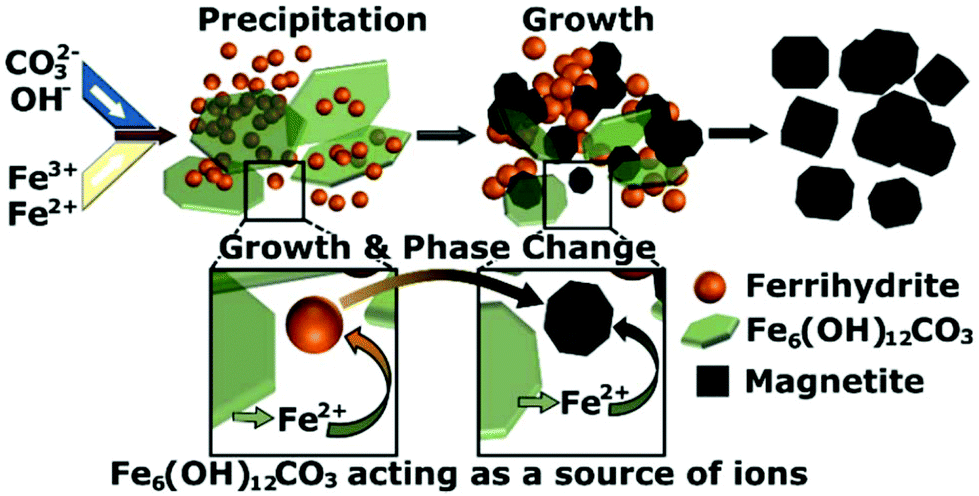
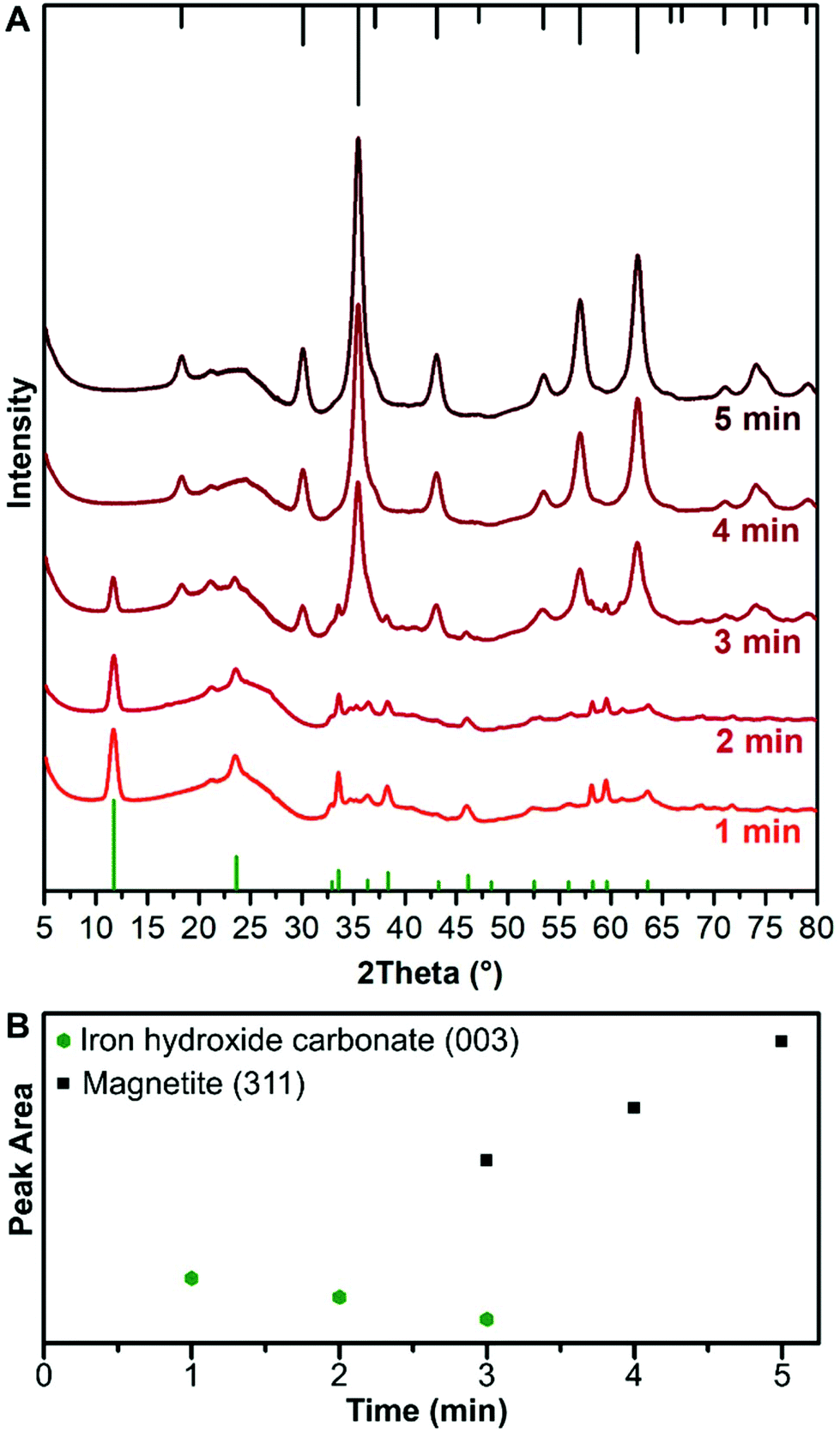
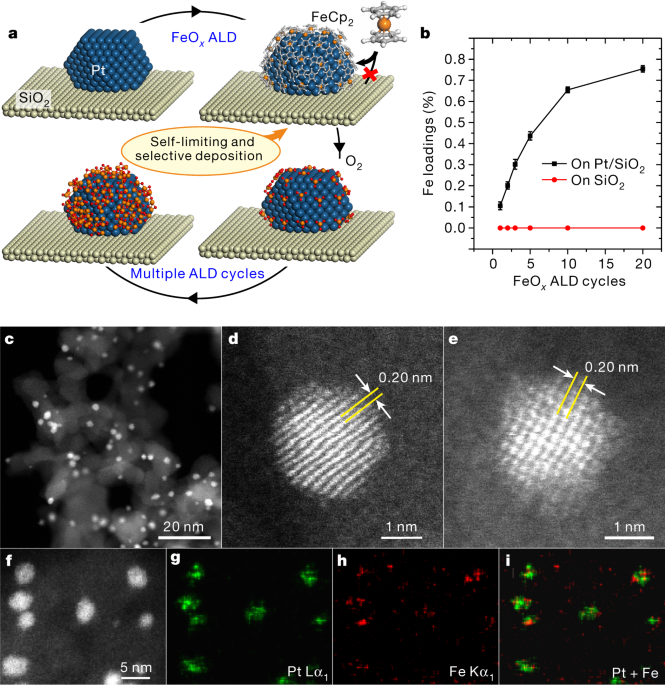
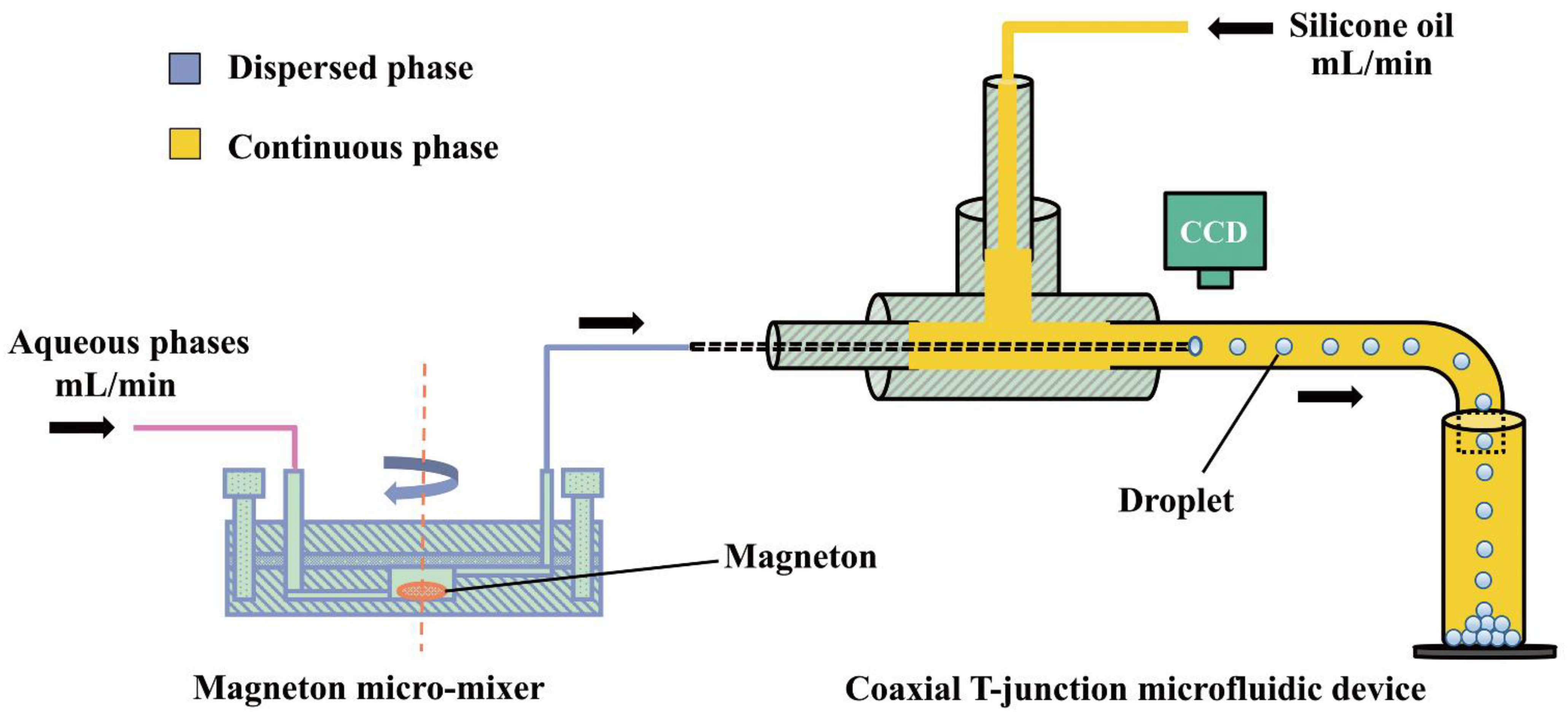
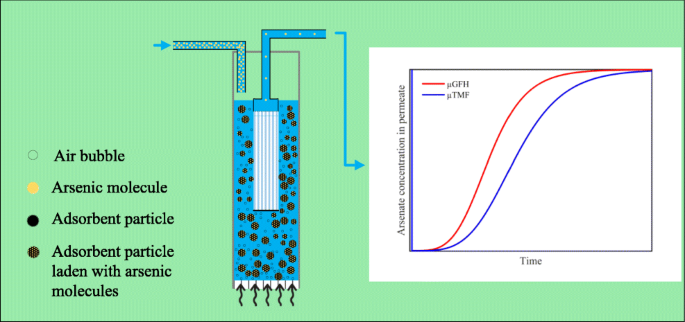
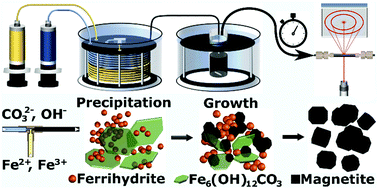
in sol of ferric hydroxide the dispersed phase is
|
Unit-1 Setting.pmd
Further a heterogeneous system of a solid as dispersed phase and a liquid as dispersion medium is called a sol. Why is ferric hydroxide/aluminium hydroxide ... |
|
THE MECHANISM OF THE MUTUAL PRECIPITATION OF CERTAIN
The so-called “meta-iron” sol of Péan de. St. Gilíes is one in which the particles of the dispersed phase are less hy- drated than in the Graham ferric oxide |
|
Sol-gel synthesis of iron(III) oxyhydroxide nanostructured monoliths
17 thg 11 2012 from the presence of the NH4NO3 crystals dispersed on the iron phase. ... Portugal |
|
اﻟﺟﺎﻣﻌﺔ اﻟﺗﮐﻧوﻟوﺟﯾﺔ ﻗﺳم اﻟﮭﻧدﺳﺔ اﻟﮐﯾﻣﯾﺎوﯾﺔ اﻟﺛﺎﻧﯾ
The examples of lyophobic sols are dispersion of gold iron (III) hydroxide and sulphur in water. The affinity or attraction of the sol particles for the medium |
|
B. Sc. II-Sem Colloidal state (1) The foundation of colloidal chemistry
(v) Dialysis can be used for removing HCl from the ferric hydroxide sol. (2) “Emulsion are the colloidal solutions in which both the dispersed phase and the ... |
|
Colloids Various phases of colloidal solution
They consist of two phases; the dispersed phase and the dispersion medium. (v) Dialysis can be used for removing from the ferric hydroxide sol. Page 5 ... |
|
Physicochemical fundamentals of colloidal systems. The colloid
dispersed phase and dispersion medium. by adding boiling water dropwise to a solution of ferric chloride one obtains the dark brown sol of ferric hydroxide. |
|
Colloids: Thomas Graham (1861) studied the ability of dissolved
Warm solutions of the dispersed phase on cooling set to a gel e.g. Example: A ferric hydroxide sol (red) made by the hydrolysis of ferric chloride is. |
|
8 COLLOIDS
Emulsions are colloidal solutions in which both the dispersed phase and dispersion medium are liquids. (Arsenious sulphide sol ferric hydroxide sol). 3. (a) ... |
|
Colloids
In order to obtain a red sol of ferric hydroxide a few drops of 30% ferric chloride solution is added to a large volume of almost boiling water and stirred |
|
Unit-1 Setting.pmd
dispersed phase and a liquid as dispersion medium is called a sol. Depending Freshly prepared ferric hydroxide aluminium hydroxide and. |
|
THE MECHANISM OF THE MUTUAL PRECIPITATION OF CERTAIN
The so-called “meta-iron” sol of Péan de. St. Gilíes is one in which the particles of the dispersed phase are less hy- drated than in the Graham ferric |
|
Sol-gel synthesis of iron(III) oxyhydroxide nanostructured monoliths
17 nov. 2012 phases and structures in the case of iron oxides/hydroxides. ... ammonium hydroxide that is often applied in sol-gel chemistry to obtain ... |
|
8 COLLOIDS
lyophilic colloidal solution has been formed then dispersed phase and of ferric hydroxide ferric hydroxide gets converted into reddish brown coloured. |
|
Colloidal Solution B.Sc-I Paper-I By Dr. Anindra Sharma Department
adsorption of positive or negative ions from the dispersion medium. For example a ferric hydroxide sol particles are positively charged because these |
|
Obtaining purification and properties of colloidal solutions. 1
Prepare colloidal solutions. Determine the charge sign of the disperse phase particles. Obtaining of a ferric hydroxide sol by the method of hydrolysis. |
|
Marine Geochemistry 1. Chemical Scavengers of the Sea
instance if ferric hydroxide is formed and precipitated in a solution of a ura- nium salt |
|
??????? ??????????? ??? ??????? ?????????? ??????
As stated above a colloidal system is made of a dispersed phase and the dispersion For example |
|
The Colloidal State Introduction: A colloid is one of the three primary
Ferric hydroxide sol is positively charged because the sol particles adsorb the ferric ions in preference to the chloride ions. Stability of colloids: Colloidal |
|
A STUDY OF THIXOTROPIC -FeOOH BY MÖSSBAUER EFFECT
1 ian. 1976 superparamagnetique en phase sol et gel. ... Les constantes hyperfmes de B-FeOOH en phase gel et sol gel& ont 6tB ... Iron hydroxide P-FeOOH. |
|
Colloid-Part II
hydroxide (the stabilizing agent) is taken in porcelain or glass (non conducting) vessel Metal to be dispersed is dipped in the vessel in the form of electrodes Electrodes are connected to the high voltage source The ends of electrodes in the dispersion medium are very near to each other |
|
[Explained] Types of Colloids and their Examples
A common method of classifying colloids is based on the phase of the dispersed substance and what phase it is dispersed in The types of colloids include sol emulsion foam and aerosol Sol is a colloidal suspension with solid particles in a liquid Emulsion is between two liquids |
|
Structural identification solubility of ferric hydroxide
solubility of ferric hydroxide precipitates and molecular structural identification Qiantao Shia George E Sterbinskyb Shujuan Zhanga Christos Christodoulatosa George P Korfiatisa and Xiaoguang Menga* aCenter for Environmental Systems Stevens Institute of Technology Hoboken New Jersey 07030 United States |
|
8 COLLOIDS - The National Institute of Open Schooling (NIOS)
phases : the dispersed phase and the dispersion medium z Dispersed Phase : It is the substance present in small proportion and consists of particles of colloids size (1 to 100 nm) Name of Property Size Filterability Settling Visibility Separation Diffusion True Solution Size of particles is less than 1 nm Pass through ordinary filter paper |
|
Searches related to in sol of ferric hydroxide the dispersed phase is filetype:pdf
Since particles of dispersed phase in lyophilic sols have an affinity for the particlesof dispersion medium these sols are more stable as compared to lyophobic sols Two factors responsible for the stability of sols are – charge and the solvation ofthe colloidal particles by the solvent |
What is the dispersed phase of ferric hydroxide?
- For example, in a colloidal solution of ferric hydroxide in water the ferric hydroxide particles are of colloidal size and constitute the dispersed phase. The medium in which the colloidal particles are dispersed is called dispersion medium.
How to prepare ferric hydroxide Sol?
- The aim of this experiment – to prepare ferric hydroxide sol. Ferric hydroxide (Fe (OH) 3) has the ability to form a lyophobic sol. Sulphides or metal hydroxides which are colloidal solution, when treated with water, are termed as lyophobic colloids. Fe (OH) 3 sol is obtained by hydrolysis of ferric chloride with boiling distilled water.
How does ferric hydroxide form a lyophobic Sol?
- THEORY: Ferric hydroxide forms a lyophobic sol with water which is the dispersion medium. It is prepared by the hydrolysis of ferric chloride with boiling distilled water as per the reaction: FeCI 3 (aq) + 3H 2 O ? Fe (OH) 3 + 3HCI (aq).
What is the solubility of ferric hydroxide in water?
- Thus due to the common ion effect, the dissociation of F e ( O H) X 3 should be very less. So, the solubility of Ferric hydroxide in water should be extremely less. By mathematical treatment also, this is justifiable. Consider the solubility of Ferric hydroxide in water to be S m o l. L ? 1. and consider the concentration of H X 3 O X + is x m o l.
|
Unit-1 Settingpmd - NCERT
dispersed phase and a liquid as dispersion medium is called a sol Depending upon the nature of Freshly prepared ferric hydroxide, aluminium hydroxide and |
|
B Sc II-Sem Colloidal state (1) The foundation of colloidal chemistry
(i) Classification based on the physical state of the dispersed phase and dispersion (v) Dialysis can be used for removing HCl from the ferric hydroxide sol |
|
Physical Chemistry
evaporating the sol, the dispersed phase obtained can be easily reconverted into the solution by Dialysis can be used to remove HCl from ferric hydroxide sol |
|
Surface Chemistry Viva Questions With Answers - SelfStudys
In sol liquid is the dispersion medium and solid the disperse phase whereas in gel solid is the dispersion medium and liquid the disperse phase |
|
Book-sample-page-f85ba7d595b6ad59635206baad72839epdf
Lyophobic sol-aluminium hydroxide, ferric hydroxide, arsenious sulphide Starch molecule (dispersed phase) + Water (dispersion medium) = Colloidal |
|
8 COLLOIDS - NIOS
phases : the dispersed phase and the dispersion medium ○ Dispersed Two beakers A and B contain ferric hydroxide sol and NaCl solution respectively |
|
SURFACE CHEMISTRY Section - chemistryworkshopjr
Colloid is heterogeneous system in which dispersed phase particles with 1nm to 1000 Eg Ferric hydroxide sol, aluminium hydroxide sol, arsenic sulphide sol, |
|
Details of Module and its structure - NROER
dispersed phase, dispersion medium, sol, gel, emulsion, to freshly prepared ferric hydroxide (Fe(OH)3), ferric hydroxide particles adsorb Fe3+ ions |
|
Physicochemical fundamentals of colloidal systems The colloid
dispersed phase (for the phase forming the particles) and dispersion medium or chloride one obtains the dark brown sol of ferric hydroxide A common feature |
|
UNIT -5 SURFACE CHEMISTRY - PUE
Sol 9 Name the dispersed phase in gel Liquid 10 Give an example for oil in Write equations for the preparation of Sulphur sol and Ferric hydroxide sol |