phosphate buffer preparation of 01 M potassium phosphate buffer at
|
phosphate bufferpdf
Gomori buffers the most commonly used phosphate buffers consist of a mixture of monobasic dihydrogen preparation of 0 1 M potassium phosphate buffer at 25° |
|
Preparation of 01M Potassium Phosphate Buffer at 25°C
Preparation of 0 1M Potassium Phosphate Buffer at 25°C Desired Volume of 1M Dilute the combined 1M stock solution to 1 litre with distilled H2O |
|
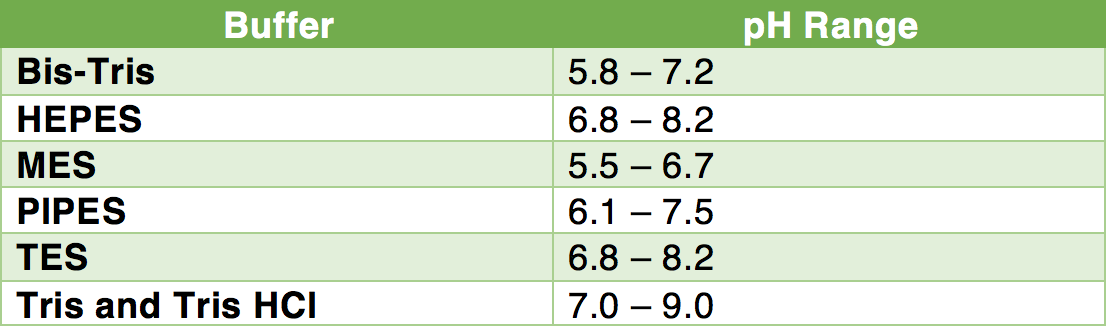
413 BUFFER SOLUTIONS
Dissolve 2 72 g of potassium dihydrogen phosphate R in 800 ml of water R Adjust the pH (2 2 3) with 1 M potassium hydroxide and dilute to 1000 ml with water R |
|
POTASSIUM PHOSPHATE
Phosphates have a very high buffering capacity and are highly soluble in water They are widely used despite a number of potential disadvantages: |
|
Potassium phosphate dibasic and monobasic
In addition to helping maintain pH it supplies essential phosphate Typical Buffer Preparation/Formulations: Prepare the following solutions: 1 M Potassium |
|
Potassium phosphate buffer 01 M pH 78 Phenylbutazone (Sigma
Potassium phosphate buffer 0 1 M pH 7 8 PB (500 txM) is preincubated for 3 min with ram seminal vesicle micro- somes or another PGH synthase preparation in |
|
Media 1M Potassium phosphate buffer pH 6
17 jan 2020 · › 868 mL 1 M KH2PO4 › Procedure Preparation of 1M Potassium phosphate buffer 1 Combine 132 mL of 1 M K2HPO4 868 mL of 1 M KH2PO4 and |
|
Buffer solutions
The buffer solution contains sodium chloride sodium phosphate and/or potassium phosphate Laboratories may follow several ways for the preparation of phosphate |
How do you make a 0.01 M phosphate buffer?
0.
1) M Phosphate Buffer is used in the preparation of Sucrose solutions for cryoprotection.
Note: Research reported in this publication was supported by the National Institute Of Mental Health of the National Institutes of Health under Award Number U19MH114830.How do you make a 1 M potassium phosphate buffer?
Weigh out the KH2PO4 and K2HPO4.
Dissolve the KH2PO4 and K2HPO4 in 100 ml of deionized water.
Adjust the pH of the solution to 7.5 using a pH meter.
Store the buffer in a cool, dark place.What is 0.1 M phosphate buffer?
0.
1) M Phosphate buffer solution pH 7.0.
Dissolve 1.361 g of potassium dihydrogen phosphate R in water R and dilute to 100.0 ml with the same solvent.
Adjust the pH (2.2. 3) using a 35 g/l solution of disodium hydrogen phosphate R.
|
TECHNICAL INFORMATION
– 1 M Potassium Phosphate Monobasic. – 1 M Potassium Phosphate Dibasic. – 5 M Sodium Chloride. Formulation Guide: 1 Liter of 0.05 M Phosphate Buffer. (25°C). 1 |
|
4.1.3. BUFFER SOLUTIONS
Dissolve 6.57 g of potassium chloride R in water R and add. 119.0 mL of 0.1 M hydrochloric acid. Dilute to 1000.0 mL with water R. Phosphate buffer solution pH |
|
TECHNICAL INFORMATION
– 1 M Potassium Phosphate Monobasic. – 1 M Potassium Phosphate Dibasic. – 5 M Sodium Chloride. Formulation Guide: 1 Liter of 0.05 M Phosphate Buffer. (25°C). 1 |
|
Calbiochem
(a) 0.1 M Potassium chloride : 7.45 g/l (M.W.: 74.5). (b) 0.1 M Hydrochloric Adjust the pH before use. Phosphate Buffered Saline (PBS) pH 7.4. 150 mM NaCl. |
|
POTASSIUM PHOSPHATE
Gomori buffers the most commonly used phosphate buffers |
|
Potassium phosphate dibasic trihydrate (P5504) - Product
A table for preparation of 0.1 M potassium phosphate buffer at 25 °C using various proportions of potassium phosphate monobasic and potassium phosphate |
|
4.1.3. BUFFER SOLUTIONS
Dissolve 6.57 g of potassium chloride R in water R and add. 119.0 ml of 0.1 M hydrochloric acid. Dilute to 1000.0 ml with water R. Phosphate buffer solution pH |
|
Potassium phosphate dibasic and monobasic
In addition to helping maintain pH it supplies essential phosphate. Typical Buffer Preparation/Formulations: Prepare the following solutions: 1 M Potassium |
|
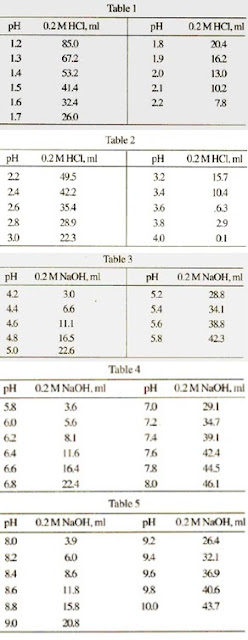
Preparation of 0.1M Potassium Phosphate Buffer at 25°C
Page 1. Preparation of 0.1M Potassium. Phosphate Buffer at 25°C. Desired. Volume of 1M. Volume of 1M. pH. K2HPO4 (mL). KH2PO4 (mL). 5.8. 8.5. 91.5. 6.0. 13.2. |
|
Phosphate buffer preparation of 0.1 M potassium phosphate buffer at
Information from cshprotocols.org: Gomori buffers the most commonly used phosphate buffers |
|
POTASSIUM PHOSPHATE
potassium phosphate monobasic Monopotassium phosphate |
|
Phosphate buffer preparation of 0.1 M potassium phosphate buffer at
updated 1/15/2007 phosphate buffer. Information from cshprotocols.org: Gomori buffers the most commonly used phosphate buffers |
|
Media 1M Potassium phosphate buffer pH 6
21?/10?/2020 132 mL 1M K2HPO4. › 868 mL 1 M KH2PO4. › Procedure. Preparation of 1M Potassium phosphate buffer. 1. Combine 132 mL of 1 M K2HPO4 ... |
|
Change in Invertase Activity of Sweet Potato in Response to
sterilized with 0.1% sodium hypochlorite solution followed by thorough washing with tap water. 10 mm potassium phosphate buffer pH 7.0 |
|
Preparation of 0.1M Potassium Phosphate Buffer at 25°C
Preparation of 0.1M Potassium. Phosphate Buffer at 25°C. Desired. Volume of 1M. Volume of 1M. pH. K2HPO4 (mL). KH2PO4 (mL). |
|
Technical Information - 1. Preparation of Mobile Phase for HPLC
(e.g.1) Preparation of 20 mmol/l phosphate buffer (pH2.5). 1. Preparation of 20 mmol/l sodium Sodium 1-hexanesulfonate (0.5 M solution) (Product No. |
|
Potassium phosphate dibasic and monobasic
In addition to helping maintain pH it supplies essential phosphate. Typical Buffer Preparation/Formulations: Prepare the following solutions: 1 M Potassium |
|
Common Buffers and Stock Solutions
3 prepare closest higher pH |
|
Preparation of Buffers Preparation of Buffers
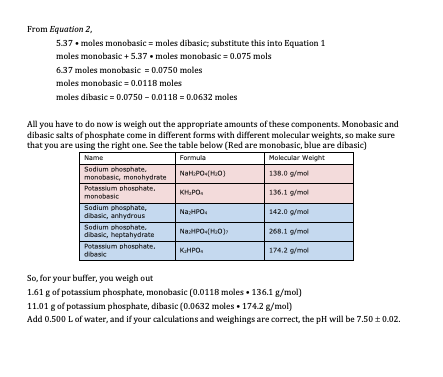
21/01/38. 7. ? Example: Describe the preparation of 10 liters of 0.045M potassium phosphate buffer pH= 7.5? • From the previous example [HA] = 0.075 molar |
|
4.1.3. BUFFER SOLUTIONS
Dissolve 1.02 g of potassium hydrogen phthalate R in. 30.0 ml of 0.1 M sodium hydroxide. Dilute to 100.0 ml with water R. 0.067 M Phosphate buffer solution pH |