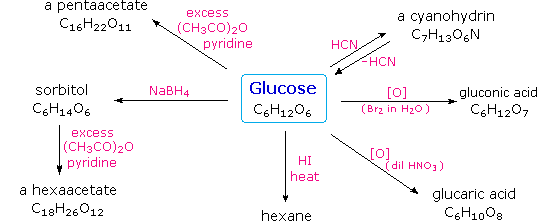
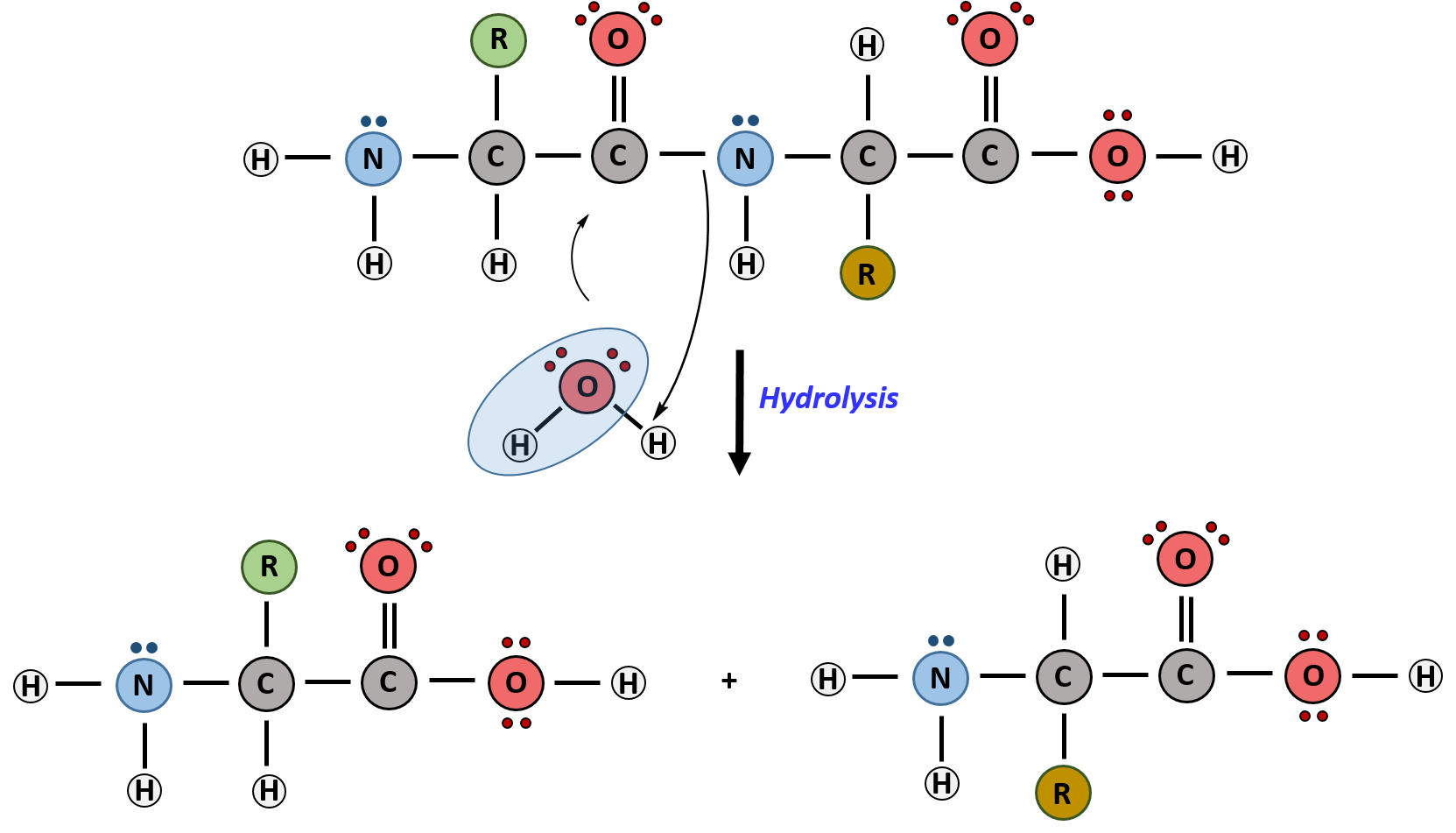
hydrolysis of starch to glucose equation
|
Saccharification of native cassava starch at high dry solids in an
Hydrolysis of starch to glucose may be written Illustration of sequence of reaction occurring during starch hydrolysis in an enzymatic membrane reactor. |
|
REACTION KINETICS FOR STARCH HYDROLYSIS WITH
ining the kinetic relationships and factors important 1n the hydrolysis of soluble starches to glucose using the glucoamylase enzyme. Analyti-. |
|
Mathematical modeling of amylase catalyzed starch hydrolysis
Product inhibition in the reaction of starch hydrolysis was also investigated. Impact of the glucose and maltose on the initial reaction rate was examined. |
|
Kinetic modelling of enzymatic starch hydrolysis
The part of the protein where substrates in this case glucose units of starch |
|
Effect of Pretreatment Methods on Enzymatic Kinetics of
Jul 8 2020 production could be reduced if enzymatic hydrolysis of starch is ... Equation (5) |
|
Process development of starch hydrolysis using mixing
difficulties in designing the starch hydrolysis process into glucose which can be further converted to high ... The reaction rate of starch. |
|
Conversion of Cassava Starch to Produce Glucose and Fructose by
Glucose is a monosaccharide with molecular formula C6H12O6. Glucose is widely used in the food and pharmaceutical industries. Enzymatic hydrolysis. |
|
Digestive Physiology: Amylase hydrolysis of starch
polysaccharide composed of repeating glucose monomers. starch is a good example of a hydrolysis reaction—the insertion of a water molecule to lyse ... |
|
Enzymatic hydrolysis of wheat starch for glucose syrup production
The inhibitory effects were studied by adding 180 g/L of glucose to the process. It was concluded that increases in the E/S ratio decreased reaction times but |
|
Experiment 10 – Enzymes
In the presence of amylase a sample of starch will be hydrolyzed to shorter polysaccharides |
|
Process development of starch hydrolysis using mixing characteristics
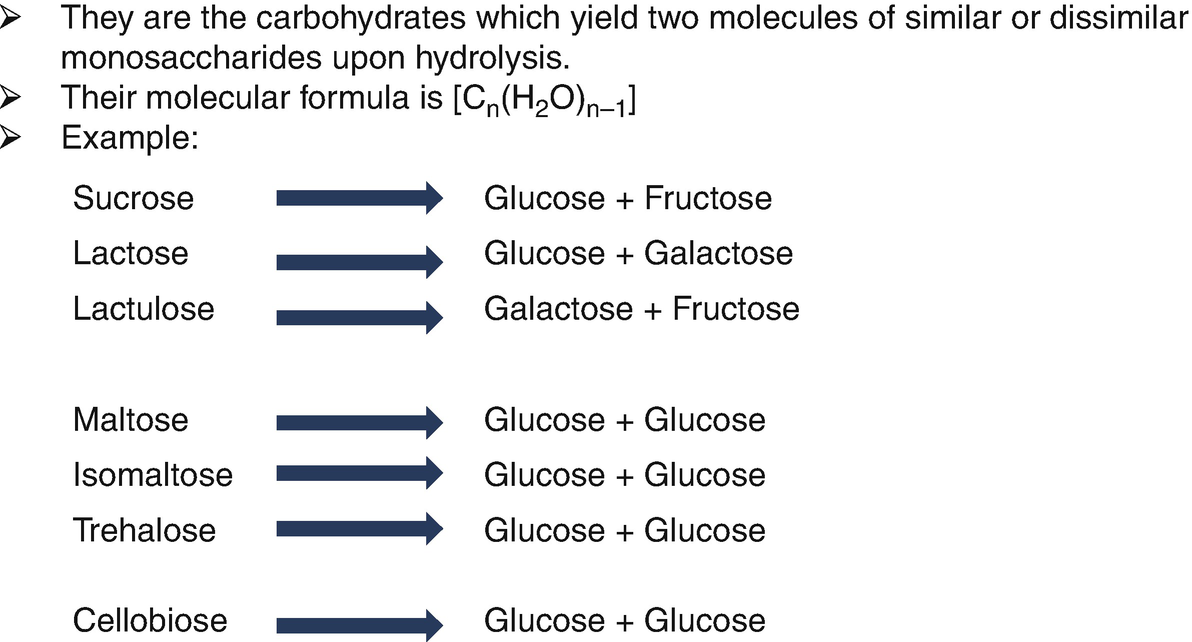
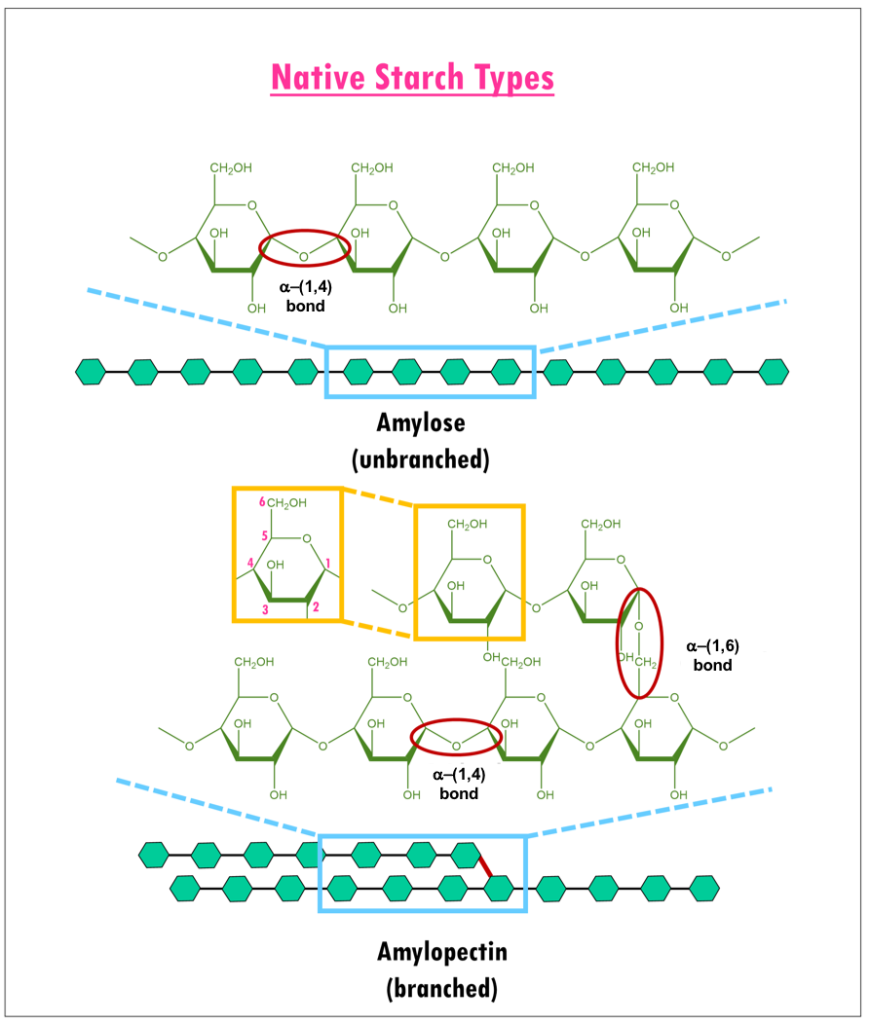
Neither starch or cellulose can be directly fermented by yeast to produce ethanol Must be hydrolyzed to fermentable sugars Degree of Polymerization (DP) 1000 100 oo10 Gl ucose = DP1 These canDextinMaltose = DP2be fermentedby yeast Maltotriose = DP3 Malto-dextrinFour or more glucose molecules = DP4+ OligosaccharideMaltoseGlucose |
|
Sweeteners from 21 Starch: Production Properties and Uses
Acid-catalyzed Hydrolysis In an acid process the slurry containing about 35–45 starch solids is pumped into a pressure vessel called a ‘converter’ and acidi?ed to a pH of about 2 0 with dilute hydrochloric acid at 140–160°C and a pressure of 80 psi (5 4 atm) |
|
Conversion of Cassava Starch to Produce Glucose and Fructose
The conversion of glucose derived from the hydrolysis is depending on the concentration of cassava starch and the reaction parameters such as temperature pressure substrate concentration |
|
Starch Agar Protocol - American Society for Microbiology
linkages between glucose subunits and allow the products of starch hydrolysis to enter the cell Starch agar is also used in differentiating members of various genera which have both amylase-positive and amylase-negative species including Streptococcus Clostridium Corynebacterium Fusobacterium Enterococcus Pseudomonas and Bacillus (8 10) |
|
Searches related to hydrolysis of starch to glucose equation filetype:pdf
An analysis of the enzymatic hydrolysis of wheat starch was performed Gelatinization stage was carried out between 90°C and 95°C for 15min In Liq?tion stage a commercial ?-amylase was used with an enzyme-substrate ratio (E/S ratio) of 0 036 w/w at 60°C and a pH of 5 8 for 4h |
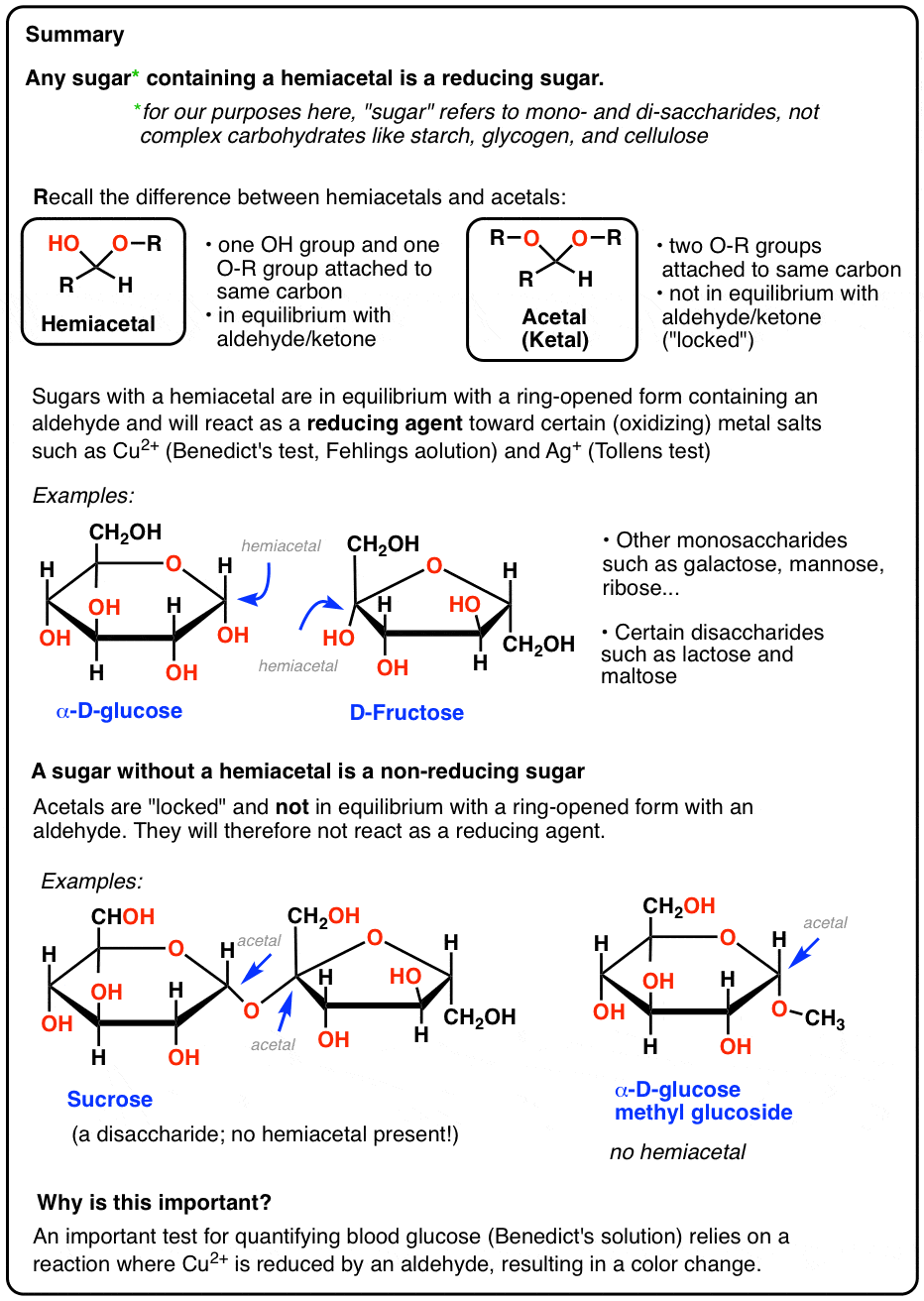
What is starch hydrolysis?
- Starchhydrolysis is an important process to hydrolyze starchinto glucose, which can be further converted to highfructose syrup for various applications in the foodindustry. Starch hydrolysis consists of two steps: gela-tinization and sacchari?cation.
What is the substrate concentration for enzymatic starch hydrolysis?
- Currently, enzymatic starch hydrolysis is carried out at a substrate concentration of 35-40 w/w % (Schenck, 2002). Increasing the substrate concentration has several advantages; such as increased volumetric productivity and a higher ?-amylase stability (Klibanov, 1983; Krishnan and Chandra, 1983; De Cordt et al., 1994).
Which enzyme catalyzes the hydrolysis of starch into maltose?
- Amylase catalyses the hydrolysis of starch (polysaccharide form of glucose) into maltose (a disaccharide of glucose). Was this answer helpful?
How is starch measured?
- Most commonly used methods for the measurement of starch in food, feeds, and ingredients employ the combined action of?-amylase and amyloglucosi- dase to hydrolyze the starch to glucose, followed by glucose determination with a glucose oxidase/peroxidase reagent. Recently, a number of questions have
|
Conversion of Cassava Starch to Produce Glucose and Fructose by
Glucose is a monosaccharide with molecular formula C6H12O6 Glucose is widely used in the food and pharmaceutical industries Enzymatic hydrolysis |
|
REACTION KINETICS FOR STARCH HYDROLYSIS - ShareOK
9 déc 2015 · ining the kinetic relationships and factors important 1n the hydrolysis of soluble starches to glucose using the glucoamylase enzyme Analyti- |
|
Production of Glucose from Hydrolysis of Potato Starch
2 mai 2020 · The SSA was explored as a solid acid for the potato starch hydrolysis The SSA was converted into polymeric frame work using Bakelite reaction |
|
Conversion of Starch into Sugars Carbohydrates and - IEA USP
carbon, hydrogen and oxygen, general formula Cx(H20)y • Carbohydrates are Saccharides, the simplest form of carbohydrates, consist of single sugar units with five or six As starch is hydrolyzed, more and more dextrose is d d d th t f d i |
|
Mathematical modeling of amylase catalyzed starch hydrolysis
reaction of starch hydrolysis was also investigated Impact of the glucose and maltose on the initial reaction rate was examined The results suggested that both |
|
Enzymatic hydrolysis of wheat starch for glucose - SciELO Colombia
The inhibitory effects were studied by adding 180 g/L of glucose to the process It was concluded that increases in the E/S ratio decreased reaction times but |




![PDF) Hydrolysis of starches by the action of an [alpha]-amylase PDF) Hydrolysis of starches by the action of an [alpha]-amylase](https://microbeonline.com/ezoimgfmt/i1.wp.com/microbeonline.com/wp-content/uploads/2020/02/Starch-Molecule-structure.png?resize\u003d678%2C404\u0026ssl\u003d1\u0026ezimgfmt\u003drs:372x222/rscb2/ng:webp/ngcb2)

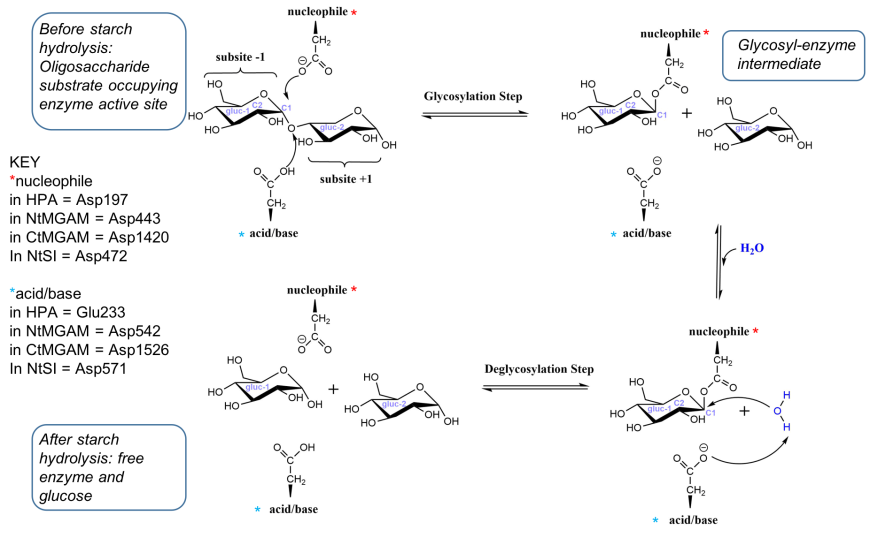






















![PDF] An original kinetic model for the enzymatic hydrolysis of PDF] An original kinetic model for the enzymatic hydrolysis of](https://pubs.rsc.org/image/chapter/bk9781782621133/bk9781782621133-00001/bk9781782621133-00001-f6_hi-res.gif)