list of buffer solutions
|
Buffering agents and Buffers
?Additives for buffer solutions preparation. ?Formulated buffers by techniques (alphabetic list and other biochemical types: buffers |
|
Biological Buffers
How can microbial contamination of buffer solutions be prevented? Alphabetical product list of biological buffers supplied by AppliChem. |
|
4.1.3. BUFFER SOLUTIONS
0.1 M Phosphate buffer solution pH 3.0. 4011500. Dissolve 12.0 g of anhydrous sodium dihydrogen phosphate R in water R adjust the pH (2.2.3) with dilute. |
|
A Guide to HPLC and LC-MS Buffer Selection
Table 2 lists several additional buffers that are sufficiently volatile for. LC-MS use. with 30% of a 33 mM phosphate buffer solution to achieve 70% ACN. |
|
PH Buffer Solutions
pH Buffers Solutions. Guaranteed Traceability. Reagecon's pH buffer standards are directly traceable to the IUPAC pH scale by an unbroken chain of |
|
AN ECONOMIC EVALUATION OF BUFFER PREPARATION
Table 2: Buffer list. Using buffer stock blending a wide range of buffers can be prepared from a relatively small number of stock solutions. |
|
List of HIV Diagnostic test kits and equipments classified according
8 days ago The list is an overview of HIV RDTs to assist Principal Recipients (PRs) of Global Fund ... Buffer solution included: 1 bottle × 5mL/bottle. |
|
LOBA Chemie Price List 2021-2022 with1st Amendment
ACETATE BUFFER SOLUTION pH 4.6. HSN: 3822.0090. CAS No.: NA. 00522 00500. 500 ml. 190.00. ACETATE BUFFER TS ACC. TO USP. HSN: 3822.0090. CAS No.: NA. |
|
Buffers
Buffer solutions are composed of a weak acid (the proton In most biochemical and toxicological work it is customary to list constants. |
|
Section 191 Acid-Base Buffer Solutions
A buffer is a concentrated solution of a weak acid (or base) together with a salt containing the conjugate base (or acid) How does a Buffer work? The Common- |
|
413 BUFFER SOLUTIONS - ResearchGate
Phosphate buffer solution pH 2 0 4007900 Dissolve 8 95 g of disodium hydrogen phosphate R and 3 40 g of potassium dihydrogen phosphate R in water R |
|
BUFFER SOLUTIONS
A buffer is an aqueous solution consisting of a mixture of a weak acid and its salt ( acidic buffer ) or a weak base and its salt ( basic buffer ) Its pH |
|
Buffers Booklet - The Wolfson Centre for Applied Structural Biology
We are pleased to present to you the newest edition of Buffers: A Guide for the Preparation and Use of Buffers in Biological Systems |
|
41 reagents standard solutions buffer solutions
Some of the reagents included in the list are toxic and are to be handled in conformity with good quality control laboratory practice Reagents in aqueous |
|
Buffer Solution - Acidic and Basic Buffers Preparations Examples
21 nov 2019 · There exist a few alternate names that are used to refer to buffer solutions such as pH buffers or hydrogen ion buffers |
|
Biological Buffers
Saturated concentrations of buffers in solution at 0°C • “Old” buffers replaced by buffers with better properties Alphabetical list of biological buffers |
|
Exp 16 Buffer solution Sp07
A buffer is a solution that resists the change in pH when limited amounts of acid or base Bronsted-Lowry acid is present list the strongest one first |
|
Lab 6 - BUFFER SOLUtiONS
Buffer: is a solution that resist changes in pH caused by the addition of a strong acid or base It contain either a weak acid and its conjugate base (the salt |
|
A Guide to HPLC and LC-MS Buffer Selection
Table 2 lists several additional buffers that are sufficiently volatile for LC-MS use Ammonium formate (2 7 |
What are 4 examples of buffer solution?
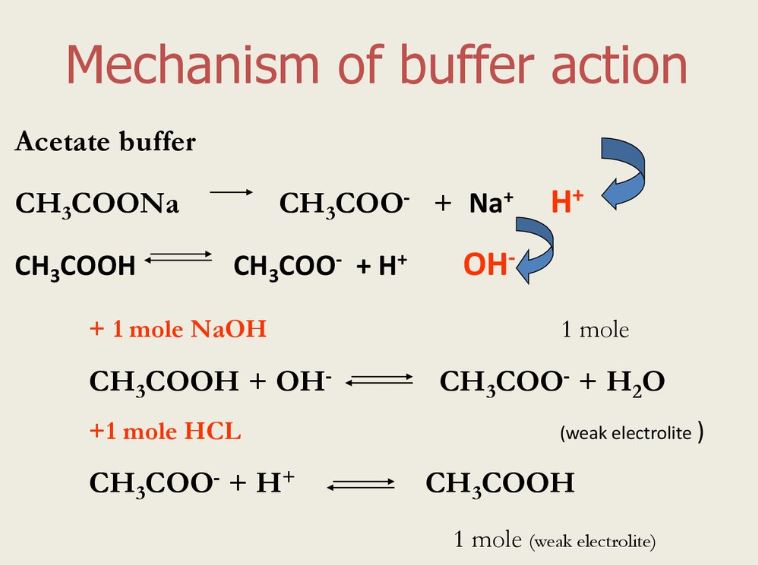
A solution of acetic acid and sodium acetate (CH3COOH + CH3COONa) is an example of a buffer that consists of a weak acid and its conjugate base, whereas a solution of ammonia and ammonium chloride (NH3 + NH4Cl) is an example of a buffer that consists of a weak base and its conjugate acid.What is buffer solution PDF?
Buffers are compounds or mixtures of compounds that, by their presence in solution, resist changes in pH upon the addition of small quantities of acid or alkali The resistance to a change in pH is known as buffer action.What are the types of buffer solutions?
There are two types of buffer solutions: acidic buffer and basic buffer: A solution with weak acid and its salts containing strong bases is called an acidic buffer solution. E.g., A solution with CH3COOH, which is weak acid and CH3COONa, which is its salt is an acidic buffer solution.- A buffer solution is defined as a solution which resists any change in its pH value even when small amounts of the acid or the base are added to it. e.g Ammonium acetate acts as a buffer.
|
Buffering agents and Buffers - INTERCHIM
Classic used buffering agents are mineral buffers [Phosphate, Tris, Borate, Citrate, Glycine ], but also several organic buffers [Glycine, DEA, ] |
|
The SOLUTION for All of Your Buffer Needs - The Wolfson Centre for
preparation of solutions from concentrated stocks with minimal pH effects from the dilution of buffer components A list of these zwitterionic buffers and their pKa |
|
Buffers Booklet - University of San Diego Home Pages
Please call us and ask for a current listing of our ever expanding Technical Resource Buffer solutions are composed of a weak acid (the proton donor) and its |
|
Buffer Table - Hampton Research
Solutions for Crystal Growth Buffer Table Crystal Growth 101 page 1 Buffer Potassium chloride Citric acid pKa 25°C (1) 3 1 (2) 4 8 (3) 6 4 Sodium citrate |
|
Buffer solutions - ResearchGate
Buffer solutions have the useful property of resisting changes in pH when Preparation of the buffer solution: mix stock A and stock B according the list below, |
|
Biological Buffers - ResearchGate
9 Technical tips 10 • How can microbial contamination of buffer solutions be prevented? pKa values of biological buffers (25°C, 100 mM), alphabetical list 16 |
|
Buffers A guide for the preparation and use of buffers in biological
Buffer solutions are composed of a weak acid (the proton donor) and its In most biochemical and toxicological work, it is customary to list constants based on |
|
Buffer Investigations - Vernier
will determine the buffering ability of a baking soda solution You will first use a pH List five materials whose buffering action might be interesting 6 List two |











![Top PDF History of Buffer Overflow [lucid78] pdf - 1Library Top PDF History of Buffer Overflow [lucid78] pdf - 1Library](https://img.youtube.com/vi/lsHq5aqz4uQ/0.jpg)