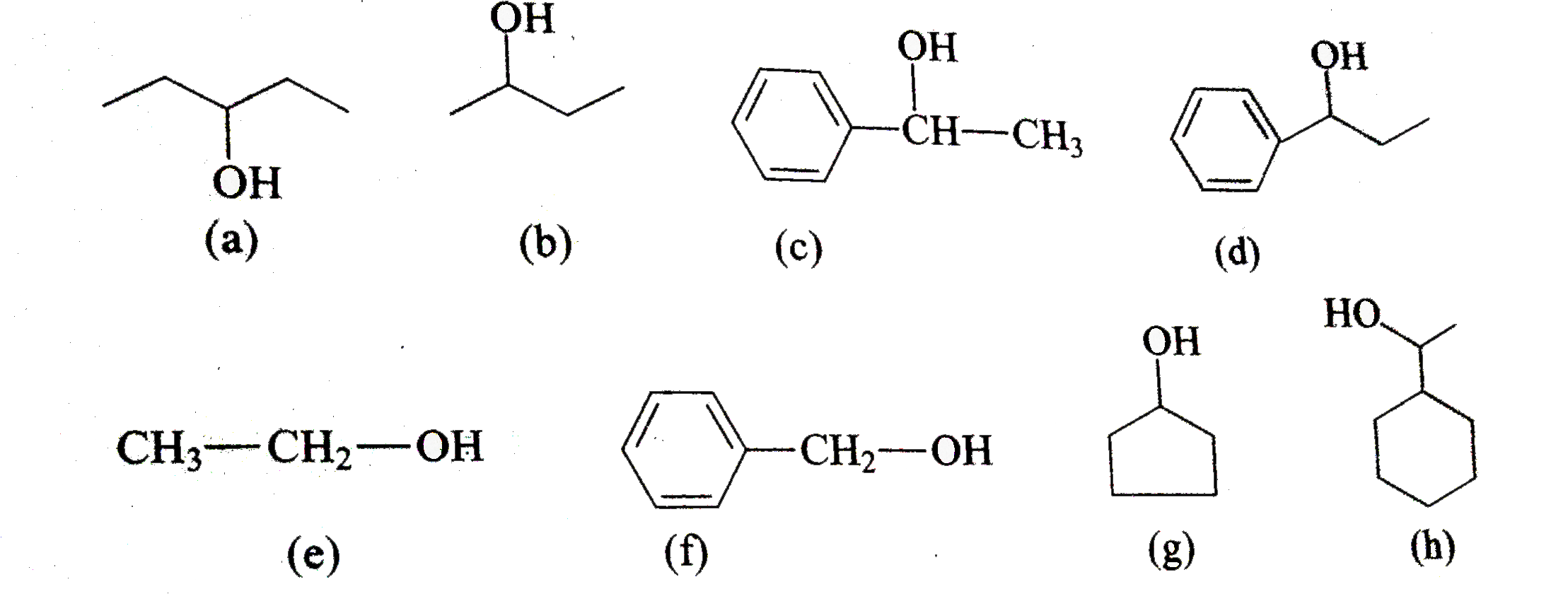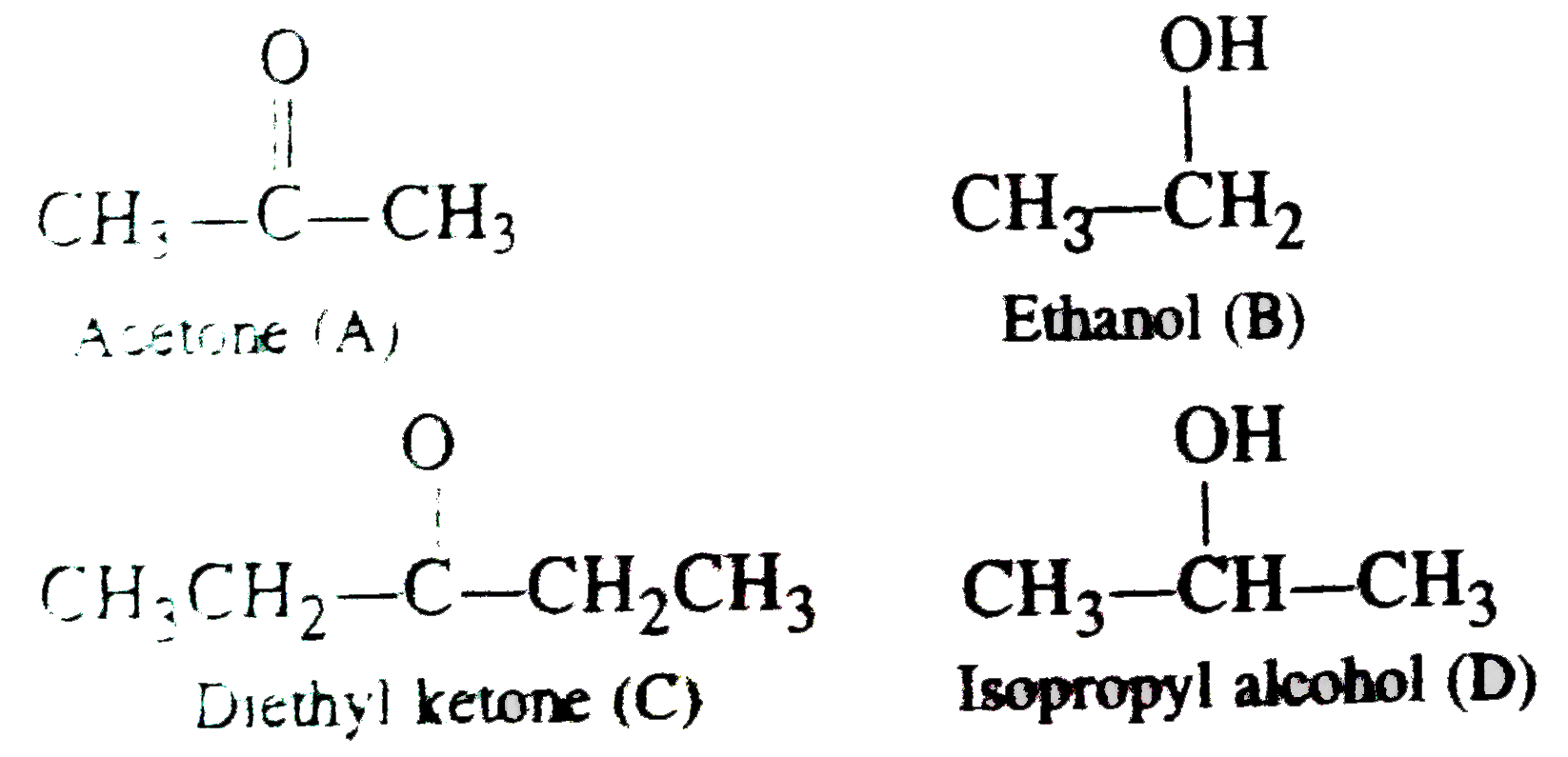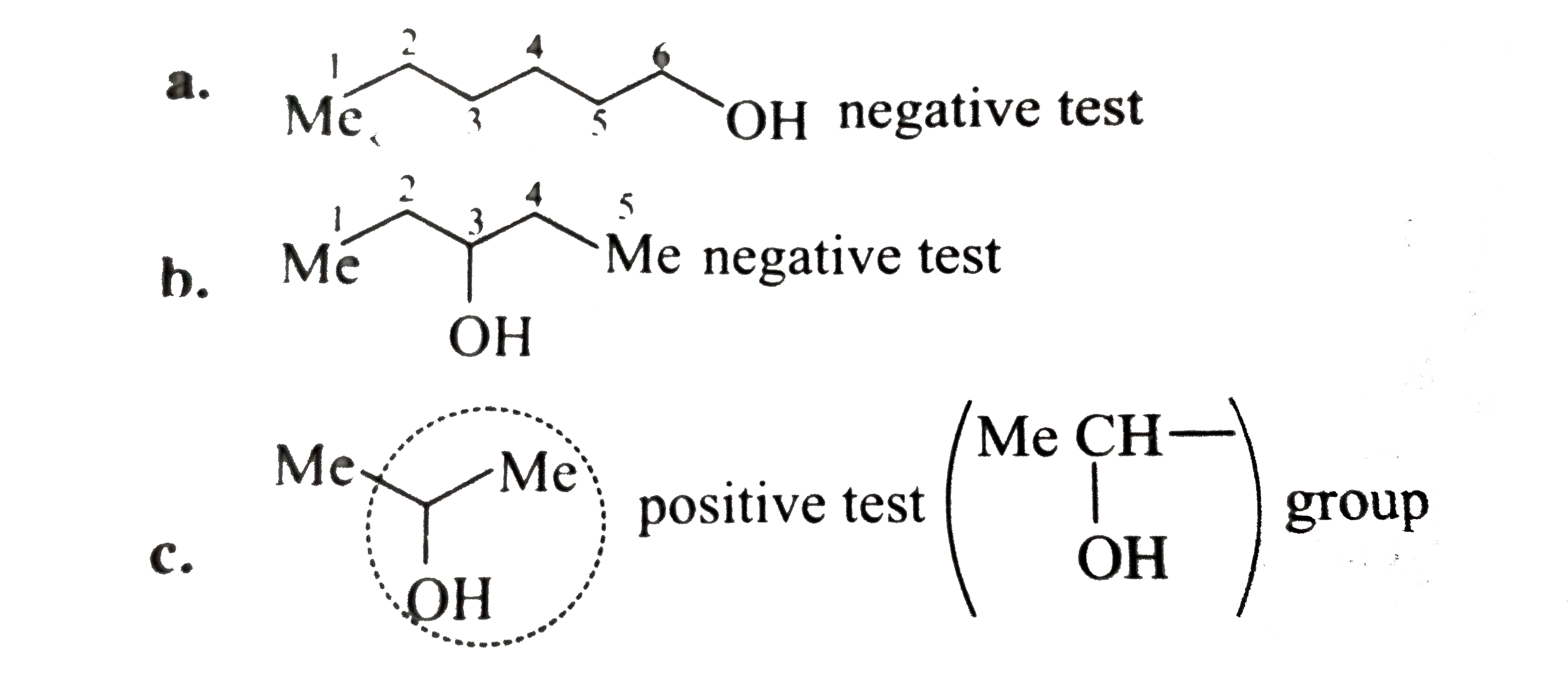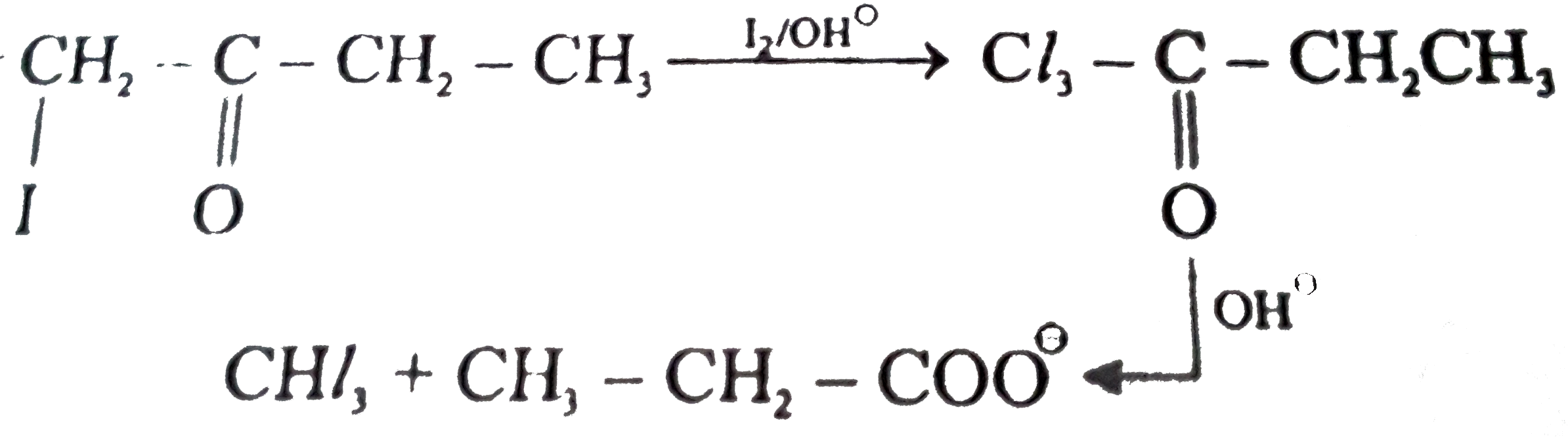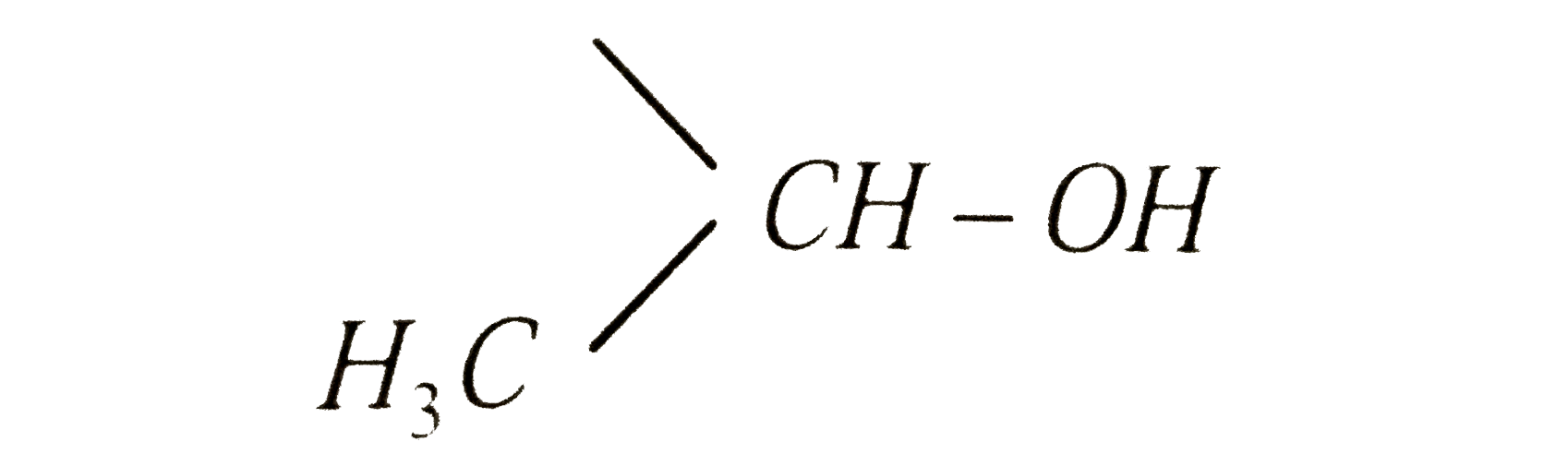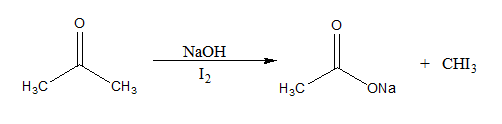iodoform test for alcohols
|
Experiment 6 – Alcohols and Phenols
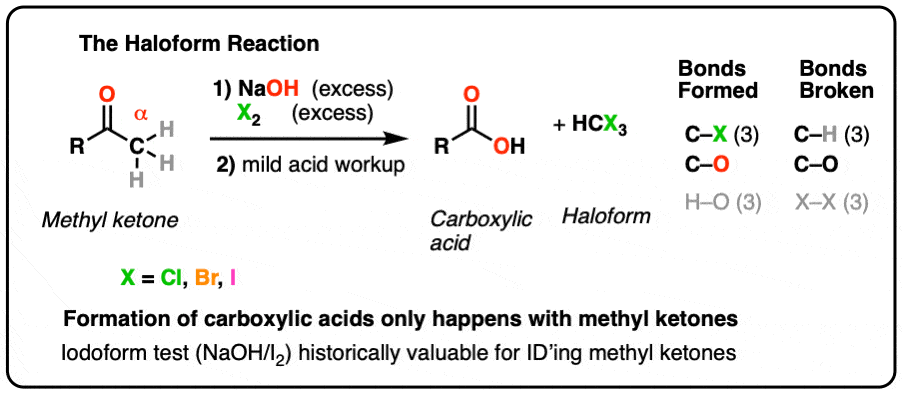
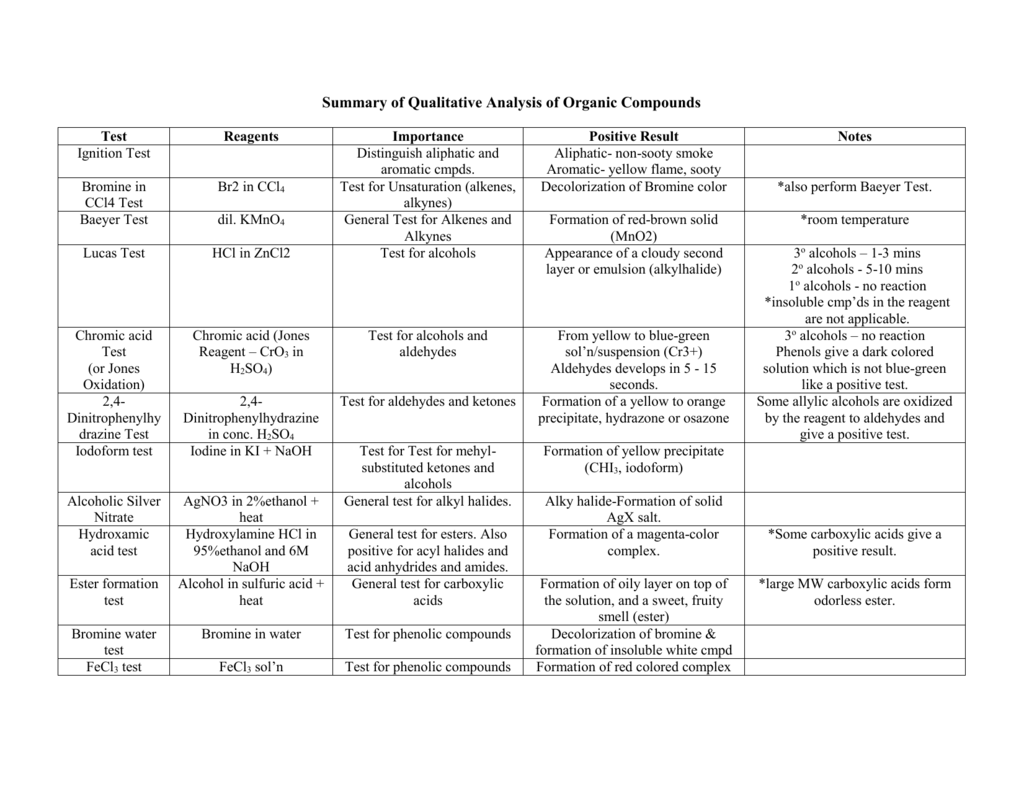
Iodoform Test. The iodoform test is used to identify secondary alcohols that have a methyl group on the alcohol carbon. This type of alcohol will react with |
|
Experiment 6 Qualitative Tests for Alcohols Alcohol Unknown
http://myweb.liu.edu/~swatson/downloads/files/Experiment_6.pdf |
|
Identification of an Unknown – Alcohols Aldehydes
https://people.chem.umass.edu/mcdaniel/chem269/experiments/aak/procedure.pdf |
|
Testsforfunctionalgroups - inorganiccompounds
Ethanol and secondary alcohols which contain CH3—CH(OH)R group (iodoform reaction) give positive iodoform test. To carry out reaction potassium iodide and |
|
The Haloform Reaction. XIV. An Improved Iodoform Test
iodoform is obtained. (4) In the case of methyl ketones heating is usually not necessary; alcohols react more. |
|
Bioethanol Production from Empty Fruit Bunch using Direct
2.5 Bioethanol production iodoform test and ethanol estimation. Method has been adapted and modified from Kuan et al. [10]. 5.0 g of fermented EFB and 1.0. |
|
4.5 The iodoform test Task
25 Feb 2014 The differences between methanol and ethanol will be carried over to the experiment with longer-chain alcohols which follow (P7172100) so that ... |
|
LABORATORY MANUAL OF EXPERIMENTAL ORGANIC
The Ritter test is a general test for alcohols or other The Lucas test and the iodoform test provide further structural information about the alcohol. |
|
Iodoform Reaction-Based Turbidimetry for Analysis of Alcohols in
8 Jun 2023 the iodoform test is necessary for the quantitation of legal alcohols —in which methanol. Page 6. Analytica 2023 4. 244 does not interfere ... |
|
Forensic Toxicology MODULE No. 21: Alcohol Poisoning SUBJECT
8.1 Ethanol (Ethyl Alcohol). For the detection of ethanol following tests are to be carried out in the exhibits. 8.1.1 Iodoform Test: Appropriate amount of |
|
Testsforfunctionalgroups - inorganiccompounds
Ethanol and secondary alcohols which contain CH3—CH(OH)R group (iodoform reaction) give positive iodoform test. To carry out reaction potassium iodide and |
|
Experiment 6 – Alcohols and Phenols
This type of alcohol will react with I2 in NaOH to give a yellow precipitate of iodoform CHI3. The reaction is shown below. R. C. R. OH. R. A tertiary alcohol. |
|
Lab 14: Qualitative Organic Analysis
Tests for the presence of 1° alcohols 2° alcohols |
|
Problem 4: Alcohol detective
The identity of the alcohol is then confirmed using standard test tube reactions (potassium dichromate and the iodoform test). Extension discussion points:. |
|
A brief freshman experience in qualitative organic analysis
tions of alkenes alcohols |
|
A Smart Colorimetric Platform for Detection of Methanol Ethanol and
13 ene 2022 The iodoform test was performed to identify the detected compound as some dyes can detect both methanol and ethanol. During the analysis |
|
Bioethanol Production from Empty Fruit Bunch using Direct
from catalysis reaction takes place in fermentation. In the second generation of 2.5 Bioethanol production iodoform test and ethanol estimation. |
|
The Haloform Reaction. XIV. An Improved Iodoform Test
iodoform is obtained. (4) In the case of methyl ketones heating is usually not necessary; alcohols react more. |
|
Identification of an Unknown – Alcohols Aldehydes and Ketones
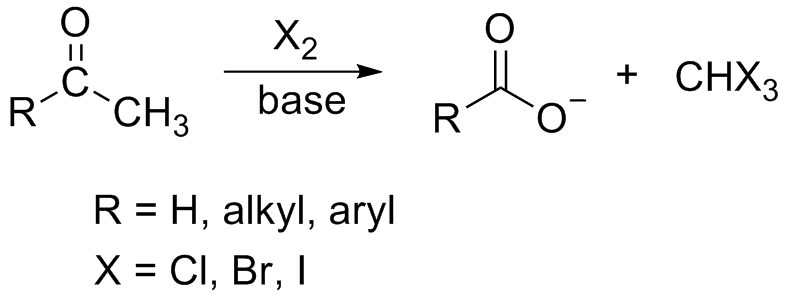
Iodoform Reaction: The iodoform test indicates the presence of an aldehyde or ketone in which one of the groups directly attached to the carbonyl carbon is |
|
Experiment 6 Qualitative Tests for Alcohols Alcohol Unknown IR of
We will do the iodoform test only twice with known alcohols once with an alcohol that contains the methyl secondary alcohol functionality and once with a |
|
INORGANICCOMPOUNDS - NCERT
Ethanol and secondary alcohols which contain CH3—CH(OH)R group (iodoform reaction) give positive iodoform test To carry out reaction potassium iodide and |
|
Identification of alcohols
Identification of Alcohols 7) Iodoform test 1 ml unk + 2 ml I2 solution (brown) + add dropwise NaoH 10 with continous rubbing &shaking till brown color |
|
Qualitative tests of Alcohols
22 nov 2018 · Iodoform test This test is given by acetaldehyde all methyl ketones and all alcohols containing CH3-CH-OH group When alcohol is warmed |
|
Jamaludin Al Anshori MSc Laboratory of Organic Chemistry
manual is designed to suit the need undergraduate student of chemistry The iodoform test also described earlier under alcohols is specific for |
|
6-Alcohols and Phenols
Iodoform Test The iodoform test is used to identify secondary alcohols that have a methyl group on the alcohol carbon This type of alcohol will react with |
|
45 The iodoform test Task
25 fév 2014 · Science - Chemistry - Organic Chemistry - 4 Alcohols (P7172000) 4 5 The iodoform test Experiment by: Anouch |
|
Iodoform Test - Description and Mechanism - Byjus
Iodoform Test is used to Check the Presence of Methyl Ketones in a given Unknown Compound It also gives Positive Result for Acetaldehyde and Ethyl Alcohol |
What is the iodoform test for alcohols?
Iodoform test for alcohol
Iodoform Test can be used to identify the presence of carbonyl compounds group in alcohols. The reaction of Iodine along with the base with methyl ketones results in the appearance of a very pale yellow precipitate of triiodomethane (previously known as iodoform).What is the indication of a positive iodoform tests for alcohols?
Indications of a positive test:
The formation of a yellow precipitate or suspension of iodoform is a positive test.How do you show ethanol in iodoform test?
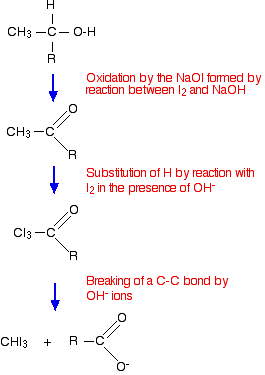
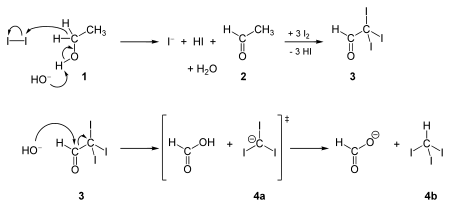
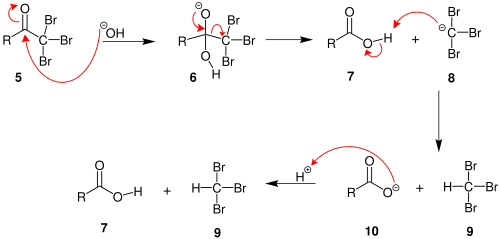
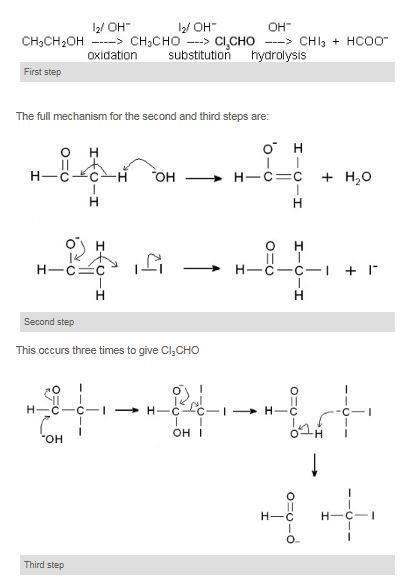
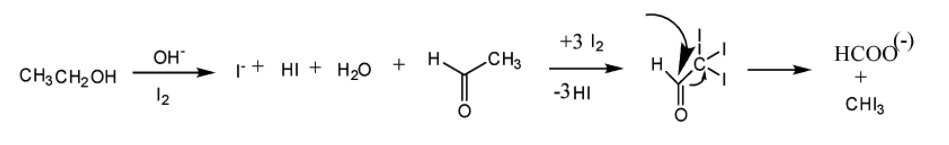
Ethanol forms acetaldehyde on oxidation, so it gives the iodoform test. The formation of a pale yellow precipitate when iodine in sodium hydroxide reacts with alcohol that can be oxidized to acetaldehyde is the iodoform reaction.- The iodoform test is used to identify secondary alcohols that have a methyl group on the alcohol carbon. This type of alcohol will react with I2 in NaOH to give a yellow precipitate of iodoform, CHI3.
|
Identification of an Unknown – Alcohols, Aldehydes, and Ketones
Iodoform Reaction: The iodoform test indicates the presence of an aldehyde or ketone in which one of the groups directly attached to the carbonyl carbon is a |
|
45 The iodoform test Task - Phywe
25 fév 2014 · Science - Chemistry - Organic Chemistry - 4 Alcohols (P7172000) 4 5 The Carry out the iodoform test with methanol and ethanol Use the |
|
LABORATORY MANUAL OF EXPERIMENTAL ORGANIC
The Ritter test is a general test for alcohols or other readily oxidizable functional groups such as aldehydes The Lucas test and the iodoform test provide further |
|
6-Alcohols and Phenols
The iodoform test is used to identify secondary alcohols that have a methyl group on the alcohol carbon This type of alcohol will react with I2 in NaOH to give a |
|
Identification of alcohols
Methanol ethanol glycerol (glycerin) isopropyl alcohol benzyl alcohol 1- Methanol, ethanol are very important organic solvents 7) Iodoform test 1 ml unk |
|
Iodoform test for aldehydes and ketones discussion - Squarespace
Regular alcohols don't react This test may be used to differentiation of most phenols from alcohol Apparatus: tubes Materials: chromonic acid, Tollen reagent , |
|
Iodoform test of aldehydes and ketones - Squarespace
Some alcohols, if not refined, may contain adide or ketone transplants Tollen test for Aldehydes Aldehyde standards Cyclohexanone and Benzaldehyde methods |