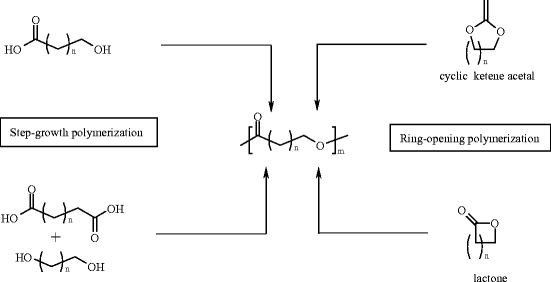
lactone hydrolysis in base
|
Chem 345 – Organic Reactions Chapter 21 Prepared by José Laboy
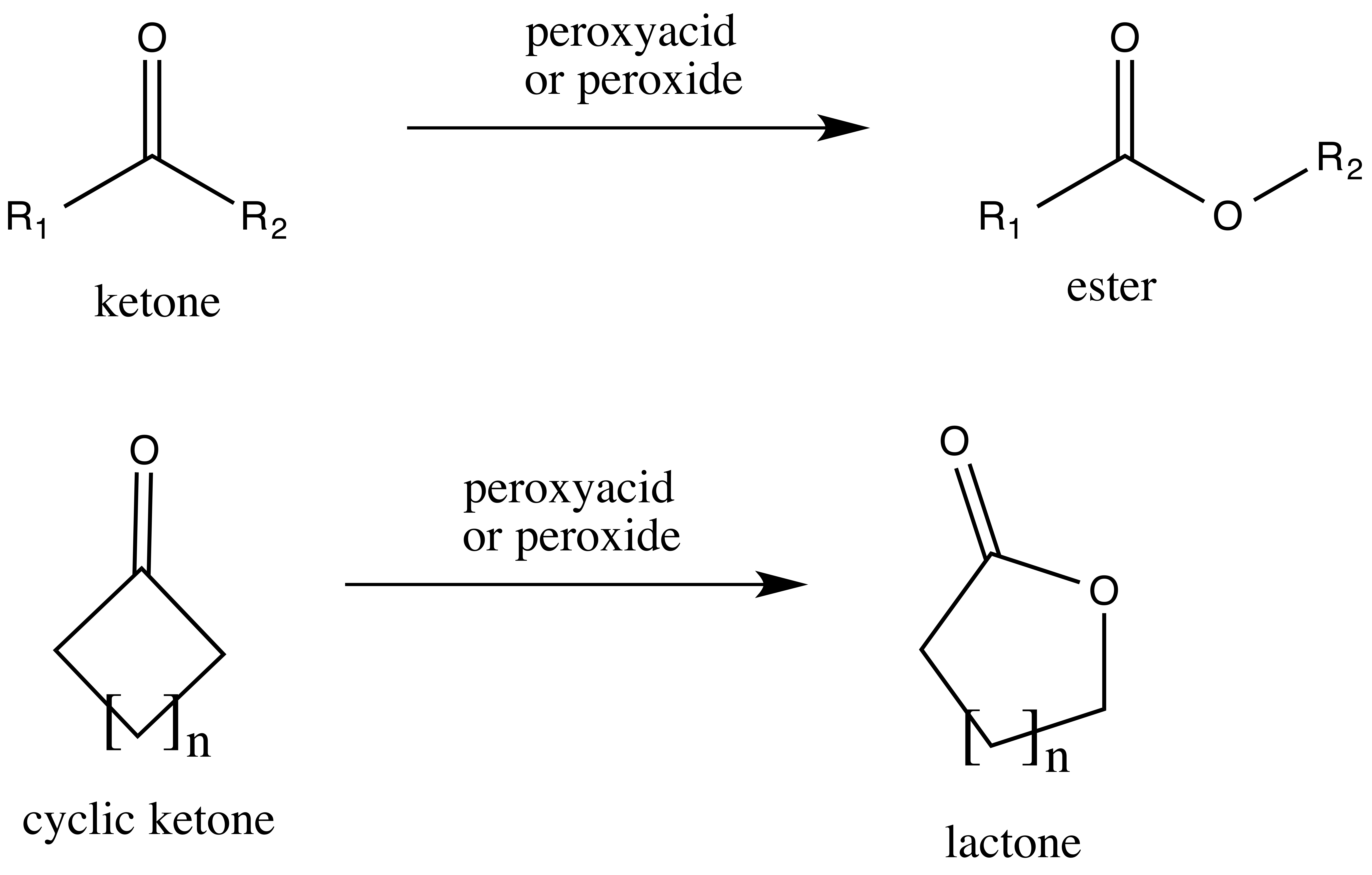
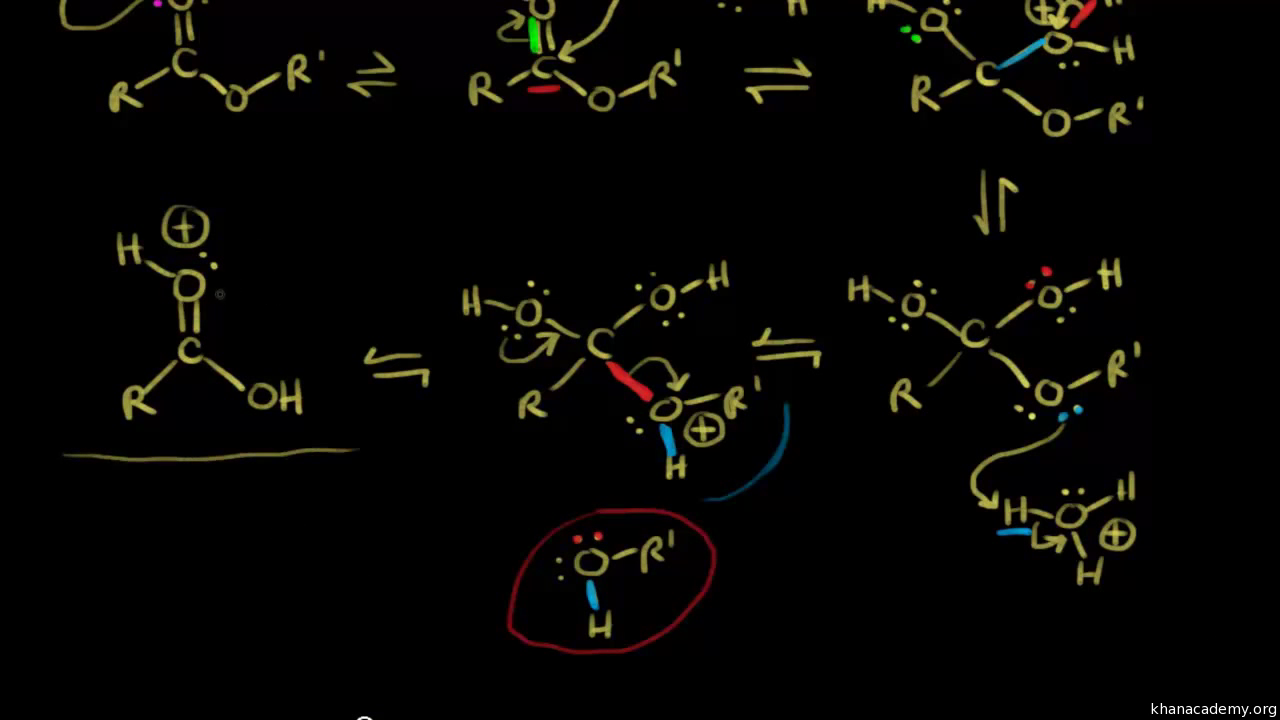
Base Catalyzed Lactone Hydrolysis. Reaction: Mechanism: Lactones are cyclic esters and readily and irreversibly hydrolyze in alkaline. |
|
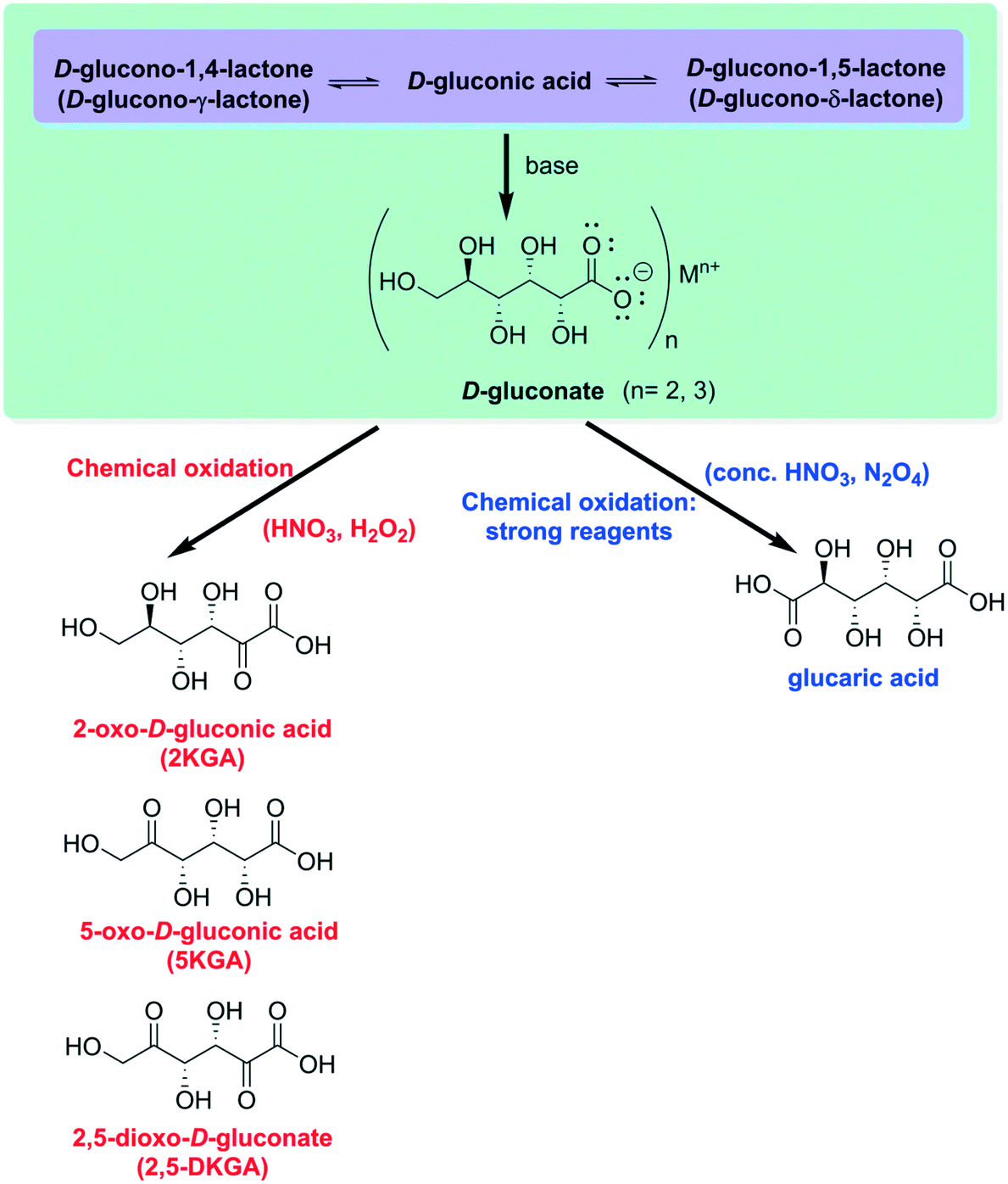
Hydrolysis of D-glucono-. delta.-lactone. II. Comparative studies of
and -lactones. The hydrolysis of D-glucono-S-lactone (eq 1) has been shown to be subject to bothintermolecular general acid and general base catalysis |
|
Hydrolysis of D-glucono-.delta.-lactone. I. General acid-base
Hydrolysis of D-Glucono-S-lactone. I. General Acid—Base. Catalysis Solvent. Deuterium. Isotope Effects |
|
Determination of the Mechanism of ?-Lactone Hydrolysis by a Mass
acid and basic hydrolysis of 7-lactones should in- volve splitting of the acyl-oxygen bond bond 5 |
|
The Hydrolysis of ?-Butyrolactone1
efficients for each of the following bases: carbonate ions phosphate ions and tetraborate ions. lactone and the study of its hydrolysis by polari-. |
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
term saponification can be used to refer to the hydrolysis in base of any carboxylic lactone from a hydroxy acid is nothing more than an intramolecular ... |
|
Formation and hydrolysis of lactones of phenolic acids
The hydrolysis of 8-hydroxy-1 -naphthoic acid lactone in the range of pH 0-10 also lactonization to that for base-catalyzed hydrolysis of the lactone. |
|
Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in Alkaline
Jun 9 2021 The classification of ester hydrolysis mechanisms proposed by Ingold [14] was adopted: the capital letters A and B stand for acid- and base- ... |
|
Iminolactones. II. Catalytic Effects on the Nature of the Products of
particularly those of a concerted acid-base nature. Results. The hydrolysis of from iminolactone hydrolysis is solely a function of pH. |
|
The Kinetics of Hydrolysis of ?-Propiolactone in Acid Neutral and
However with ß- lactones an added reaction with water enters.2. With /3-butyrolactone Olson and co-workers3·1 have shown that the hydrolysis in basic and in. |
|
Chem 345 – Organic Reactions Chapter 21
Base Catalyzed Lactone Hydrolysis Reaction: Mechanism: Lactones are cyclic esters and readily and irreversibly hydrolyze in alkaline |
|
Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions
11 jui 2013 · ABSTRACT: The neutral and base-catalyzed hydrolysis of nine carboxylic acid esters was studied using a hybrid supermolecule-PCM approach |
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · AAC2 to AAC1 hydrolysis takes place as acidity increases A parallel work addresses the neutral and base-catalyzed hydrolysis of lactones |
|
Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions
11 jui 2013 · The neutral and base-catalyzed hydrolysis of nine carboxylic acid esters was studied using a hybrid supermolecule-PCM approach including six |
|
Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in - MDPI
9 jui 2021 · BAC2 (base catalyzed nucleophilic addition followed by hydrolysis) and AAC2 (acid cata- lyzed hydrolysis) mechanisms are the two most important |
|
Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions
7 avr 2023 · Request PDF Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions The neutral and base-catalyzed hydrolysis of nine |
|
HYDROLYSIS
Hydrolysis reactions are generally enhanced by both acids and bases and three independent reaction mechanisms account for neutral acid and base hydrolysis |
|
Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in - NCBI
9 jui 2021 · The classification of ester hydrolysis mechanisms proposed by Ingold [14] was adopted: the capital letters A and B stand for acid- and base- |
|
Lactonization and protonation of gluconic acid - eScholarship
Though the formation of lactone is catalyzed by a hydrogen ion lactonization as well as its reverse reaction (lactone hydrolysis) does not alter the |
What is the hydrolysis reaction of lactone?
Hydrolysis of Lactones
Lactones (Cyclic esters) undergo typical reactions of esters including hydrolysis. Hydrolysis of the lactone under acidic conditions creates a hydroxyacid.What is the mechanism of base hydrolysis?
Mechanism of Base Hydrolysis of Esters
The electrophilic reagent that is present is attacked by hydroxide nucleotides at C=0; thus creating the tetrahedral intermediate. When the intermediate collapses, C=O will result in the loss of leaving the group alkoxide.What products are formed by hydrolysis of each lactone or lactam with acid?
Lactones belong to the category of cyclic esters and are obtained from hydrocarboxylic acids. The hydrolysis of lactone leads to the formation of hydroxyl carboxylic acid.- Lactone's hydrolysis condensation reaction is a reversible reaction with equilibrium. Lithium aluminium hydride reduces lactones to diols. The reaction involves the breaking of the ester bond and then the carboxylic acid group is reduced to the alcohol group. Polyesters are formed by lactones.
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · lactones 1 INTRODUCTION As is also the case with neutral and base-catalyzed mechanisms, the acid-catalyzed hydrolysis of esters has |
|
Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions
11 jui 2013 · ABSTRACT: The neutral and base-catalyzed hydrolysis of nine carboxylic acid esters was studied using a hybrid supermolecule-PCM |
|
Base Catalyzed Lactone Hydrolysis - Chem 345 – Organic
Lactones are cyclic esters and readily and irreversibly hydrolyze in alkaline media to produce a hydroxycarboxylate Although carboxylic acids are weak acids they are completely ionized in basic media |
|
HYDROLYSIS
Hydrolysis reactions are generally enhanced by both acids and bases and three independent reaction mechanisms account for neutral, acid and base |
|
STEROID LACTONES - ScienceDirectcom
the Worcester Foundation for Experimental Biology, The oxidation of a steroid ring ketone to a lactone according to the scheme - ? / C=O in 57 to 63 per cent yield, was purified by alkaline hydrolysis, followed by carbonation of the |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
term saponification can be used to refer to the hydrolysis in base of any carboxylic acid lactone from a hydroxy acid is nothing more than an intramolecular |
![PDF] Mechanisms of lactone hydrolysis in neutral and alkaline PDF] Mechanisms of lactone hydrolysis in neutral and alkaline](https://ars.els-cdn.com/content/image/3-s2.0-B9780123736468500093-u06-56-9780123736468.jpg)












![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://demo.vdocuments.mx/img/378x509/reader018/reader/2020011320/55a14cab1a28ab4e398b45bd/r-2.jpg)
![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://i.ytimg.com/vi/7s2cdaQtVs0/maxresdefault.jpg)

![Studies on the B AL 2 mechanism for ester hydrolysis - [PDF Document] Studies on the B AL 2 mechanism for ester hydrolysis - [PDF Document]](https://0.academia-photos.com/attachment_thumbnails/48945268/mini_magick20190202-1828-122ecn0.png?1549098871)




![PDF] Mechanisms of lactone hydrolysis in neutral and alkaline PDF] Mechanisms of lactone hydrolysis in neutral and alkaline](https://imgv2-2-f.scribdassets.com/img/document/290613517/149x198/72fdf1f7a5/1448097314?v\u003d1)











![PDF] Mechanisms of lactone hydrolysis in neutral and alkaline PDF] Mechanisms of lactone hydrolysis in neutral and alkaline](https://static-01.hindawi.com/articles/jpol/volume-2015/157267/figures/157267.fig.009.svgz)

