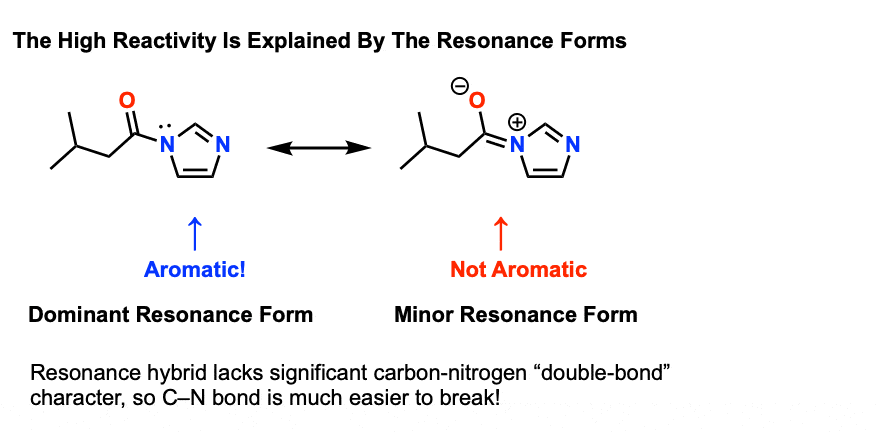
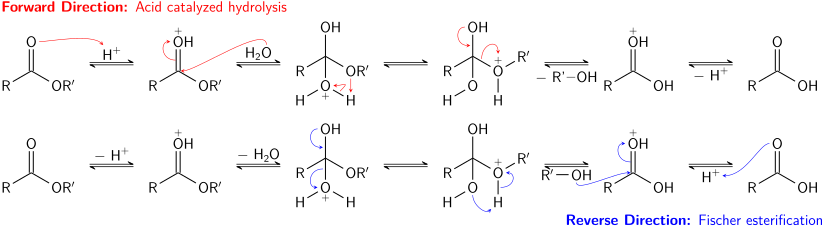
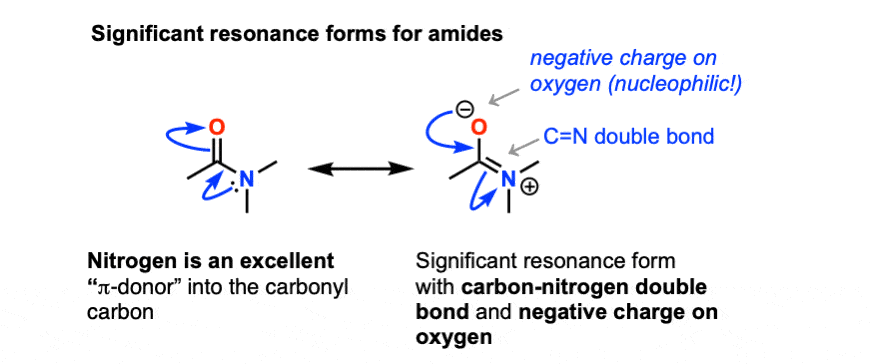
is acid hydrolysis of amides reversible
|
Lecture 6: Hydrolysis Reactions of Esters and Amides - Objectives
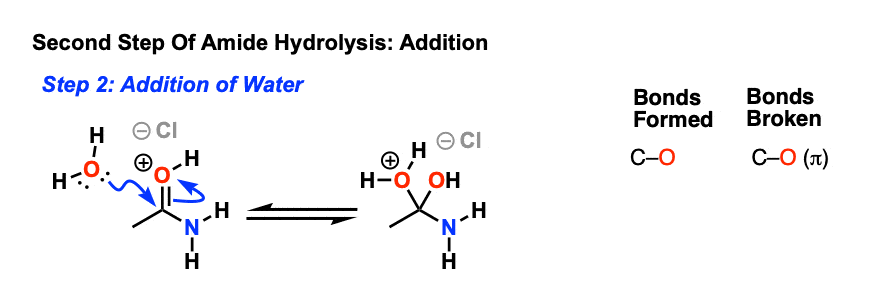
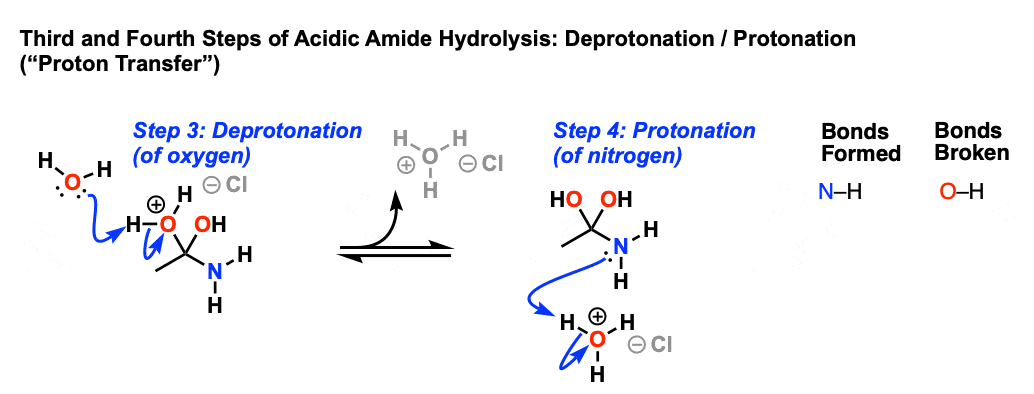
draw the mechanism of hydrolysis of amides under acidic and basic reaction Acid-Catalysed Hydrolysis of Methyl Esters ... This step is reversible. |
|
Reversible formation of intermediates during hydronium-catalyzed
Acid-catalyzed amide hydrolysis is generally considered to proceed by pathways involving irreversibly formed tetrahedral intermediates.1. |
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
alyzed ester hydrolysis is reversible but saponification is irreversible |
|
Sans titre
7.6 Hydrolysis of Amides. The reversibility of this reaction means that an amide can hydrolyze to form an amine and a carboxylic acid. |
|
20_13_16.html.ppt [Read-Only]
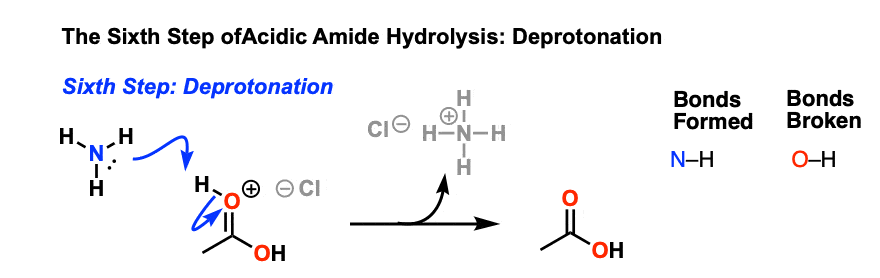
Hydrolysis of Amides. Hydrolysis of amides is irreversible. In acid solution the amine product is protonated to give an ammonium salt. |
|
Mechanisms of Acid Hydrolysis of Carboxylic Acid Esters and Amides
much less reversible than the hydrolysis of esters. Ordinary amides have w = 1.2 to 2.6 indicative of water acting as a nucleophile.28 |
|
Nâ•Alkylboronic acids as bifunctional reversible inhibitors of αâ
At %aw) % [S] o the hydrolysis of amides and esters catalysed by a-chymotrypsin can be represented as follows (1) [9] : h&. E+S -E+Pl+Pz. |
|
Reversible Twisting of Primary Amides via Ground State N–C(O
14 déc. 2017 ring16f |
|
Reversible Hydrolysis and Synthesis of Anandamide Demonstrated
8 nov. 1997 Reversible Hydrolysis and Synthesis of Anandamide. Demonstrated by Recombinant Rat. Fatty-Acid Amide Hydrolase. Yuko Kurahashi Natsuo Ueda |
|
Stable and ordered amide frameworks synthesised under reversible
hydrolysis of amides7 are of the order of 10. ?11 s. ?1. ) including in acidic or to acid chlorides followed by reaction with amines |
|
76 Hydrolysis of Amides
7 6 Hydrolysis of Amides The reversibility of this reaction means that an amide can hydrolyze to form an amine and a carboxylic acid |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
This step is reversible • Collapse of the tetrahedral intermediate back to a carbonyl compound can proceed either by expulsion of the hydroxyl ( |
|
Amide Hydrolysis Using Acid Or Base - Master Organic Chemistry
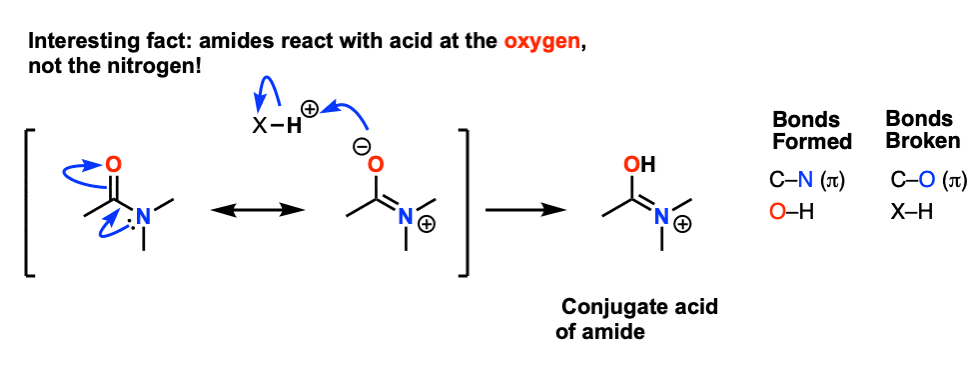
7 oct 2019 · As we noted the first step is the reversible protonation of the amide on oxygen to give the conjugate acid |
|
244: Hydrolysis of Amides - Chemistry LibreTexts
5 mar 2021 · Generally amides can be hydrolyzed in either acidic or basic solution The mechanisms are much like those of ester hydrolysis |
|
Mechanisms of Acid Hydrolysis of Carboxylic Acid Esters and Amides
much less reversible than the hydrolysis of esters Ordinary amides have w = 1 2 to 2 6 indicative of water acting as a nucleophile 28 |
|
Facilitated Reversible Formation of Amides from Carboxylic Acids in
The facilitative roleof carboxylic grouping on formation and hydrolysis of certain amido acids is shown to be mediated by formation of acid anhydrides |
|
Hydrolysis of Esters and Amides Dalal Institute
Also acidic ester hydrolysis is a reversible process and does not complete with 100 yield (like esterification) Page 2 434 A Textbook of Organic Chemistry |
|
THE HYDROLYSIS OF AMIDES IN THE ANIMAL BODY
namely that acetamide is mainly hydrolyzed in the rabbit because he could recover from the urine only a small fraction of the amide given As will be seen |
|
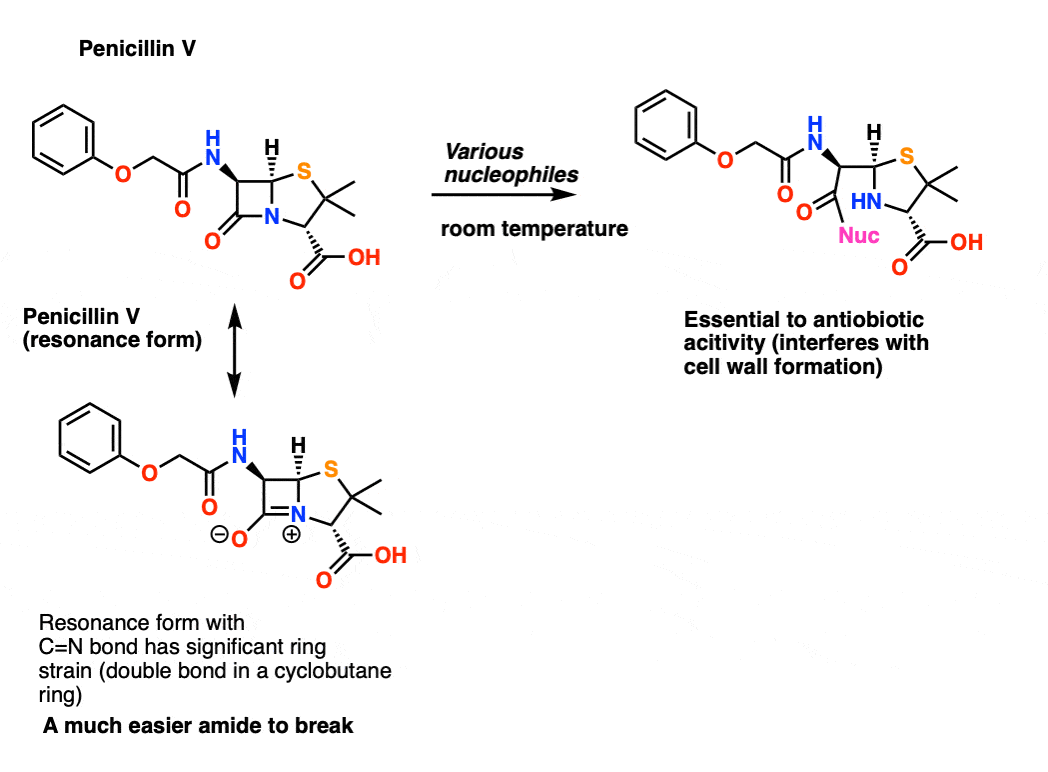
Stability of Medium-Bridged Twisted Amides in Aqueous Solutions
We examined the effect of structure on the reversibility of amide bond hydrolysis which we attributed to the transannular nature of the amino acid analogs |
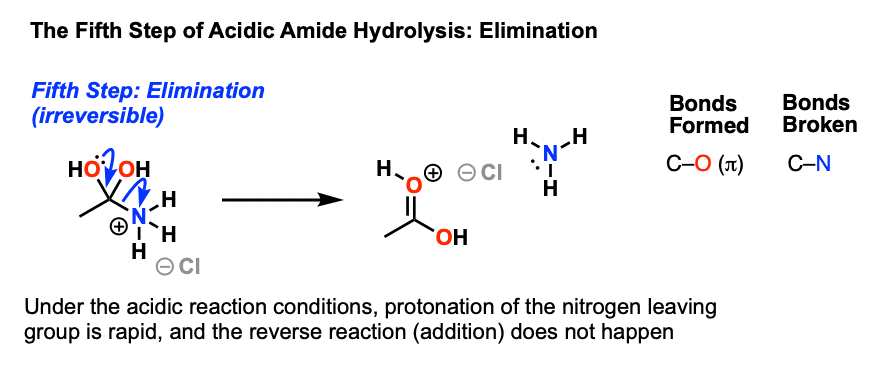
Is acid hydrolysis of amide reversible?
The reversibility of this reaction means that an amide can hydrolyze to form an amine and a carboxylic acid. This reaction is reversible exergonic.Why is acid catalyzed amide hydrolysis irreversible?
Acid-catalysed hydrolysis reactions are reversible. The forward reaction is driven over to product by using an excess of water, usually as the solvent. Base-mediated hydrolyses, however are essentially irreversible because the product is a non-electrophilic carboxylate salt.Is acid catalyzed hydrolysis reversible?
Acid-catalyzed hydrolysis of ester is reversible and occurs by SN1 pathway. Acid catalysts speed up the reaction by protonating carbonyl oxygen and thus rendering carbonyl carbon more susceptible to nucleophilic attack.- Hydrolysis is a reversible reaction, meaning that the alcohol and acid groups can react with each other to produce a polyester bond and water as a by-product.
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of hydrolysis of amides under acidic and basic reaction conditions Acid-Catalysed Hydrolysis of Methyl Esters This step is reversible |
|
76 Hydrolysis of Amides
The reversibility of this reaction means that an amide can hydrolyze to form an amine and a carboxylic acid This reaction is reversible exergonic + H2O _____ > |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
Acid-Catalyzed Ester Hydrolysis Because esterification of an acid with an alcohol is a reversible reaction (Sec 20 8A), esters can be hydrolyzed to carboxylic |
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · the acid-catalyzed hydrolysis of esters has seldom been the subject of reversible and using very harsh conditions 7,8 In this work, the |
|
COMMUNICATIONS - Chemistry Department at Brown University
cysteine nucleophile of the enzyme with reversible formation of a hemithioacetal adduct interested in developing catalysts of amide hydrolysis, and we reasoned that the pionic acid to tetrahydropyranone under neutral conditions in 100 |
|
51C Chapter 22 S15NEW
Both carboxylic acids and 1° and 2° amides can form hydrogen bonds, and When weaker nucleophiles add to aldehydes ketones, the reaction is reversible The hydrolysis of esters is a very slow reaction because water is a poor |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Acid halides • Amides • We begin our study of carbonyl compounds with the study of When an alcohol adds reversibly to an aldehyde or ketone, the product is called a Hydrolysis occurs in both acid and base media, but the reactions are |