how to determine the rate of a reaction
|
Sample Exercise 14.1 Calculating an Average Rate of Reaction
Analyze We are asked to determine an instantaneous rate from a graph of reactant concentration versus time. Plan To obtain the instantaneous rate at t = 0s we |
|
Experiment 5 Kinetics: The Oxidation of Iodide by Hydrogen
To determine the effect of a catalyst on the rate of reaction. Discussion. When hydrogen peroxide is added to a solution of potassium iodide the iodide ions |
|
Sample Exercise 14.1 Calculating an Average Rate of Reaction
From the data given in the caption of Figure 14.3 calculate the average rate at which. A disappears over the time interval from 20 s to 40 s. For the reaction |
| Rate of Reaction of Sodium Thiosulfate and Hydrochloric Acid |
|
Chapter 14 Chemical Kinetics
The temperature at which a reaction occurs: The rates of chemical We can then use the rate constant to determine the reaction rate for any given. |
|
Enzyme Kinetics: Velocity
20 févr. 2013 The rate of the reaction catalyzed by enzyme E ... C. Measure the velocity by determining the rate of product formation. |
|
??????? 1
Sample Exercise 14.1 Calculating an Average Rate of Reaction. From the data given in the caption of Figure calculate the average rate at which A. |
|
Chemistry 1011
Reaction Rate. • Define the average rate and instantaneous rate of a chemical reaction in terms of reactant and/or product concentration. |
|
Differential and Integrated Rate Laws
On more thing – I must insist: it is not possible to predict the rate law from the overall balanced chemical reaction; rate laws must be determined |
|
Untitled
Measuring Reaction Rates. 1. A chemist wishes to determine the rate of reaction of zinc with hydrochloric acid. The equation for the reaction is:. |
|
Chapter 14 Chemical Kinetics
Knowing the concentrations of reactants and the rate of a reaction with these concentrations we can determine the rate constant 5 4 x 10-7 M/s = k (0 0100M)( |
|
CHAPTER 13 – Rates of Reactions
a) Calculate the average rate of this reaction in this time interval b) Predict the rate of change in concentration of H + during this time interval |
|
Sample Exercise 141 Calculating an Average Rate of Reaction
Sample Exercise 14 1 Calculating an Average Rate of Reaction Solution Analyze We are given the concentration of A at 20 s (0 54 M) and at 40 s (0 30 M) |
|
Chemical Kinetics
- A rate law shows the relationship between the reaction rate and the concentrations of reactants - The exponents tell the order of the reaction with respect |
|
CHEMICAL KINETICS/RATE OF CHEMICAL REACTION 1
? This is a reaction whose rate is independent of concentration (i e initial concentration) ? A product ? Rate equation in differential form is R |
|
Types of chemical reactions Rate of a reaction Factors affecting rate
Molecularity of a complete reaction has no significance and overall kinetics of the reaction depends upon the rate determining step Slowest step is the rate- |
|
Chemical Kinetics: Rates and Rate Laws
Rates of chemical reactions are usually defined by comparing the change in reactant or product concentration over time Consider the reaction N2 + 3 H2 2 NH3 |
|
KINETICS: RATES OF CHEMICAL REACTIONS
The rate of reaction is a positive quantity that describes how the concentration of a reactant or a product varies with time • Consider the reaction: N2 + 3H2 |
|
Chapter 14 Chemical Kinetics - UNF
? Determine the rate law and rate constant for a reaction from a series of experiments given the measured rates for various concentrations of reactants ? Use |
|
RATE OF REACTION
The rate of a chemical reaction may be described as the quantity of product produced per unit time or the quantity of reactant used up per unit time This |
|
Reaction Rates: Chemical Kinetics
For the general reaction: Rate Law Reaction Order aA + bB → cC + dD x and y are the reactant orders determined from experiment x and y are NOT the |
|
Sample Exercise 141 Calculating an Average Rate of Reaction
Plan The average rate is given by the change in concentration, ∆[A], divided by the change in time, ∆t Because A is a reactant, a minus sign is used in the calculation to make the rate a positive quantity |
|
Chapter 14 Chemical Kinetics
Some rates do not depend on reactant concentration These are zero order reactions The rate law is: Rate = k • These reactions are linear in concentration The half-life, t1/2 is the time needed for the concentration of a reactant to decrease to half of its initial value |
|
Chapter 14 Chemical Kinetics
Rate = A [C4H9Cl] A [C4H9OH] Initial [ NO2-] (M) Observed initial rxn Example 2: IF ONE REACTANT IS INVOLVED (usually a decomposition reaction) The initial rate of decomposition of acetaldehyde, CH3 CHO, Example 3: TWO REACTANTS 2H2(g) + 2NO (g) → N2(g) + 2H2O (g) III D V rate = k[A][B] |
|
PDF chapter, Determining the rate law and obtaining - IOPscience
Kinetic data may be analysed to determine the reaction rate, orders with respect to each reactant, and the rate constant for the reaction As we shall see, these |
|
The effect of concentration on rate - Nuffield Foundation
Rate cannot be directly measured, so instead chemists often measure the concentration of a reactant or volume of gaseous product as the reaction progresses |
|
RATES OF CHEMICAL REACTIONS OBJECTIVES 1 To
temperature and the presence of a catalyst affect the rates of reactions 2 The effect of temperature will be determined by observing the reduction of potassium |
|
Chemical Kinetics - NCERT
Kinetic studies not only help us to determine the speed or rate of a chemical reaction but also describe the conditions by which the reaction rates can be altered |




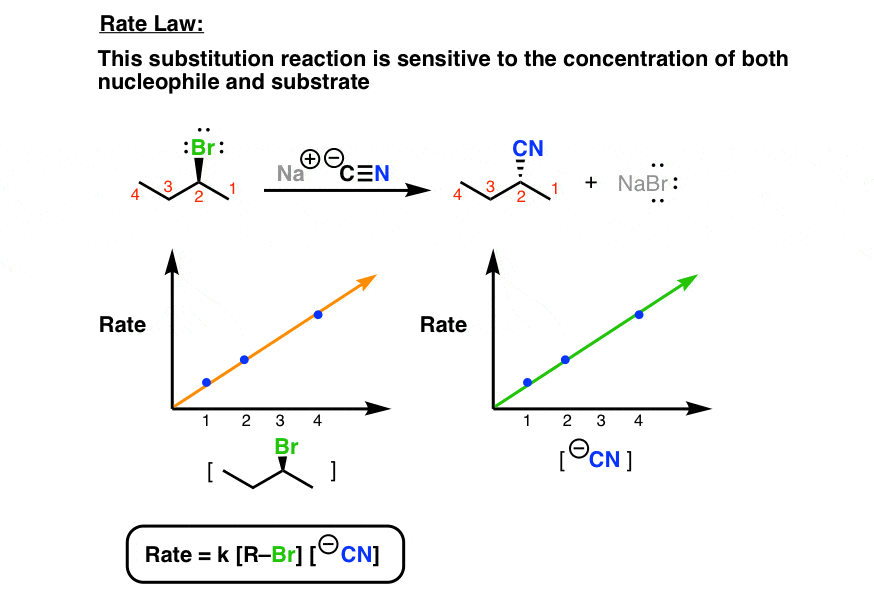





![a) For a reaction A + B→ P the rate is given by Rate \u003d k[A][B a) For a reaction A + B→ P the rate is given by Rate \u003d k[A][B](https://cdn.kastatic.org/googleusercontent/UmxM1vFrq4y98KboPSA_-Kq9A8RRxR4leB7yNMOYGUhFK4L2Ie6WM4ZCuohNKE2NC6m9jZZJ8KsZgHzK1jN4uuaL)
