hydrolysis of acid anhydride mechanism
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: V
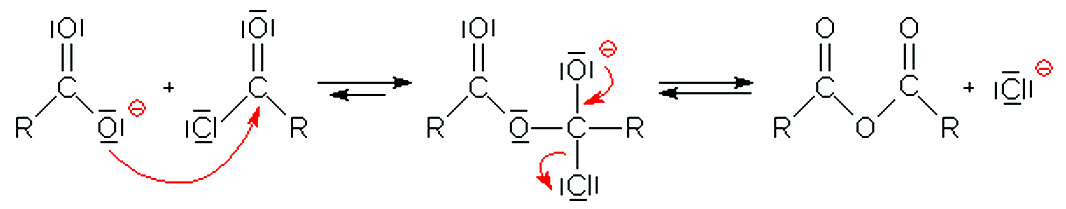
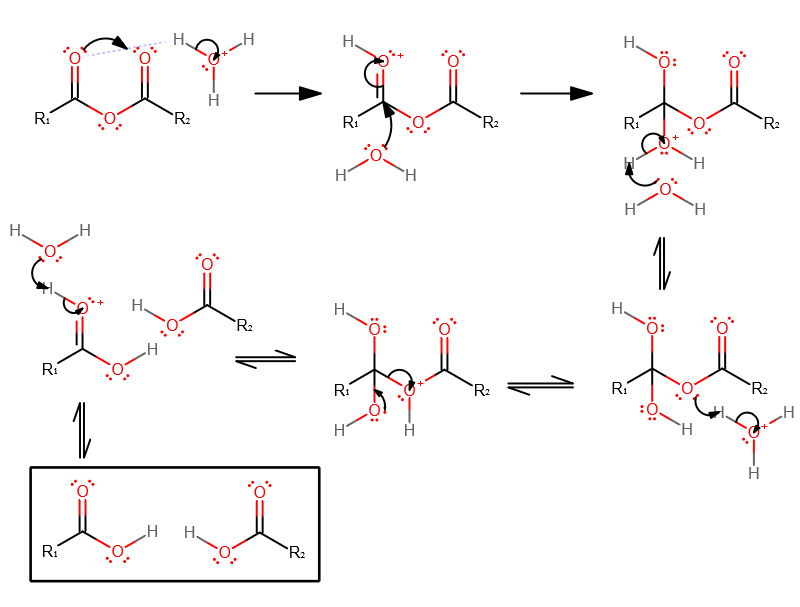
The acid-catalyzed hydrolysis of acetic anhydride has been studied by Kilpatrick (I) and by Gold and I-Iilton (23). It is i~sually accepted that the first step |
|
PicoSpin 45: Hydrolysis of Acetic Anhydride with Heavy Water (D2O)
The hydrolysis of acetic anhydride (Ac2O) to acetic acid (AcOH) serves as a Lowry T.H.; Richardson |
|
Kinetics and Activation Energy Parameters for Hydrolysis of Acetic
The hydrolysis reaction of acetic anhydride in a water/acetone cosolvent systems forming acetic is pseudo-first order. The reaction mechanism begins by acetic |
|
The Hydrolysis of Carboxylic Anhydrides. V.12 The Acid Hydrolysis
The acid hydrolysis of trimethylacetic anhydride follows an A-2 mechanism in both water and aqueous di- oxane and is slower than that of acetic anhydride. |
|
Solvent Isotope Effects in Catalyzed Hydrolysis of Carboxylic Acid
The latter mechanism is discarded for carboxylic acid derivatives with good leaving groups two mechanisms; benzoic anhydride hydrolysis. |
|
The Hydrolysis of Substituted Benzoic Anhydrides1
The mechanism of anhydride hydrolysis is discussed. Although the hydrolysis of aromatic esters acid chlorides and amides has been frequently studied. |
|
Reactions of Anhydrides Mechanism of Anhydride Substitution
Esters are hydrolyzed with water in the presence of either acid or base to form carboxylic acids or carboxylate anions respectively. Reactions of Esters. • |
|
ACETIC ANHYDRIDE HAZARD SUMMARY IDENTIFICATION
* Acetic Anhydride is a HIGHLY CORROSIVE. CHEMICAL and contact can severely irritate and burn the skin and eyes with possible eye damage. * Breathing Acetic |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
Very fast zinc-catalyzed hydrolysis of an anhydride. Model for the
Received August 9 1974. Very Fast Zinc-Catalyzed Hydrolysis of an. Anhydride. A Model for the Rate and Mechanism of Carboxypeptidase A Catalysis. |
|
Chapter 21: Carboxylic Acid Derivatives
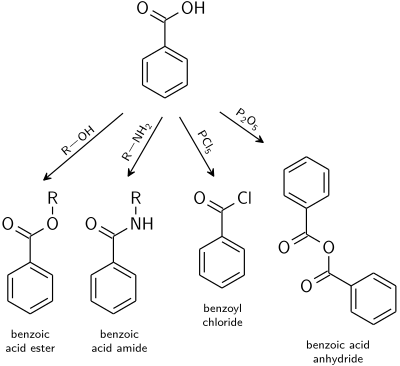
Hydrolysis of Acid Derivatives (21-7) This reaction is the reason all of these compounds are considered acid derivatives because they produce carboxylic acids upon hydrolysis This reaction can be performed under acidic or basic conditions Acid Chlorides/Anhydrides These two compounds are so reactive that the |
|
General Reaction of Anhydrides Reactions of Anhydrides
• Amides are hydrolyzed in acid or base to form carboxylic acids or carboxylate anions • In acid the amine by-product is protonated as an ammonium ion whereas in base a neutral amine forms Reactions of Amides • The mechanism of amide hydrolysis in acid is the same as the mechanism of ester hydrolysis in acid |
|
Acetic anhydride - Wikipedia
Hydrolysis of Maleic Anhydride Anhydrides are derivatives of carboxylic acids All derivatives ofcarboxylic acids can be converted to the corresponding acid through reactionwith water Some less reactive derivatives require an catalyst for thereaction to occur in a reasonable time but anhydrides do not |
|
The Simple and General-Base Catalyzed Aqueous Hydrolyses of
two-step mechanism for simple hydrolysisbelow room temperatureand a single-step mechanism for propanoate- and butanoate-catalyzed hydrolysis for all temperatures Implications of these results and comparison with acetic anhydride hydrolysis are discussed |
|
Searches related to hydrolysis of acid anhydride mechanism filetype:pdf
Hydrolysis of the anhydride ~6-mL of distilled water was added to 1 23 g of anhydride and the mixture heated until boiling The diacid was crystallized in an ice-water bath and collected via vacuum filtration Melting points and test for unsaturations (Bromine and Baeyer test) |
What catalysts are used in anhydride hydrolysis?
- The Tennessee Eastman acetic anhydride process involves the conversion of methyl acetate to methyl iodide and an acetate salt. Carbonylation of the methyl iodide in turn affords acetyl iodide, which reacts with acetate salts or acetic acid to give the product. Rhodium chloride in the presence of lithium iodide is employed as catalysts.
What is the reaction of anhydride hydrolysis?
- Hydrolysis of the anhydride causes a constant additional delivery of vesicle-forming material, so that the amount of vesicle present increases during the poly (A) synthesis. These experiments demonstrated a model for a minimal cell.
How is the hydrolysis of an anhydride into a carboxylic acid achieved?
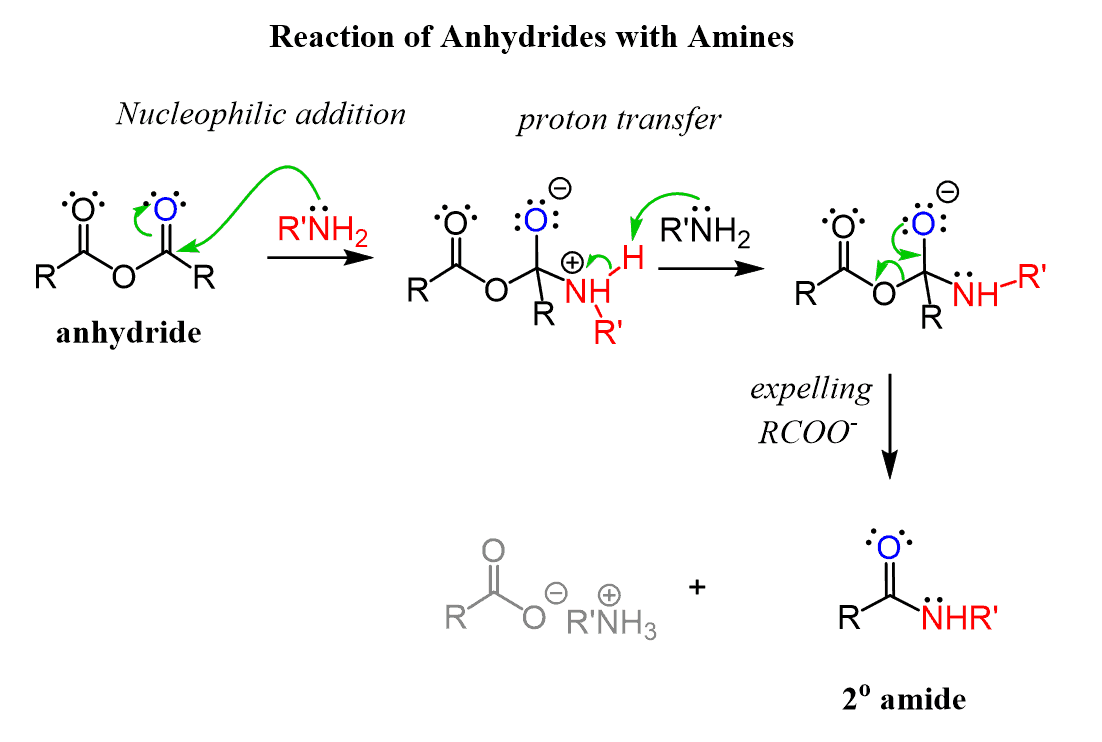
- Acid anhydrides undergo hydrolysis and nucleophilic acyl substitution reactions. Acid anhydrides readily hydrolyze to carboxylic acids. In many cases, this reaction is an unwanted side reaction and steps will be taken in the lab to keep the system "dry" (aka water free). The presence of pyridine facilitates proton transfers during the reaction.
What is the electron-dense atom in anhydride hydrolysis?
- It is practically identical for anhydrides as it is for acyl halides (particularly acyl chlorides) and esters, and similar variations are seen in the acid-catalyzed hydrolysis of nitriles, amides, etc, wherein the electron-dense atom (e.g. O in a carbonyl or N in a nitrile) takes on a proton and becomes susceptible to nucleophilic attack.
|
Reactions of Anhydrides Mechanism of Anhydride Substitution
Esters are hydrolyzed with water in the presence of either acid or base to form carboxylic acids or carboxylate anions, respectively Reactions of Esters • Basic |
|
Hydrolysis of Acetic Anhydride with Heavy Water - Thermo Fisher
In a hydrolysis reaction, a chemical bond is broken by the addition water The hydrolysis of acetic anhydride (Ac2O) to acetic acid (AcOH) serves as a model example of the hydrolysis reaction Acetic anhydride rapidly hydrolyzes in the presence of water, alcohol and catalyzing acid, in this case water |
|
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
derivatives (acid chlorides and anhydrides) carboxylic acid amide acid chloride acid anhydride Mechanism of the base-promoted hydrolysis, Fig 20 4, p 851 |
|
Carboxylic acid deri
General mechanism: acid ester amide acid chloride acid anhydride thioester acyl phosphate acid Mechanism of the base-promoted hydrolysis (Figure 21 9) |
|
Carboxylic Acid Derivatives Addition/Elimination
The Mechanism of formation starting with an anhydride: addition/elimination again The products for anhydride hydrolysis are two carboxylic acids, the extent to |
|
II CARBOXYLIC ACIDS AND DERIVATIVES One of the more
Nitriles can be hydrolyzed to the corresponding carboxylic acids incorporation of two moles of Grignard (or hydride), in a mechanism which involves an As with acid halides, reduction of anhydrides with LiAlH4 results in the addition of two |