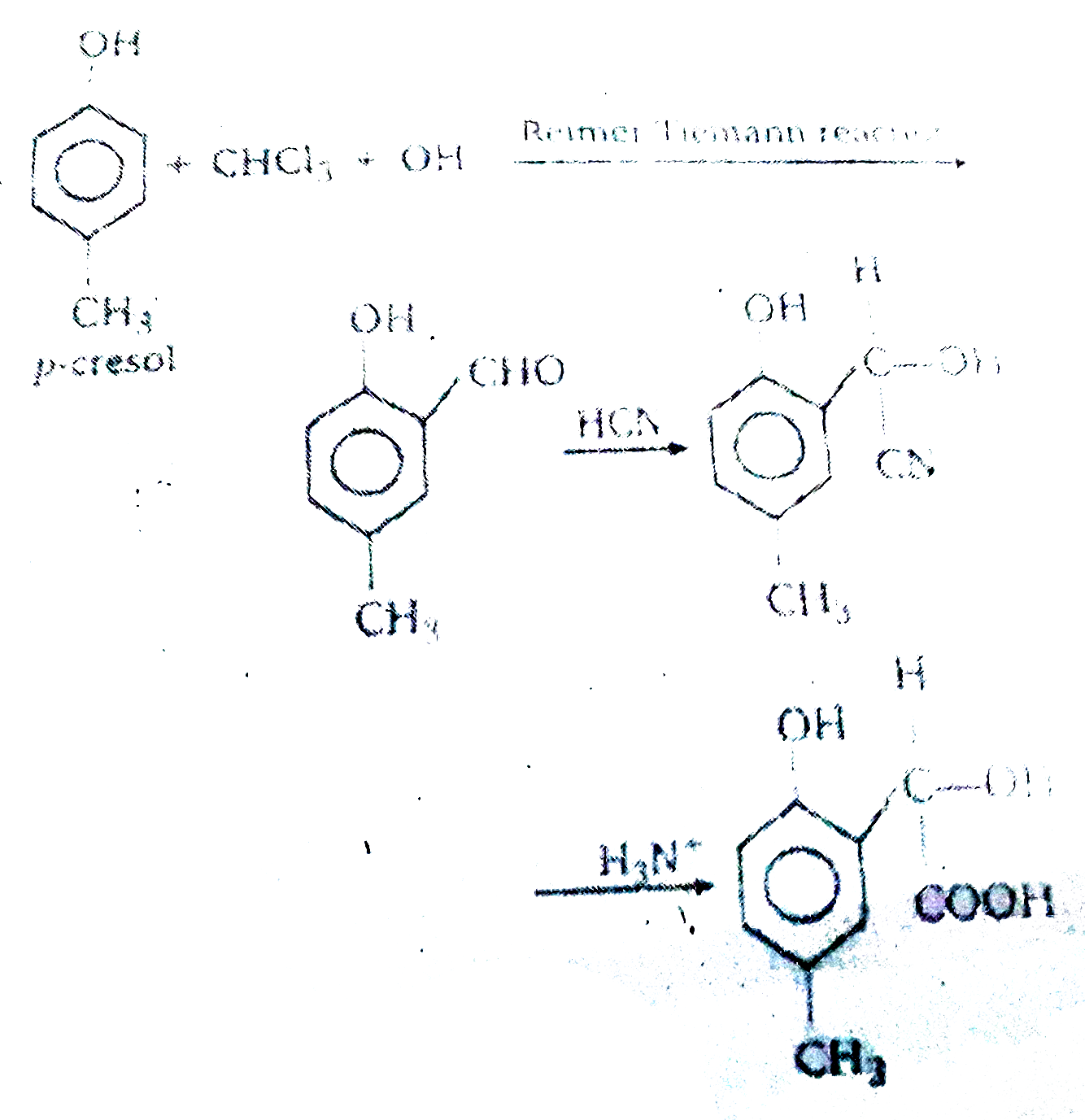
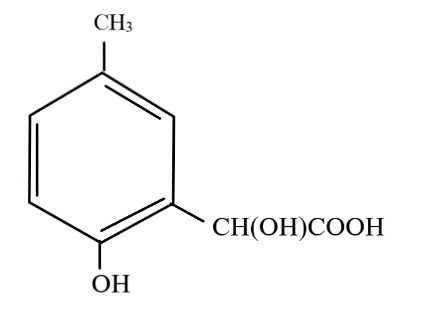
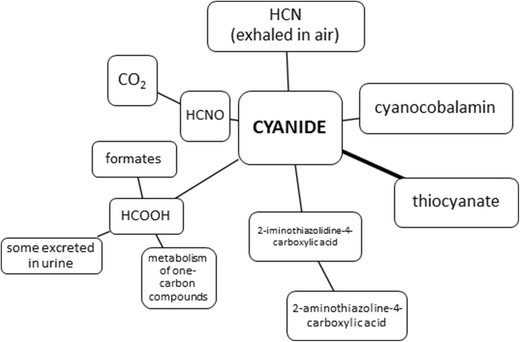
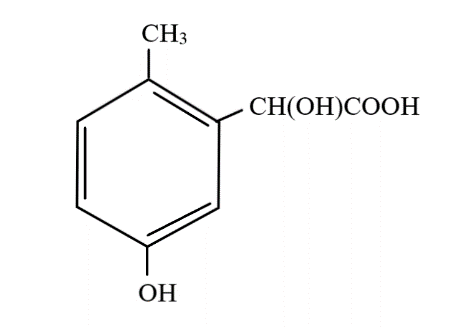
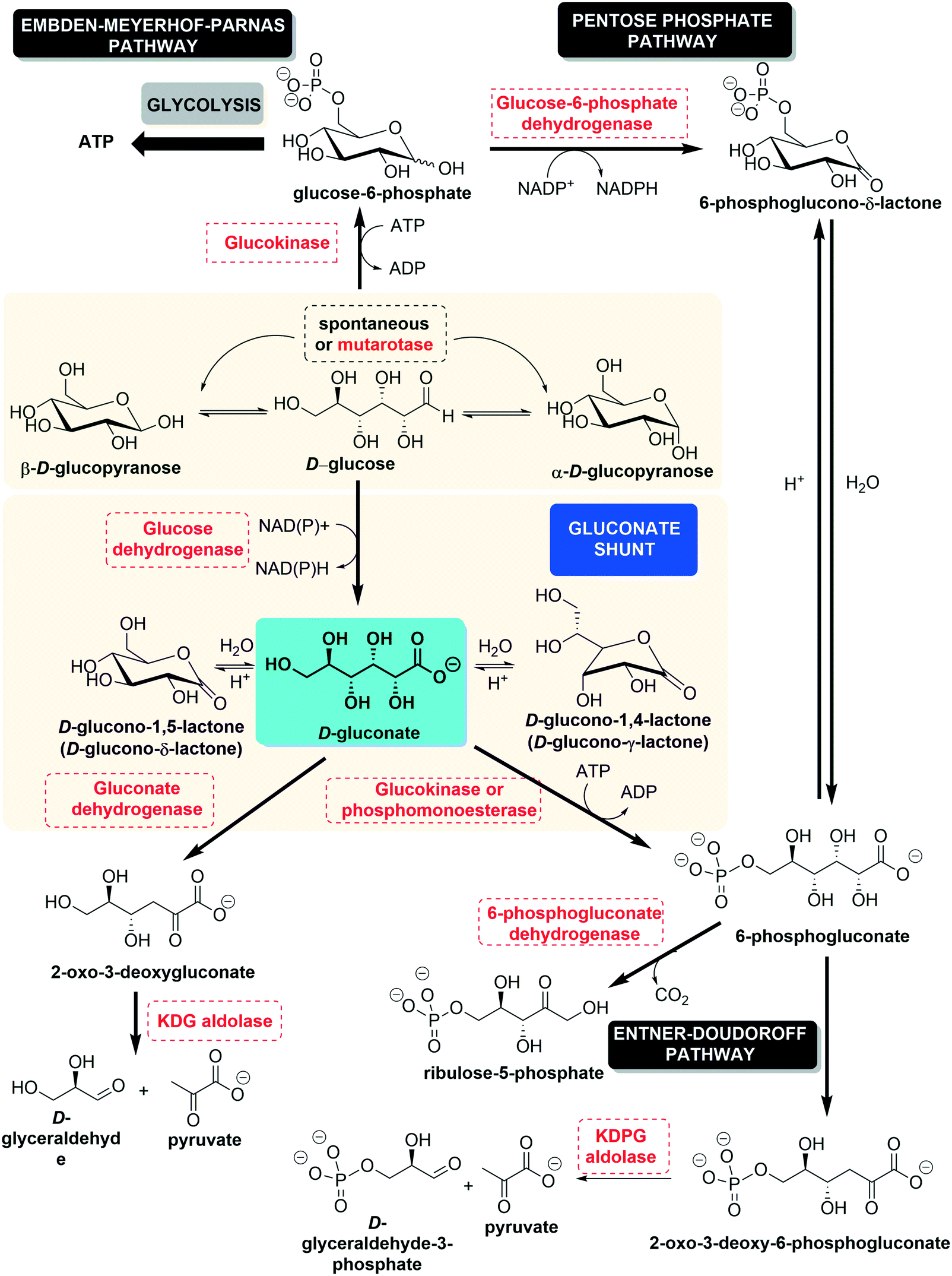
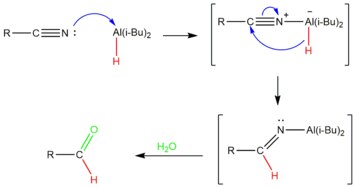
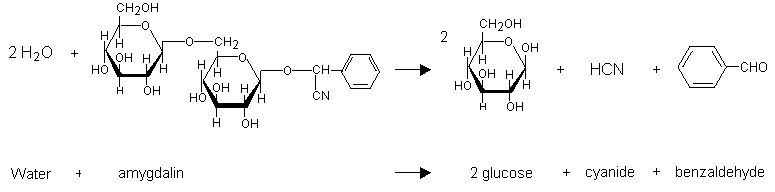
hydrolysis of cyanide in acidic medium
|
THE HYDROLYSIS OF HYDROGEN CYANIDE BY ACIDS1
Air freed of ammonia was sucked through this solution and then through standard acid. The excess acid was titrated and the amount of am- monium salt present |
|
A Mechanism for the Hydrolysis of Cyanamide in Acid Solution
hydrobromic sulfuric and nitric acid solution |
|
TOXICOLOGICAL PROFILE FOR CYANIDE
However the relatively slow rates of hydrolysis reported for hydrogen cyanide in acidic solution (Krieble and McNally 1929;. Krieble and Peiker 1933) and of |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
In general nitriles and amides are exceptionally stable to acid and basic and nitriles using a methanolic solution of NaOH in a less polar aprotic solvent. |
|
Acid–base behaviour of the ferricyanide ion in perchloric acid media
In basic medium cyanide reduces femcyanide to ferrocyanide in 48 11; this but in acid medium this reaction is not possible |
|
1448 J.C.S. Perkin I1
withdrawing cyano-group. CL-CYANOBENZYLILENEANXLINES (I) are fairly readily hydrolysed in acidic or alkaline solution but not in and hydrogen cyanide only. |
|
Partial hydrolysis and acetolysis of cellotriose-1-14C
hydrolysis of glycosides in acidic solution is a reaction well known to the nitrile was hydrolyzed in solution by heating at 80° for 5 hr. The solution ... |
|
THE ACID HYDROLYSIS OF GLYCOSIDES: I. GENERAL
In a very strongly acidic solution the conversion of the glycoside to its conjugate acid Use of mercuric cyanide. (43 44) rather than silver carbonate always ... |
|
Detoxification Effect of Fermentation on Cyanide Content of Cassava
hydrocyanic acid on hydrolysis. This is Generally there was a decrease in pH value with days indicating that the medium becomes more acidic with days of. |
|
Fast disproportionation of hexacyanomanganate(III) in acidic
The uv spectra of. M~(CN)~- in acidic and cyanide-containing solutions differ in acidic medium with time. Reaction of decomposition: Mn(IV) 5 X. M ; H2SO4 ... |
|
Dry hydrolysis of nitriles by sodium perborate and copper salts in
Nitriles react with sodium perborate or copper salts to yield carboxylic acids under mild acidic conditions. The multiphase reaction can be carried out in |
|
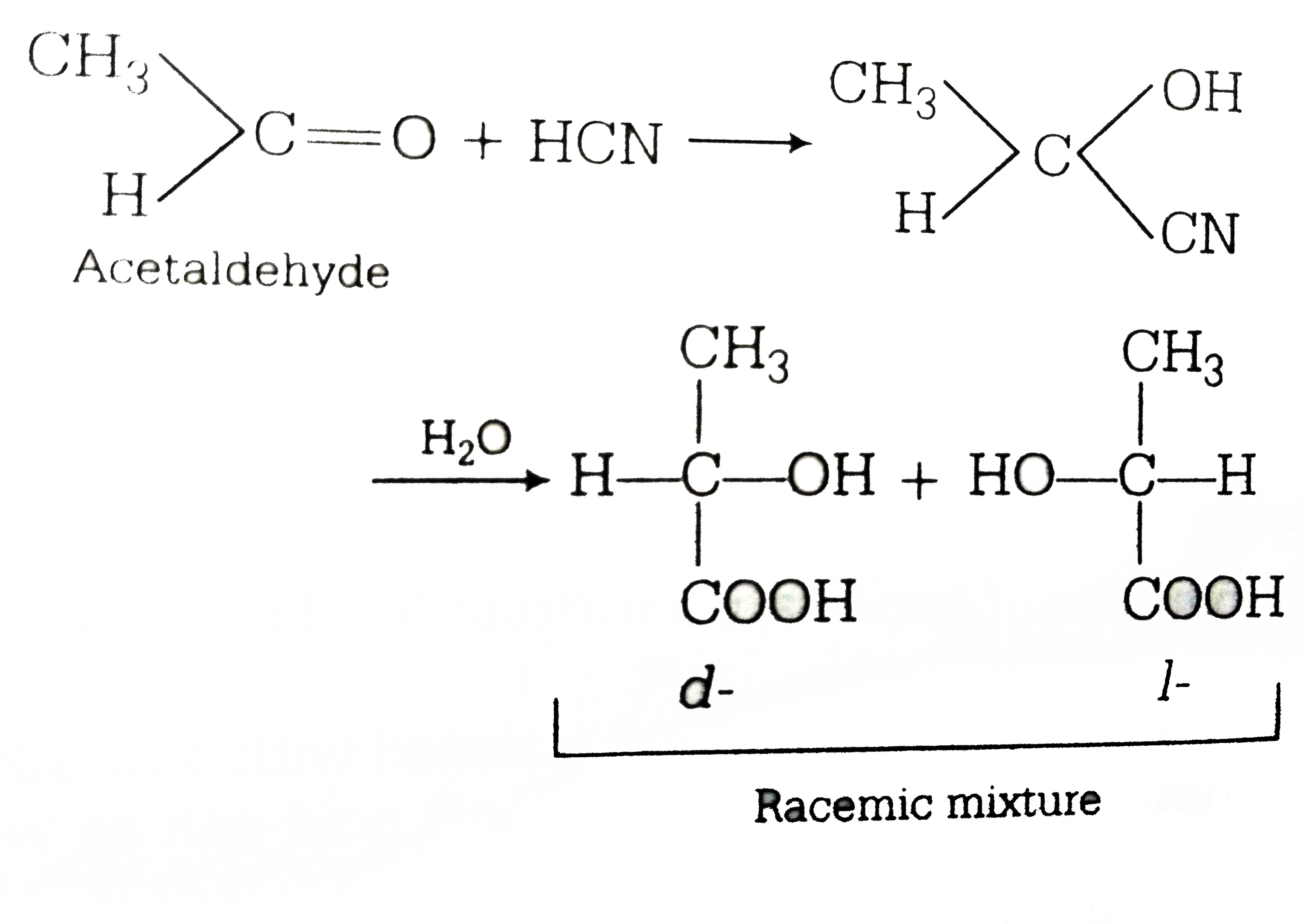
Anchimeric?Assisted Spontaneous Hydrolysis of Cyanohydrins
Conditions: Implications for Cyanide Initiated Selective Transformations. both the acidic and basic conditions but hydrolysis to the amide was observed ... |
|
Kinetics of Alkoxysilanes and Organoalkoxysilanes Polymerization
21 mars 2019 The activation energy in the acidic medium ranged from 11 to 16 kcal mol?1. The hydrolysis rate constant ranged between 0.002 and 0.5 M?1 ... |
|
TOXICOLOGICAL PROFILE FOR CYANIDE
slow rates of hydrolysis reported for hydrogen cyanide in acidic solution (Krieble and McNally 1929;. Krieble and Peiker 1933) and of cyanides under |
| Enzymatic hydrolysis of heterocyclic nitriles |
|
The Hydrolysis of Hydrogen Cyanide by Acids. II
ion concentration of the solution. tion ofthe hydrolysis of hydrogen cyanide with mineral acids are given. ... able to obtain the acidity. |
|
Partial hydrolysis and acetolysis of cellotriose-1-14C
addition using sodium cyanide-C14 hydrolysis of the resultingnitrile |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
Nitriles are hydrolyzed first to amides and further to carboxylic acids and ammonia with even more strong reaction conditions. The hydrolysis of amides and |
|
A mild and Selective Method of Ester Hydrolysis
nitriles remain unaffected under this condition and 6-keto double bonds3 under acidic hydrolysis are also possible. On the other hand hydrolysis under ... |
|
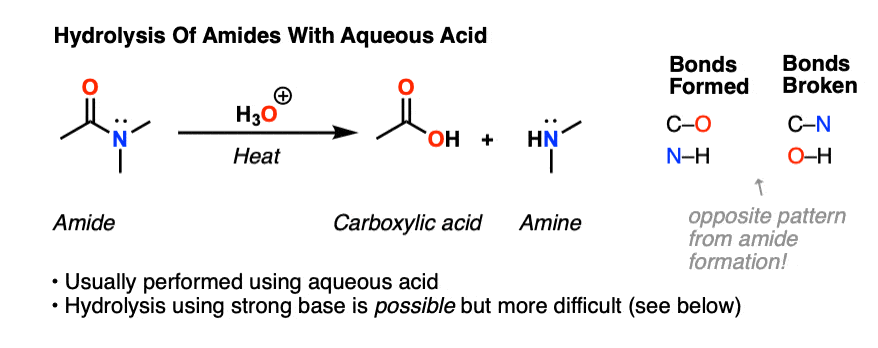
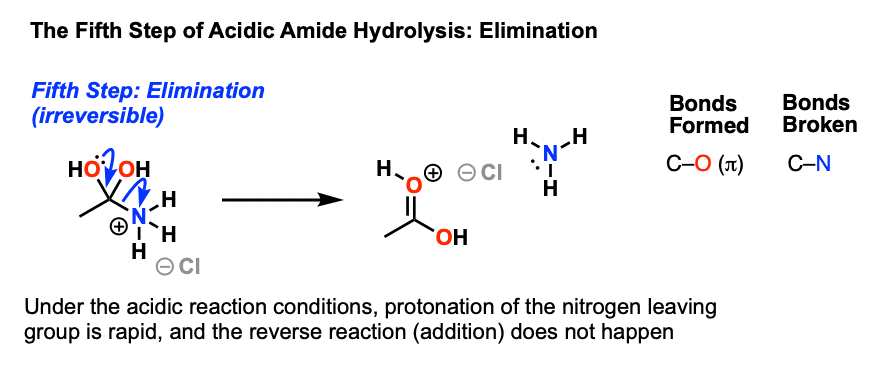
Hydrolysis of Nitriles
O H + + Page 8 Hydrolysis of Nitriles In basic solution the carboxylic acid product is deprotonated to give a carboxylate ion + – RCO O HO + – |
|
NF Jain and CE Masse Carboxylic acid - thieme-connectde
Hydrolysis of carboxylic acid esters, amides, and nitriles can be either catalyzed by achieved by the use of a large excess of water in the reaction medium |
|
Theoretical study of nitrile hydrolysis by solid acid catalysts - Pure
1 jan 2000 · Subject headings: reaction mechanism; hydrolysis/ nitrile/ quantum shown that acetonitrile can be protonated in super-acid medium as |
|
Reductive hydrolysis of nitriles to aldehydes by - ScienceDirectcom
The reductive hydrolysis of nitriles to the corresponding aldehydes has been in two ways: (i) by means of catalytic hydrogenation in sulfuric acid medium |
|
PDF (Chapter 24) - Caltech Authors
Primary amides have two N-H bands of medium intensity near 3500 cm-l The hydrolysis of nitriles is a satisfactory method for preparation of unsubsti- tuted amides Generally, amides can be hydrolyzed in either acidic or basic solution The |