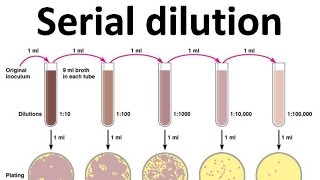
how to make 1 10 serial dilutions
|
Microbiologics Dilutions Guide
Dilution is the process of making a solution weaker or less concentrated. In microbiology serial dilutions (log dilutions) are used to decrease a bacterial |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
serial dilution of a bacterial culture. The initial step combines 1 unit volume culture (10 l) with 99 unit volumes of broth (990 l) = 1:100 dilution. In |
|
Serial-Dilution-Protocols.pdf
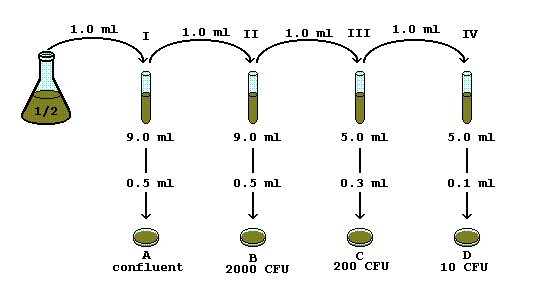
Sep 30 2005 The amount used to make that pour plate = 0.1ml (convert to 1/10 - it is easier to multiply fractions and decimals together). 51 colonies. = 51 ... |
|
Calculations There are a variety of calculations that you will be
For example for a series of 1:10 dilutions with final volumes of 10ml |
|
Dilution Chart
1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000. 2500ml 1000ml 500ml 250ml |
|
Titering phages Titering phages is probably the most common
10-8 dilution and so on through all of your serial dilutions from 10-8 to 10-1 |
|
Thermo Scientific Orion ISE Calibration: Preparing Ammonia
The following application note describes how to prepare ammonia standards through serial dilutions. the 10 ppm 1 ppm and 0.1 ppm standards into separate 150 ... |
|
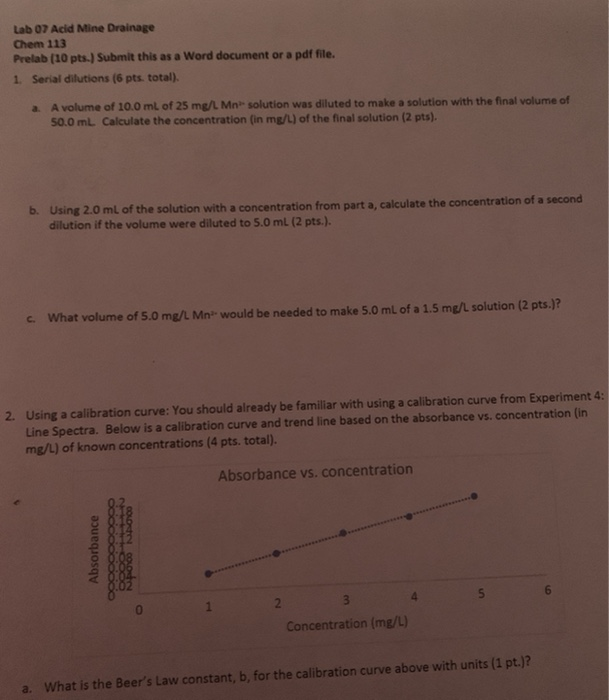
Lab 3: Making Solutions
prepare the dilutions. Table 3.5 Serial Dilution (1:10) of the 300X Stock CuSO4. Concen. -tration. Volume. Calculation of volume of stock needed. Vol. of. V1 to ... |
|
2 Amount and concentration: making and diluting solutions
Serial dilutions are a quick and convenient way to prepare a wide range of Take a 1mM stock solution of adrenaline dilute it 1:10 (0.1ml + 0.9ml) to give… |
|
Food Product Sampling and Standard Plating Methods
100 grams to make your initial 1:10 (10-1) dilution. 4. Stomach sample for 90 To produce 10-fold serial dilutions |
|
Laboratory Math II: Solutions and Dilutions
To make a complex solution with two or more solid solutes treat each solute individually. ? 5 liters of 50mM NaCl |
|
Microbiologics Dilutions Guide
of making a solution weaker or less concentrated. In microbiology serial dilutions (log dilutions) are used to decrease a bacterial concentration to a |
|
Product Manual
1 ml x 10 1. Prepare dilutions of the BSA standard solution as shown below. ... serial dilutions if necessary) into 1.5 ml microtubes. Perform at least ... |
|
Thermo Scientific Orion ISE Calibration: Preparing Ammonia
Prepare a 1 ppm ammonia standard by measuring 100 mL of the 10 ppm ammonia Please Note: A serial dilution could be started by making a 10 ppm ammonia ... |
|
Sample Validation Test (Spike and Recovery Test)
unknown preliminary testing of a serial-dilution of the sample |
|
Adeno-X™ Rapid Titer Kit User Manual
2005. 9. 30. Adeno-X Expression System User Manual (PT3414-1). Applications ... Using PBS or medium as diluent prepare 10-fold serial dilutions of your. |
|
Adeno-X™ Rapid Titer Kit User Manual
PT3651-1 www.clontech.com. Clontech Laboratories Inc. Version No. PR043520 Using PBS or medium as diluent |
|
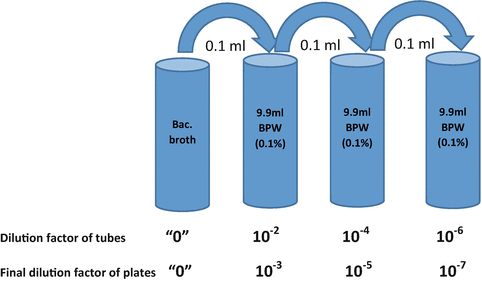
6223 easySpiralRange_B_EN v7.indd
easySpiral Dilute® is a 2-in-1 automatic diluter and plater which allows you to do 5 x 1/10th serial dilutions. It will then automatically plate on 1 |
|
Lentivirus titering protocol
24 hrs later make 5-fold serial dilution of viral stock in a round bottom 96-well 10. Mix by gently turn the plate from one side to another side;. |
|
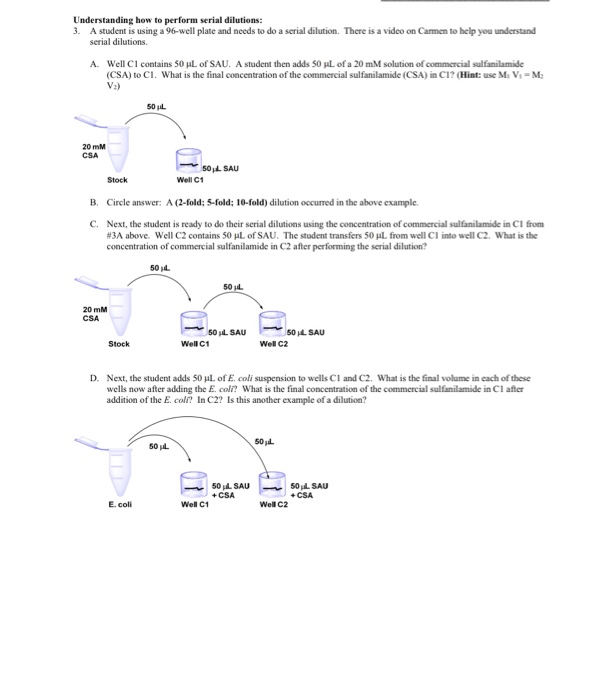
Cell Cloning by Serial Dilution 96 Well Plates Protocol
1. Cell Cloning by Serial Dilution in 96 Well Plates. Protocol This method is fast and easy; however like most ... 1 2 3 4 5 6 7 8 9 10 11 12. |
|
Serial Dilution Protocols - American Society for Microbiology
30 sept 2005 · 1ml added to 9ml = 1/10 (2nd tube) X previous dilution of 1/10 (1st tube) = total dilution of 1/100 for 2nd tube STEP 3: Determine the amount |
|
Formula 3 Serial Dilutions
This type of dilutions describes the ratio of the solute to the final volume of the dilute solution -For example to make 1:4 dilution of a 2 0 M KCl |
|
Serial Dilutions - Biomol
A serial dilution is a step-wise and geometric series of dilutions which starts with a small amount of starting material and amplifies the dilution factor |
|
Serial Dilution Method Definition Procedure Application
18 jan 2023 · The main purpose of serial dilution technique is to find out the concentration or the cell counts of an anonymous sample by counting the number |
|
How to Make a Serial Dilution - ASSET
Serial dilutions are a step by step dilution of a solution The following example is for a tenfold dilution where the dilution factor is 1:10 at each dilution |
|
Microbiologics Dilutions Guide
In microbiology serial dilutions (log dilutions) are used to Example: 1:10 dilution - if the concentration is 1000 CFU a one log |
|
Making Serial Dilutions - PhagesDBorg
Remove 10 ?L of the “neat” (undiluted) phage sample and add it to the first dilution tube (labeled 10–1) Vortex for five seconds to mix 4 Remove 10 ?L with |
|
Accurate and consistent Serial Dilutions made easy with Andrew
Figure 2: The three steps in the example 1/10 serial dilution where the final volume is 180 µl 1 Add sample (20 µl) 3 Move to next |
|
Serial Dilution Protocol PDF - Biology Exams 4 U
Transfer 1 ml of solution from test tube C to test tube D and mix well and continue up to the required dilution Here we are making 10-4 dilution Serial |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
Anderson's How to Make Simple Solutions and Dilutions webpage) milligram = mg = 1/1000 of a g or 10-3 g serial dilution of a bacterial culture |
How do you make a 1 10 serial dilution?
1:10 dilution is performed by placing 1 ml of the re-hydrated pellet solution into 9 ml of buffer = 100 - 1,000 CFU/ml. 3. 0.1 ml of the organism suspension plated to an agar = 10 - 100 CFU per 0.1 ml. 99 ml 3.5 x 101 (35 CFU/ml) 1 ml from the 99 ml solution will provide 35 CFU/ml.How do you make a 1 10 dilution from 1 100?
Serial dilutions are often performed in steps of 10 or 100. They are described as ratios of the initial and final concentrations. For example, a 1:10 dilution is a mixture of one part of a solution and nine parts fresh solvent. For a 1:100 dilution, one part of the solution is mixed with 99 parts new solvent.- For example, a 10:1 ratio means you mix 10 parts water to 1 part chemical. The amount of each liquid changes depending on the ratio used, and the size of the container.
|
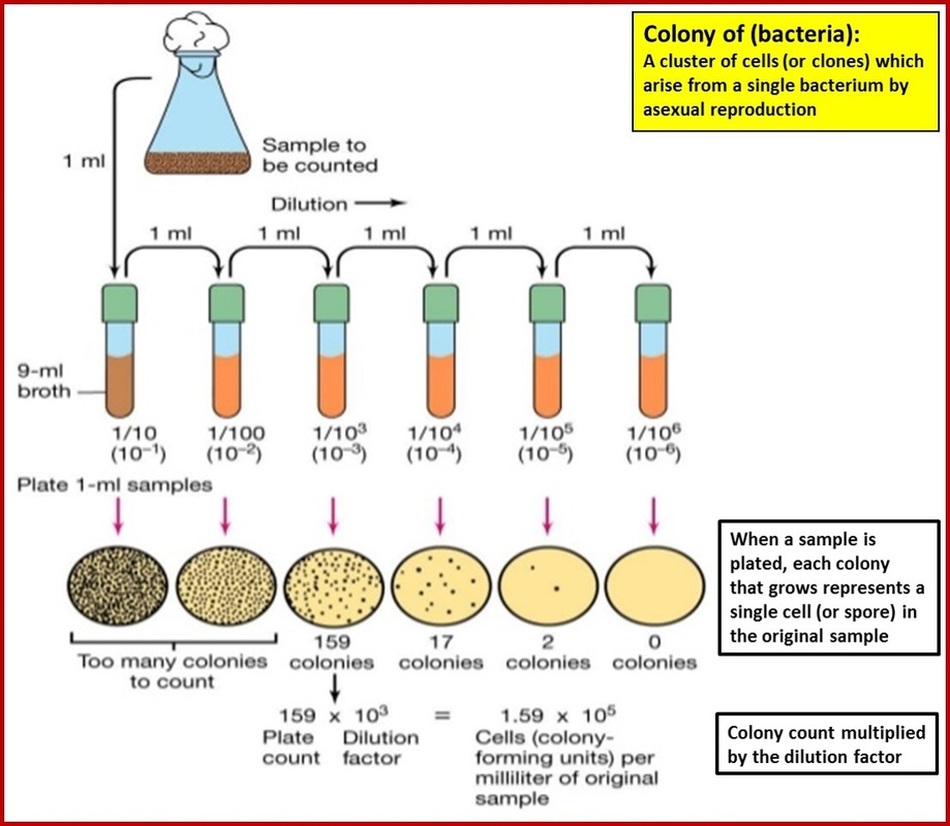
Serial Dilutions
What is the total dilution that we need to make? Stock concentration: 10 mg/mL= 10-2 g/mL Final concentration: 1 ng/mL = 10-9 g |
|
How to Make a Serial Dilution - ASSET - Cornell University
Pipette 20 μl of the D1 solution into your D2 tube Gently press the plunger on the pipette up and down to mix Now you have a dilution of 1:100 (10-2) |
|
Serial Dilutions
Traditionally serial dilutions were done with 1mL pipettes and 99mL sterile dilution bottles because they were If you start out with a broth culture and do a 10 -6 dilution Using a new sterile 1mL pipette, tranfer 1mL from the 1:10,000 ( 10 -4 ) |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
milligram = mg = 1/1000 of a g or 10-3 g gram = g However, if you are asked to make a 1:3 dilution of acetic acid in ethanol, you would mix A serial dilution is simply a series of simple dilutions which amplifies the dilution factor quickly |
|
Math and Dilutions
Microbiology is full of Math and Dilution Terminology Luckily, once 10-1=1/10= 0 1 (Ten to the minus one or one tenth) make a serial dilution of 10-2 overall |
|
Dilutions Occasionally a solution is too concentrated to be used as it
1 Make a 1 to 10 dilution of serum in saline 2 Make a 1 in 10 dilution of serum in saline 3 Make a This type of dilution series is referred to as a serial dilution |
|
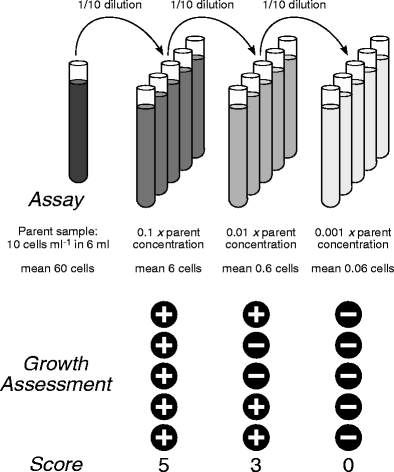
SERIAL DILUTIONS – TUBE METHOD Principle Serial dilution is a
A small amount of serum or solute can be serially diluted by transferring aliquots to diluent One of the most common series doubles the dilution factor with each |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
looking at how to make a solution from a solid substance or “solute” being This means that you add 10 milliliters of the 5M NaCl stock solution to a While serial dilutions are a great way to save on both reagents and space in the lab, |
|
Making Dilutions
You could make a liter of 10mM NaCl with 0 29 g, because Making Dilutions 3 Figure 1 A serial dilution A small volume (vt) of solution is transferred to a |



























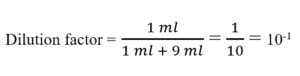



![Solved] Your Turn In lab you will use a stock solution that is 2 Solved] Your Turn In lab you will use a stock solution that is 2](http://sciencefair.math.iit.edu/techniques/SerialDilution/Serial_dilution.jpg)