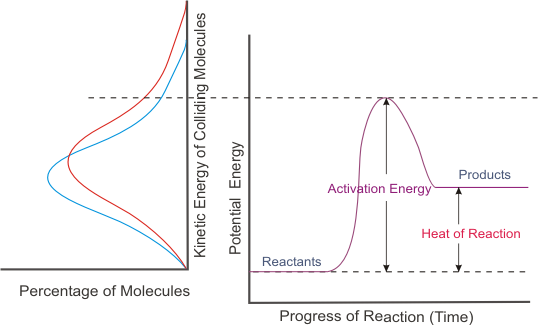





![a) For a reaction A + B→ P the rate is given by Rate \u003d k[A][B a) For a reaction A + B→ P the rate is given by Rate \u003d k[A][B](https://cdn.kastatic.org/googleusercontent/UmxM1vFrq4y98KboPSA_-Kq9A8RRxR4leB7yNMOYGUhFK4L2Ie6WM4ZCuohNKE2NC6m9jZZJ8KsZgHzK1jN4uuaL)