hydrolysis of ethylethanoate in acid produces
|
Value of Coproduction of Ethanol and Furfural from Acid Hydrolysis
15 mai 1984 furfural coproduced with ethanol by the acid hydrolysis of cellulose can be produced at a lower unit cost and .in sufficient quantity to ... |
|
Lignocellulosic Biomass to Ethanol Process Design and Economics
acid prehydrolysis with simultaneous saccharification (enzymatic) and better cellulose hydrolysis enzymes and ethanol-fermenting organisms ... |
|
NREL 2012 Achievement of Ethanol Cost Targets: Biochemical
enzymatic hydrolysis and cofermentation of the resultant biomass sugars to polymer-to-sugar yields and sugar-to-ethanol yields because acetic acid ... |
|
Process Design and Economics for Biochemical Conversion of
Ethanol. Dilute-Acid Pretreatment and. Enzymatic Hydrolysis of Corn Stover produces excess steam that is converted to electricity for use in the plant ... |
|
Ethanol Production by Enzymatic Hydrolysis: Parametric Analysis of
The plant discussed in the report is a large-scale facility producing price for ethanol and for ethanol produced from biomass via an acid hydrolysis. |
|
Combination of enzymatic hydrolysis and ethanol organosolv
14 févr. 2021 matic hydrolysis and ethanol organosolv pretreatments: effect on lignin ... During autohydrolysis acetic acid is produced from xylans and ... |
|
Ethanol Production by catalytic hydration of ethylene
processes used in the industry to produce ethanol detailing the catalysts used as well as the di if hydrolysis is made by a diluted acid |
|
Bioethanol potential of raw and hydrothermally pretreated banana
calculated as a percent ratio of the actual ethanol produced with respect to the ethanol expected from the total glucose available after acid hydrolysis |
|
Optimization of acid pretreatment and enzymatic hydrolysis on the
sugars which eventually produced ethanol by yeast fermentation. Banana peel |
|
The Effect of Differentiation of Hydrolysis Time towards Ethanol
4 mars 2021 Ethanol Levels Produced Through Ulva Lactuca Fermentation ... The acid hydrolysis stage is required to damage the cellulose structure. |
|
Reaction Rate and Rate Constant of the Hydrolysis of Ethyl Acetate Wit
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) HCl was used as catalyst to accelerate it 1ml and 2ml of ethyl acetate was injected separately into the 500ml reactor vessels which contains 0 05N of NaOH and thoroughly mixed |
|
Determination of rate of reaction and rate constant of the
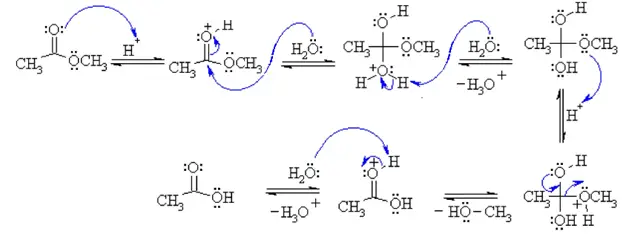
The hydrolysis of an ester such as ethyl acetate illustrates a bimolecular reaction that gives sodium acetate and ethanol as the product from which second order rate constant can be calculated: CH3COOC2H5+NaOH?CH3COONa+C2H5OH present in it direct or indirect titration with a standard solution of an acid |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE - unibask
Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl acetate using sodium hydroxide This experiment illustrates a bimolecular reaction (reacting species are ethyl acetate and sodium hydroxide): CH 3 –COO–CH 2–CH 3 + NaOH ? CH 3–COONa + CH 3–CH 2–OH E |
|
9701/2 W02 Chem - XtremePapers
(c)A mixture of 6 0 g of ethanoic acid and 6 0 g of ethanol was added to 4 4 g of ethylethanoate and the overall mixture allowed to reach equilibrium It was found that0 040 mol of ethanoic acid was present in the equilibrium mixture |
|
Searches related to hydrolysis of ethylethanoate in acid produces filetype:pdf
hydrolysis of ethyl acetate which can be represented by the chemical equation: CH 3COOC 2H 5 + OH ? CH 3COO ? + C 2H 5OH The kinetics data will be obtained • at 25 C under three different initial mixing conditions (Ethyl acetate in excess hydroxide in excess second order conditions – ie 1:1 mixing) • at ~35 C under second order |
What is the hydrolysis of ethyl acetate?
- The hydrolysis of ethyl acetate illustrates a 1 bimolecular reaction that gives sodium acetate and ethanol as the product from which second order rate constant can be calculated:
What is the mechanism of hydrolysis of ethyl ethanoate?
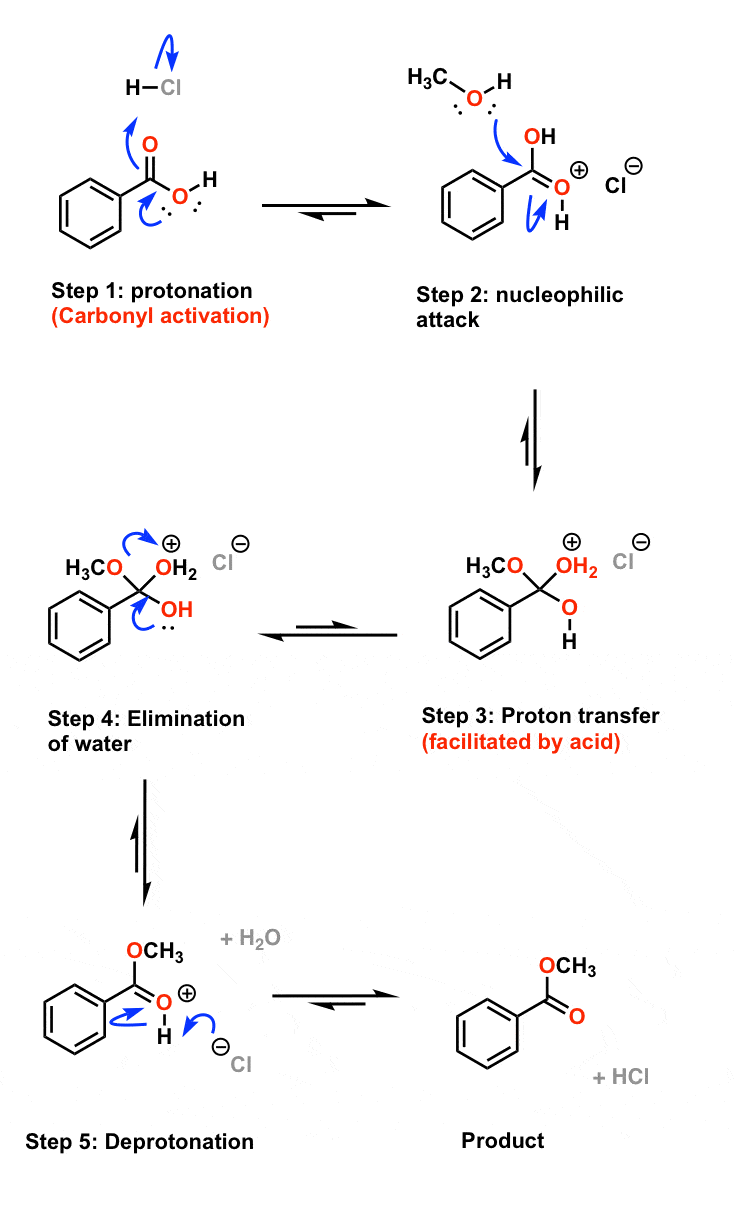
- It uses ethyl ethanoate as a typical ester. The mechanism for the hydrolysis of ethyl ethanoate A reminder of the facts Ethyl ethanoate is heated under reflux with a dilute acid such as dilute hydrochloric acid or dilute sulphuric acid. The ester reacts with the water present to produce ethanoic acid and ethanol.
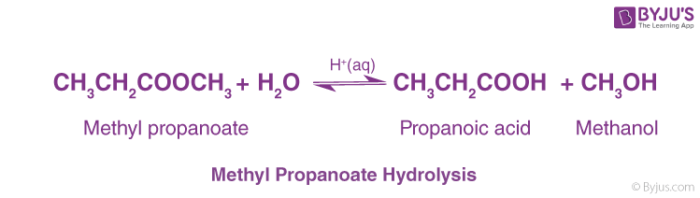
What is ethyl propanoate acidic hydrolysis?
- Ethyl propanoate acidic hydrolysis gives ethanol and propanoic acid. Concentrated H 2 SO 4 is used on heating to get products. Reaction is catalyzed by acids (H 2 SO 4) by making the carbonyl carbon more positive and therefore more susceptible to attack by the nucleophile. Neucleophiles desire to attack positively charged atoms.
How does ethyl acetate react with ethanol?
- OH Ethyl acetate sodium hydroxide sodium acetate ethanol As the reaction proceeds, each hydroxide ion removed in the formation of ethanol removes one molecule of ethyl acetate, being the number of moles of either OH- or ethyl acetate so removed, the concentration of the reactants decreases.
|
Reaction rate and rate constant of the hydrolysis of ethyl - Sci Hub
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide), HCl was used as catalyst to the presence of a mineral acid gives acetic acid and |
|
IV SEMMESTER
To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as gives the end point and this increase in conductivity is due to the fast moving |
|
Chemistry - Pearson
5 jui 2018 · 8 Methanol is produced by the catalysed reaction between carbon monoxide (b ) A 0 0400 mol dm−3 solution of pentanoic acid with pH = 3 13 was used in a ( i) Write the equation for the hydrolysis of ethyl ethanoate using |
|
National Quali cations 2016 X713/76/02 Chemistry Section 1 - SQA
18 mai 2016 · (i) One of the fatty acids produced by the hydrolysis of olive oil is linoleic acid ethanol + ethanoic acid ⇌ ethyl ethanoate + water mass of |
|
Download - Bradford Scholars - University of Bradford
The Acid-Catalyzed Hydrolysis of Carboxylic Esters 278 7 1 3 3 Hence if a change in solvent produces a change in any or all of the "y" terms, then the dioxane on the rate of alkaline hydrolysis of ethylacetate at different temperatures |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
Under neutral conditions, the reaction generally proceeds via addition to the carbonyl carbon to produce a tetrahedral intermediate The slow step is attack by |
|
Kinetics of ethylacetate hydrolysis on mordenite
Liquid phase hydrolysis of ethylacetate on mordenite catalyst has been studied The performance Zeolites are important solid acids used in many commercial processes channel as octahedral AI, which creates diffusion- al hinderance for |
|
Esters - Cathkin High School
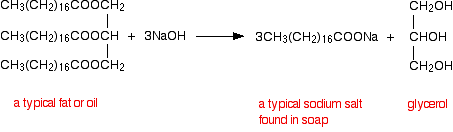
In a hydrolysis reaction a molecule Ethyl ethanoate would have the shortened structural formula The hydrolysis of fats and oils produces fatty acids and |
|
39 Carboxylic acids and esters
Conditions: Ruflux • The hydrolysis of any carboxylic acid derivative results in the carboxylic acid Ethyl ethanoate b Methyl ethanoate c If its melting point is above room temperature it is a solid which makes it a fat, eg margarine and |



















![PDF] Acid-based hydrolysis processes for ethanol from PDF] Acid-based hydrolysis processes for ethanol from](https://data01.123dok.com/thumb/zp/26/4ooy/Pki4ZiPT5VIqTD1so/cover.webp)