inductive effect chemistry a level
|
Detailed Notes - Topic 3.11. Amines - AQA Chemistry A-level
03-Mar-2011 Positive inductive effect. This means aliphatic amines are strongerbases and aromatic amines are weaker. https://bit.ly/pmt ... |
|
Tuning Redox Transitions via Inductive Effect in Metal Oxides and
molecular chemistry electrochemistry |
|
Methyl group inductive effect in the toluene ions. Comparison of
(20) The value of hi has negligible effect on the energy level ordering. Journal of the American Chemical Society / 90:23 / November 6 1968. Page 3 |
|
Organic Chemistry Himansu Pandey-min.pdf
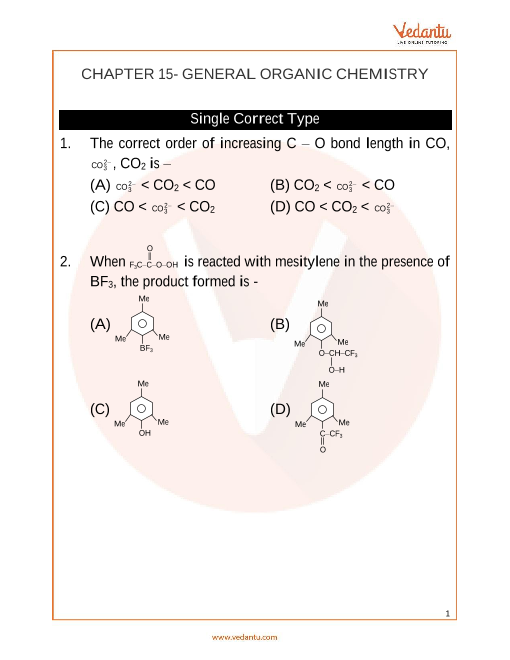
General Organic Chemistry. (EXERCISE ONLY ONE CORRECT ANSWER. LEVEL. 1. Which of (b) Inductive effect. (d) Electromeric effect. (b) Aldehydes. 43. Which of ... |
|
Cambridge International AS & A Level
CHEMISTRY. 9701/41. Paper 4 A Level Structured Questions M4: butanoic acid: positive inductive effect / electron donating effect of propyl / alkyl / R group. |
|
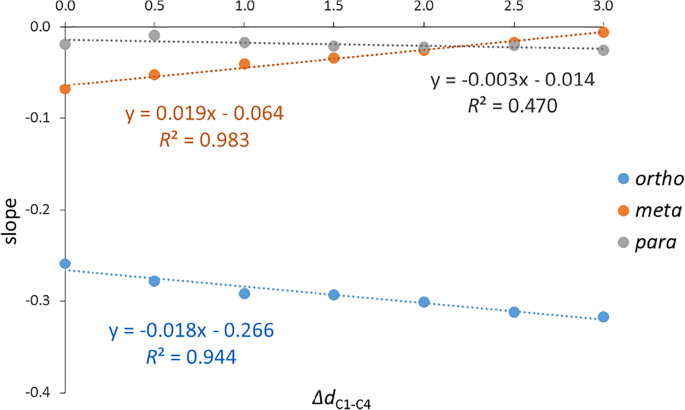
Phase Separating Electrode Materials Chemical Inductors
16-Jun-2022 Inductive effects in low frequency part of spectrum are seen for lithiation levels within the miscibility gap (i.e. lithiation levels from 0.2. |
|
Phase separating electrode materials - chemical inductors?
16-Jan-2023 Inductive effects in the low frequency part of the spectrum are seen for lithiation levels within the miscibility gap (i.e. lithiation levels ... |
|
Level-1 Test: Syllabus for Computer-based Multiple Choice (MCQ
inductive effects in alkyl halides;. Reactive intermediates (carbocations ... Chemistry of s-and p-block elements. Alkali and alkaline earth metals –electronic ... |
|
Inductive effect of uncharged groups: dependence on electronegativity
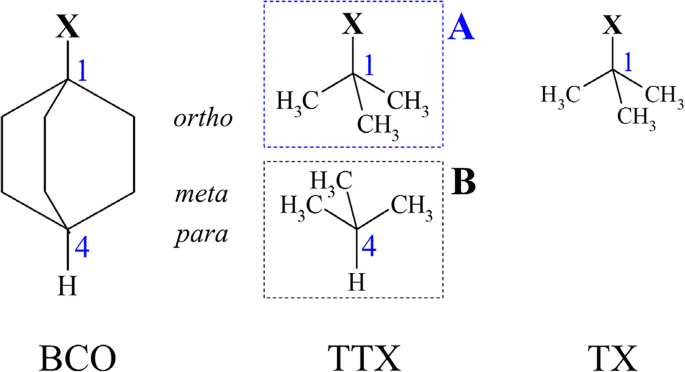
quantum chemical methods at various levels;1517–20 derivatives of bicyclo[2.2.2]octane played an important role.15 |
|
Subject: Chemistry Semester VI Paper No.: CHB-605 Topic: IR
Fermi resonance requires that the vibrational levels be The electronic effect including inductive effect mesomeric effect |
|
Detailed Notes - Topic 3.11. Amines - AQA Chemistry A-level
03-Mar-2011 Positive inductive effect. This means ?aliphatic amines are stronger?bases and ?aromatic amines are weaker?. https://bit.ly/pmt ... |
|
Positive inductive effect of methyl groups in nine simple alcohols
As a result of this chemical surrounding the electron density between the bonded atoms may decrease or decrease modifying the reactivity of a given bond. The |
|
Chemistry Questions and Answers – Inductive Effect 1
Chemistry Questions and Answers – Inductive Effect. 1. Electronegativity is defined as the power of an atom in a molecule to ______. |
|
Inductive effect of uncharged groups: dependence on electronegativity
KEYWORDS: inductive effect; electronegativity; substituent effect; density functional theory; quantum chemical methods at various levels;1517–20. |
|
Organic Chemistry Himansu Pandey-min.pdf
GENERAL ORGANIC CHEMISTRY. § Exercise-1: Only One Correct Answer. Level-1. Level-2 Which of the following exhibit electromeric effect ?. (a) Alkanes. |
|
The Inductive Effect in Organic Chemistry
The “inductive effect” remains an icon of chemical education and chemical thought. It is and also the MK model) at the RHF/6-31G level do indicate. |
|
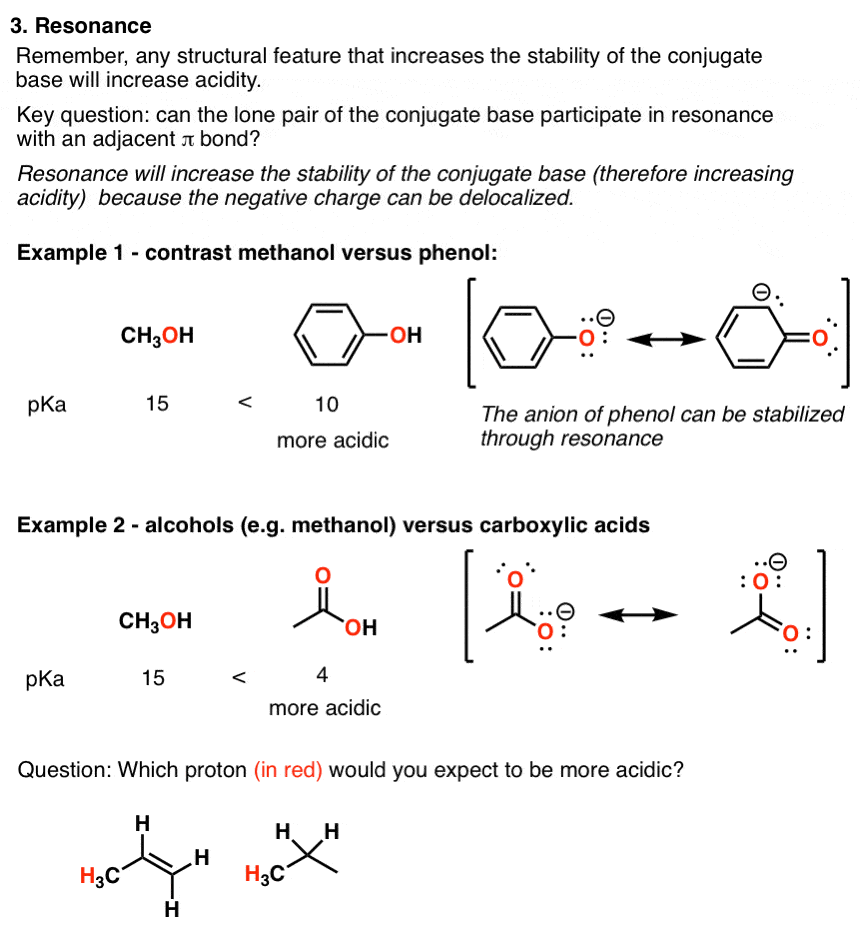
CARBOXYLIC ACIDS
Substituents mainly exert their influence on the acidity of aliphatic carboxylic acid through the inductive effect. Since the inductive effect operates through |
|
Phase Separating Electrode Materials Chemical Inductors
16-Jun-2022 We discover presence of chemical inductive effects in phase ... level experimental observation of inductance during impedance measurements. |
|
Inductive effect
11-Mar-2021 In chemistry the inductive effect is an effect regarding the transmission of unequal sharing of the bonding electron through a chain of ... |
|
Phase Separating Electrode Materials – Chemical Inductors?
We discover presence of chemical inductive effects in phase separating ion level experimental observation of inductance during impedance measurements. |
|
RESONANCE AND INDUCTION TUTORIAL Jack DeRuiter
Induction or the inductive effect of an atom or functional group is a function of that groups 1) electronegativity 2) bonding order and charge and 3) position within a structure Inductive effects refer to those electronic effects of an atom or functional group can contribute through single bonds such as saturated (sp 3) carbon atoms! This is |
|
Write short note on inductive effect and resonance - Brainlyin
M3 reason for increased stability of intermediate in terms of greater number of alkyl groups showing largest inductive (electron releasing) effect greater (positive) inductive effect due to two alkyl groups OR greater electron donation of two alkyl groups 1 4(c) dehydrating agent / cause dehydration 1 4(d)(i) oxidation 1 4(d)(ii) |
|
Introduction to Organic Chemistry
Inductive effect - shifting of electrons in response to the EN of nearby atoms Lewis Structures: represent what covalent bonds are present in a molecule electron bookkeeping |
|
A-level CHEMISTRY (7405/3) - Exam QA
Stage 2: the inductive effect • The unionised COOH group contains two very electronegative oxygen atoms • therefore has a negative inductive (electron withdrawing) effect • The CH3 group has a positive inductive (electron pushing) effect Stage 3: how the polarity of OH affects acid strength • The O–H bond in the ethanedioic acid is |
|
14: Substituent Effects - UC Santa Barbara
The inductiveeffect of F on the acidity of the CO2H group is a result of the positively polarizedCH2 carbon to which the CO2H group is attached How C-F Polarity Affects Acidity Fluoroacetic acid is an acid because itdonates a proton to water or other bases (Figure 14 07) |
|
Searches related to inductive effect chemistry a level filetype:pdf
Cambridge International AS & A Level CHEMISTRY 9701/22 Paper 2 AS Level Structured Questions February/March 2022 MARK SCHEME Maximum Mark: 60 Published This mark scheme is published as an aid to teachers and candidates to indicate the requirements of the examination It shows the basis on which Examiners were instructed to award marks |
|
The Inductive Effect in Organic Chemistry - UNL Digital Commons
The present paper is concerned with the ability of alkyl groups to serve as sigma inductive electron donors The “inductive effect” remains an icon of chemical |
|
Chemistry Questions and Answers – Inductive Effect
Which of the following is an application of inductive effect? a) Bond length b) Dipole moment c) Strength of carboxylic acids d) All of the mentioned |
|
Markovnikovs Rule (328) CIE A Level Chemistry Revision Notes
Avis 48 |
|
Resonance and induction tutorial
Induction or the inductive effect of an atom or functional group is a function of that groups 1) electronegativity 2) bonding order and charge and 3) |
|
Resonance and Inductive Effects
Introduction to Chemical Reactivity of Organic (v) Inductive effects are caused by differences in electronegativity between bonded atoms which leads |
|
Types of Inductive Effect Applications Stability - Byjus
The inductive effect refers to the phenomenon wherein a permanent dipole arises in a given molecule due to the unequal sharing of the bonding electrons in |
|
Detailed Notes - Topic 311 Amines - AQA Chemistry A-level
3 mar 2011 · More alkyl groups means more 'pushing' Example: Positive inductive effect This means aliphatic amines are strongerbases and aromatic amines |
|
Block – 2: Organic reaction mechanism - UOU
inductive effect mesomeric (or resonance) effect electromeric effect and hyperconjugative effect Of these electromeric effect is temporary while rest is |
|
BSc Chemistry - e-PG Pathshala
There are four types of electronic effects mainly viz inductive effect mesomeric (or resonance) effect electromeric effect and hyperconjugative effect Of |
|
Química - Elsevier
Abstract Electronic substituent effects are usually classified as inductive (through ?-bonds) and resonance effects (via ?-bonds) The alkyl group has been |
What is the inductive effect in chemistry?
- In chemistry, the inductive effect is an experimentally observed effect of the transmission of unequal sharing of the bonding electron through a chain of atoms in a molecule, leading to a permanent dipole in a bond. In short, alkyl groups tend to donate electrons, leading to the inductive effect. I Hope It Will Help!
What is the inductive effect of a ? bond?
- The polarization of a ? bond due to electron-withdrawing or electron-donating effect of adjacent groups or atoms is called inductive effect. The inductive effect arises due to the electronegativity difference between two atoms forming a sigma bond. Similarly, a carbocation will be destabilized by an electron-donating group.
What is the inductive effect of a carbocation?
- The inductive effect arises due to the electronegativity difference between two atoms forming a sigma bond. Similarly, a carbocation will be destabilized by an electron-donating group.
What is the difference between inductive effect and resonance effect?
- This is termed as inductive effect. The polarity produced in a molecule by interaction of two ?-bonds or between a ?-bond and lone pair of electrons present on an adjacent atom is termed as resonance effect. Still have questions?
|
Research Article Topological Model on the Inductive Effect - CORE
19 fév 2014 · of Pure and Applied Chemistry (IUPAC) the inductive effect is defined as the bond distances at the experimental level We propose to |
|
PDF :8 - BSc Chemistry
TABLE OF CONTENT 1 Learning outcomes 2 Introduction 3 Types of Electronic effects 3 1 Inductive Effect 3 2 Mesomeric or resonance effect 4 Summary |
|
Inductive Effects
Induction or the inductive effect of an atom or functional group is a function of that groups 1) electronegativity derived from amide chemistry Typically amides |
|
Chapter 1: Introduction - University of Arizona
these cognitive resources will improve chemistry curriculum at all levels positive inductive effect decreases acid strength because electron density is |
|
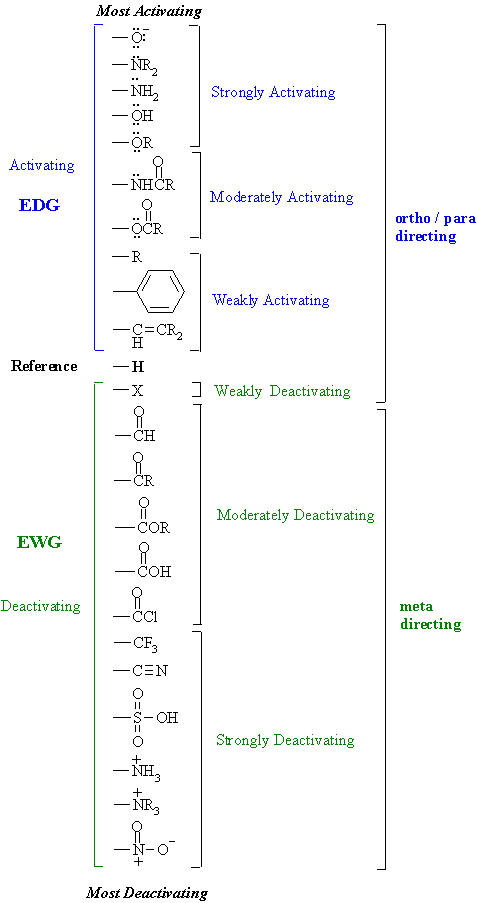
Substituent Effects
Alternatively, we can use substituent effects to understand chemical We will divide the electronic influence of substituents into inductive effects and resonance arbitrarily put the absolute energy levels of F-CH2CO2H and H- CH2CO2H at |
|
Grids to Illustrate Induction and Resonance Effects - Amazon S3
18 fév 2020 · Resonance Effects: Electrophilic Aromatic Substitution Advanced undergraduate, organic chemistry for chemistry majors, (basic knowledge of EAS), structure (a 3x3 grid, games last 15-30 min), pedagogy (varied levels of |
|
Detailed Notes - Topic 311 Amines - AQA Chemistry A-level
3 mar 2011 · Positive inductive effect This means aliphatic amines are strongerbases and aromatic amines are weaker https://bit ly/pmt |