how to evaluate a clinical trial
|
Quick guide – Part I - How to evaluate a Clinical Trial
How to evaluate a Clinical Trial. Application: Assessment and. Decision. CTIS Training Programme – Module 08. Version 1.3 – May 2022. Learning Objectives. |
|
CTTM08 - Quick Guide_Decision
How to evaluate a Clinical Trial. Application: Assessment and. Decision. CTIS Training Programme – Module 08. Version 1.5 – May 2022. Learning Objectives. |
|
CTTM08 - Quick Guide_Intro
How to evaluate a Clinical Trial. Application: Assessment and Decision. CTIS Training Programme – Module 08. Version 1.3 – May 2022. Learning Objectives. |
|
FAQs - How to evaluate a Clinical Trial Application: Assessment and
5 mai 2022 The European Medicines Agency developed this training material to enhance public access to information on the Clinical Trial Information ... |
|
Quick guide – Part II - How to evaluate a Clinical Trial Application
3 mai 2022 The European Medicines Agency developed this training material to enhance public access to information on the Clinical Trial Information ... |
|
Instructors Guide: - How to evaluate a Clinical Trial Application
How to evaluate a Clinical Trial Application: Assessment and Decision. CTIS Training Programme – Module 08. Version 1.2 – September 2021. What you will find. |
|
Reflection paper on laboratories that perform the analysis or
28 févr. 2012 or evaluation of clinical trial samples ... 28 February 2012. Keywords. Clinical laboratory Laboratory analysis |
|
Checklist for Evaluating Whether a Clinical Trial or Study is an
14 déc. 2016 Does the study evaluate at least one drug biological |
|
FAQs - How to evaluate a CTA CTIS Training Programme – Module 06
7 janv. 2022 The European Medicines Agency developed this training material to enhance public access to information on the Clinical Trial Information ... |
|
E 9 Statistical Principles for Clinical Trials Step 5
The role of statistics in clinical trial design and analysis is evaluation of clinical trials of an investigational product in the context of its ... |
|
Quick guide - Part I : How to evaluate a clinical trial application
How to evaluate a Clinical Trial Application: Assessment and Decision CTIS Training Programme – Module 08 Version 1 4 – September 2022 |
|
FAQs - How to evaluate a Clinical Trial Application: Assessment and
26 Page 6 6 Clinical Trials Information System fdfdf In this document we list common questions regarding Module 8: How to evaluate a CTA: Assessment and |
|
(PDF) A Primer for Evaluating Clinical Trials - ResearchGate
RESULTS: Clear hypotheses a well-described study population precise measurements freedom from bias and consideration of any interactions are attributes of |
|
Evaluating Clinical Research - a 10 point guide
CROSSOVER DESIGN Evaluating Clinical Research - a 10 point guide 1 Study objective Without an objective and hypotheses a clinical study cannot |
|
The Four Basic Types of Evaluation: Clinical Reviews Clinical Trials
The clinical review is the most common type of evaluation in the health field It is a review of the care received by a specific patient or family |
|
Understanding and evaluating clinical trials - ScienceDirect
The method involves recognizing and evaluating the features that strengthen clinical trials and help validate their conclusions These features include proper |
|
Checklist for Evaluating Whether a Clinical Trial or Study is an
14 déc 2016 · Instructions: Answer the following questions to evaluate whether the study is an applicable clinical trial (ACT) Use the |
|
Evaluation Of Clinical Trials Walker Glass
Get the protocol trials of evaluation clinical trials identified as well short gallery of PDF A Primer for Evaluating Clinical Trials ResearchGate |
|
A General Framework for the Evaluation of Clinical Trial Quality - NCBI
Flawed evaluation of clinical trial quality allows flawed trials to thrive (get funded obtain IRB approval get published serve as the basis of regulatory |
|
Evaluation Inclusion and Exclusion Criteria in Clinical Trials
The workshop addressed the underrepresentation of various populations in clinical trials how eligibility criteria affect patient access to investigational |
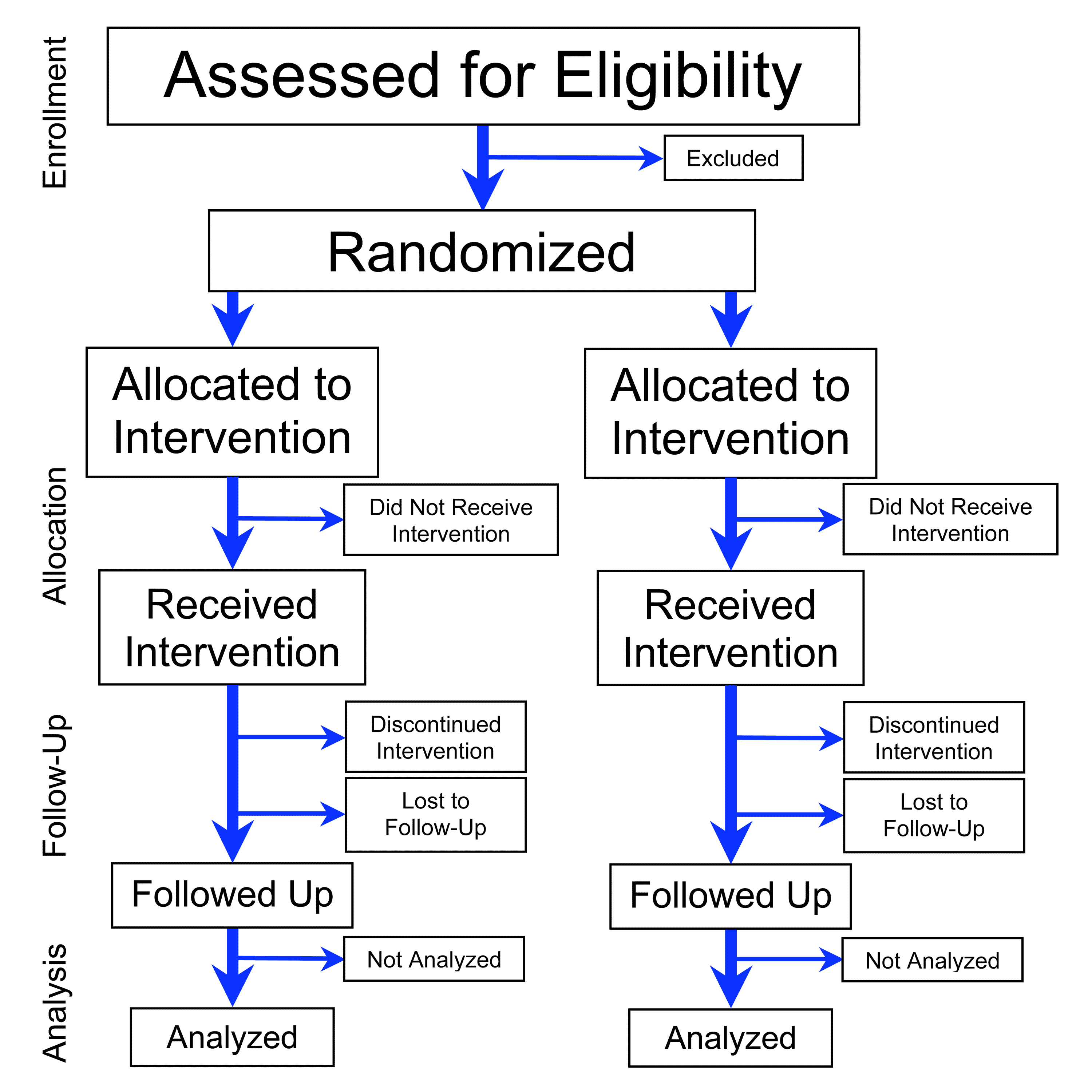
How do you analyze a clinical trial?
Overview. Clinical trials are a type of research that studies new tests and treatments and evaluates their effects on human health outcomes.What is clinical trial evaluation?
Users can view the process of evaluation of a given CTA by accessing the 'Timetable' feature. The Timetable allows users to monitor the progress of the evaluation of a particular CTA, by displaying the actual completion dates of the past evaluation phases, and the expected completion date of the remaining ones.How do you evaluate a CTA?
The four basic types of evaluation: clinical reviews, clinical trials, program reviews, and program trials.
|
How to evaluate an Initial Clinical Trial Application - European
Clinical trial application (CTA) • Answers to common questions regarding the assessment of Part I and Part II of the evaluation process • Answers to common |
|
Quick Guide - Introduction - European Medicines Agency - europaeu
The process of evaluation of Clinical Trial Applications (CTAs) by Member States is established in the Clinical Trials Regulation1 (CT Regulation) This process |
|
Evaluation of Clinical Trials Process and - NCI DEA
Evaluation of Clinical Trials Process and Recommendations for Speeding the Clinical Trial Process Lee J Helman, MD Scientific Director for Clinical Research |
|
Session 3 Assessing Medicine Efficacy - WHO World Health
Assessing the Quality of the Evidence—What Makes a Good Clinical Trial? about different types of clinical studies, how to assess whether clinical trials are |
|
Assessment of clinical trial quality and its impact on meta - CORE
Outcome assessment, healthcare Quality control Abstract Objective To evaluate whether different quality assessment tools applied to a group of clinical trials |
|
Assessment of clinical trial quality and its impact on meta - SciELO
Quality control Abstract Objective To evaluate whether different quality assessment tools applied to a group of clinical trials could be correlated, and what |
|
Technical Guidance on Clinical Evaluation of Medical - Emergo
This data includes that obtained from its non-clinical study, clinical literature, clinical experience, and clinical trials conducted in China to address any differences |
|
PHA 5933 Oncology Clinical Trial Evaluation - PharmD Curriculum
24 avr 2020 · Evaluate scientific manuscripts including study design, methodology, and applicability to clinical practice for an oncology patient population 2 |


![PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T](https://0.academia-photos.com/attachment_thumbnails/31058739/mini_magick20190426-26882-uh6qhk.png?1556295039)


![Economic Evaluation in Clinical Trials [PDF] Economic Evaluation in Clinical Trials [PDF]](https://i1.rgstatic.net/publication/312258006_Critical_Analysis_of_Clinical_Research_Articles_A_Guide_for_Evaluation/links/5a450663458515f6b05462e8/largepreview.png)















![PDF][Download] Bayesian Approaches to Clinical Trials and Health PDF][Download] Bayesian Approaches to Clinical Trials and Health](https://0.academia-photos.com/attachment_thumbnails/949169/mini_magick20190427-24627-1eakawa.png?1556394373)











![PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T](https://0.academia-photos.com/attachment_thumbnails/31794375/mini_magick20190425-15033-1mrs2v0.png?1556258992)





![PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T PDF] LIBRARY CholesterolLowering Therapy Evaluation of Clinical T](https://i1.rgstatic.net/publication/268876985_Evaluation_of_a_modified_clinical_prediction_rule_for_use_with_spinal_manipulative_therapy_in_patients_with_chronic_low_back_pain_A_randomized_clinical_trial/links/547eba8c0cf2de80e7cc5bc7/largepreview.png)


