infusion systems - Conception
|
Conception et réalisation dune micropompe intelligente
23 janv. 2014 Les MEMS (en anglais : MicroElectroMechanical Systems) développés au ... l'infusion lente de médicaments (injection de quelques microlitres ... |
|
MDCG 2021-24 Guidance on classification of medical devices
The explanations provide some simplified concepts and are not exhaustive. For details see the intended for use with an infusion pump. |
|
POMPE A PERFUSION Autres désignations Pompe volumétrique
Infusion Systems. Applications médicales. Utilisation Principale : conception et de fonctionnement. Le système de détection de fin de poche se fait. |
|
MDCG 2021-24 Guidance on classification of medical devices
The explanations provide some simplified concepts and are not exhaustive. For details see the intended for use with an infusion pump. |
|
AVIS révisé de lAnses relatif à lactualisation des repères
23 déc. 2019 Source : IOM Pregnancy Weight Guidelines (Rasmussen Catalano |
|
ATTESTATION CE / EC CERTIFICATE
Ambulatory infusion pumps and accessories. pour la conception la production et le contrôle final - des dispositifs médicaux énumérés ci-dessus est ... |
|
Using diabetes technology in pregnancy
Diabetes technology currently refers to insulin pump therapy (continuous subcutaneous insulin infusion (CSII)) glucose sensors (real-time continuous glucose |
|
Une approche paradigmatique de la conception architecturale des
“Fundamental concepts or properties of a system in its environment La nécessité de contrôler et réguler les systèmes à bon escient (infusion des apports ... |
|
Les années 1970–1980 et lacquisition de la « preuve du concept
Group on Artificial Insulin Delivery Systems and Pancreas Transplantation » de l'Association Européenne pour l'Étude du Diabète (European Association for |
|
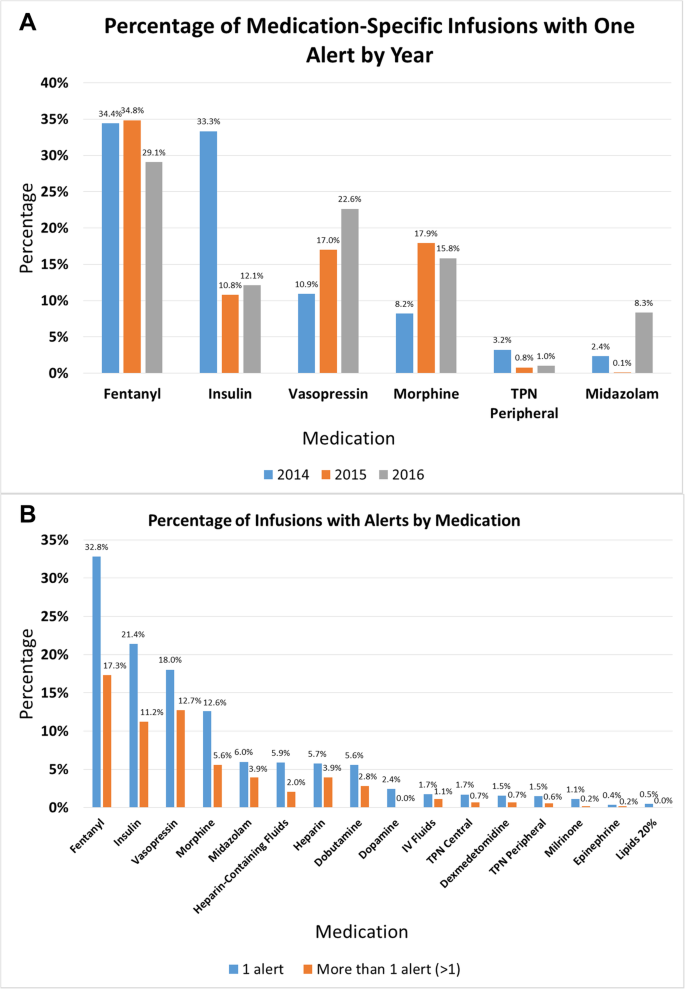
Building a Smart Infusion Figure 1 Phases drug order tes ting
Figure 1 Phases System Drug Library Introduction smart infusion system is designed to minimize programming errors and the related risk of patient harm Each facility develops a customized infusion system drug library which includes dosing ranges and other safety limits for individual drugs |
|
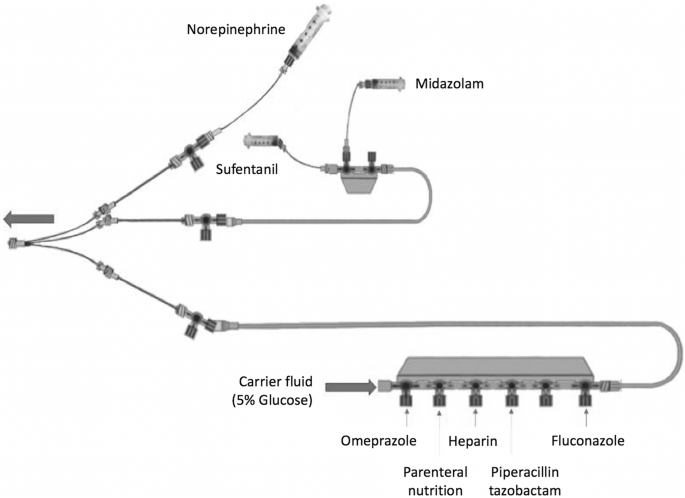
WIRELESS MEDICAL INFUSION - NIST
10 begins the process to identify the actors interacting with infusion pumps define the 11 interactions between the actors and the system perform a risk assessment identify 12 mitigating security technologies and provide an example implementation 13 Clinicians and patients rely on infusion pumps for safe and accurate administration of |
|
Infusion Systems Product Catalog - BBraunUSAcom
Infusion Systems INFUSION SYSTEMS Description Product Code Unit Per cs Infusomat® Space Pump Sets with SAFELINE ® Needleless IV System and Pre-attached ASV Valve (60 drops/mL) Universal spike slide clamp pressure limited check valve two SAFELINE injection sites 82 in and 6 in |
|
Evaluation of Intravenous Infusion Systems for Adenosine
adenosine infusion actually occurs during co-administration of a radiopharmaceutical into the infusion line MATERIALS AND METHODS Five infusion systems were evaluated They were a gravity infusion system (Buretrol Travenol Labs Deerfield IL) two syringe pumps (Harvard Apparatus Co Millis MA Model |
|
0118 - Intravascular Devices and Infusion Systems
Topic: 01 18 - Intravascular Devices and Infusion Systems 01 18 - Policy 11-28-22 - Revised 1994 - Author 01 18 - Intravascular Devices and Infusion Systems Purpose To provide infection control guidelines for the proper placement and management of intravascular devices and infusion systems |
|
Technology Infusion for Complex Systems: A Framework and Case
2Engineering Systems Division Massachusetts Institute of Technology 77 Massachusetts Avenue Cambridge MA 02139 TECHNOLOGY INFUSION FOR COMPLEX SYSTEMS: CASE STUDY Received 1 July 2008; Revised 16 March 2009; Accepted 24 March 2009 after one or more revisions Published online 17 June 2009 in Wiley InterScience (www interscience wiley com) |
|
VANTAGECONTROLSCOM VANTAGE TECHNOTES
Every system upgrade will need the following hardware i e a Main Enclosure Terminal Board and correct InFusion Controller or a DIN InFusion Controller Please enter the quantity needed for each of these items based on the information determined from TABLE # ONE above |
|
Searches related to infusion systems conception filetype:pdf
Infusion time must be documented (start and stop) If health care professional administering substance/drug is continuously present to administer injection and observe the patient bill as a Push If infusion time is 15 minutes or less bill as a Push Infusion intervals of > 30 minutes beyond 1-hour |
What is an infusion system?
- An infusion system is the process by which an infusion device and any associated disposables are used to deliver fluids or drugs in solution to the patient by the intravenous, subcutaneous, epidural or enteral route. Healthcare professional Clinicians judgement. Always in accordance with Manufacturers instructions/directions.
What is a concurrent infusion?
- Concurrent Infusions – infusion of a new substance/drug at the same time as another substance/drug through same IV line or when two distinct infusions are given in two separate lumens in a multi-lumen catheter IV site. Not time-based and may only be reported once per day.
How do I report a first IV push after a TX/Pro/DX infusion?
- Example: First IV push given subsequent to an initial one-hour tx/pro/dx infusion is reported using a subsequent IV push code. Do not code first IV push with code 96374 (initial) but rather code 96375 for first IV push given after (subsequent to) the initial infusion.
What is the maximum hang time for an infusion?
- There is no nationally published guideline recommending a maximum hang time for an infusion. If longer hang times are used, hospital leadership; medical, nursing and pharmacy staff; infection control; risk management; and other stakeholders should weigh the risk of infection against the need to conserve intravenous solutions.
|
Infusion pump standards guide - NAMDET
10 mar 2014 · Examples of product standards that are applicable to infusion pump design opportunity for the manufacturer to state their intended concept of |
|
Optimisation dun processus de conception par - Thèses
4 jui 2019 · Etat de l'art de la conception des dispositifs médicaux au regard de la prise and a new user interface for an infusion pump developed with a |
|
SUMMARY OF PRODUCT CHARACTERISTICS - Sanofi India
When used with an insulin infusion pump or intravenously, Apidra® should not be indicates no safety issues in use of insulin glulisine during pregnancy or on |
|
UNDERSTANDING INSULIN PUMPS - Diabetes Australia
planning for, and during, pregnancy 4 Understanding insulin CGM System is compatible with the Animas Vibe insulin infusion pump (distributed by AMSL) |
|
External Insulin Infusion Pump - Blue Shield of California
1 août 2020 · A first-time external insulin infusion pump with or without continuous glucose monitoring women with diabetes is also lower in pregnancy |




![PDF] Developing the control system of a syringe infusion pump PDF] Developing the control system of a syringe infusion pump](https://i1.rgstatic.net/publication/261048886_Design_and_development_of_insulin_delivery_system_prototype/links/58db327345851578dfe88af3/largepreview.png)




![PDF] Control System Design of Syringe Infusion Pump and MATLAB PDF] Control System Design of Syringe Infusion Pump and MATLAB](https://i1.rgstatic.net/publication/329419659_Efficient_treatment_of_chronic_endometritis_through_a_novel_approach_of_intrauterine_antibiotic_infusion_a_case_series/links/5fc23625a6fdcc6cc677c31e/largepreview.png)





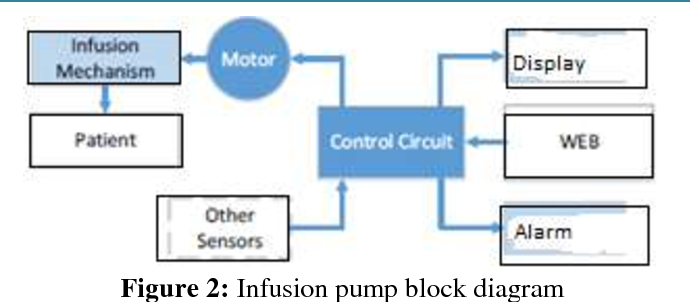
![PDF] Control System Design of Syringe Infusion Pump and MATLAB PDF] Control System Design of Syringe Infusion Pump and MATLAB](https://d3i71xaburhd42.cloudfront.net/ec794453f0e1b51fbbd3424b3d860410b8514fa1/2-Figure3-1.png)








![PDF] Developing the control system of a syringe infusion pump PDF] Developing the control system of a syringe infusion pump](https://0.academia-photos.com/attachment_thumbnails/34693564/mini_magick20190321-8138-1th83bx.png?1553190048)



![PDF] Control System Design of Syringe Infusion Pump and MATLAB PDF] Control System Design of Syringe Infusion Pump and MATLAB](https://rsds.org/wp-content/uploads/2014/12/low-dos-ketamine-infusion-emergency-dept-patients-severe-pain-pdf.jpg)



![PDF] Developing the control system of a syringe infusion pump PDF] Developing the control system of a syringe infusion pump](https://i1.rgstatic.net/publication/335169966_Types_and_Frequency_of_Infusion_Pump_Alarms_and_Infusion-Interruption_to_Infusion-Recovery_Times_for_Critical_Short_Half-Life_Infusions_Retrospective_Data_Analysis/links/5d5d2dbb299bf1b97cfa6b70/largepreview.png)

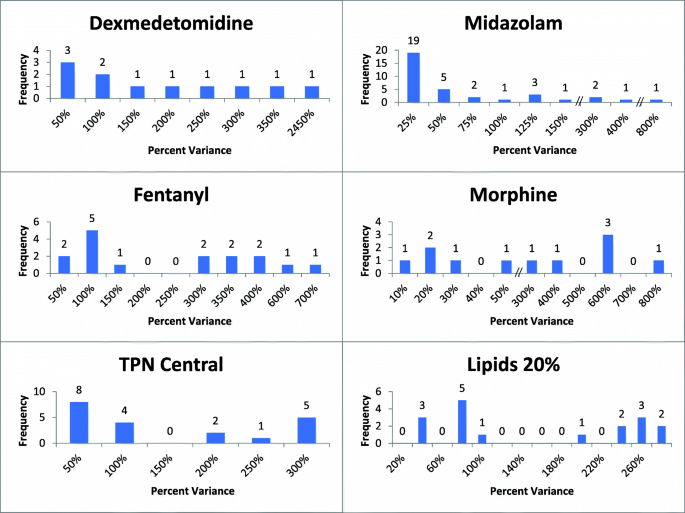
![PDF] Control System Design of Syringe Infusion Pump and MATLAB PDF] Control System Design of Syringe Infusion Pump and MATLAB](https://onlinelibrary.wiley.com/cms/asset/9e7d725e-1c5e-48d5-b029-11042e8e97b6/dmrr2653-fig-0002-m.jpg)