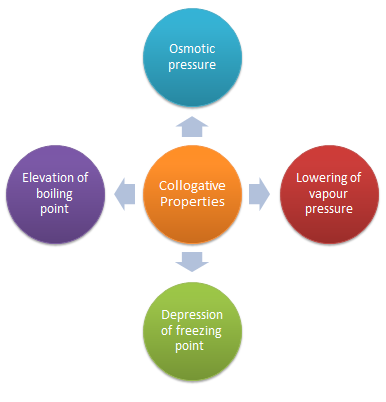
list three colligative properties of solutions
|
16.3 Colligative Properties of Solutions
In this section you will discover how a solute can change the freezing point of a solution. Guide for Reading. Key Concepts. • What are three colligative. |
|
MODULE HANDBOOK CHEMISTRY 1
Jan 7 2020 3. Private learning : 2 x 60 = 120 minutes (2 hours) per week. Credit points ... Solution |
|
How to develop colligative properties of solution chemistry e- book
ISBN: 978-602-6697-36-3. 225. How to develop colligative properties of solution chemistry e- book based science process skills approach with 4S TMD models. |
|
Lehninger Principles of Biochemistry 4th Edition
in solutions than in pure water. The effect of solute con- centration on the colligative properties of water is in- dependent of the chemical properties of |
|
MATA KULIAH COURSE Nama Mata Kuliah Course Name : Kimia I
Course Name. : Kimia I 3 sks. Semester. : I. DESKRIPSI MATA KULIAH. Description of Course ... Solution Concentration |
|
Chemistry Assessment: #3 Part 1 Solubility and Colligative Properties
Write the equation for molarity: 3. Explain in words how molality and molarity differ. 4. What is the molality of a solution in which 0.32 moles AlCl3 has been |
|
2nd Semester Chem FINAL EXAM GUIDELINE & REVIEW
3-5 Write the formula for each of the following: a. sulfur trioxide ____SO3___ Name the three colligative properties of solutions: _vapor. |
|
Experiment on Colligative properties Colligative properties are the
The list of colligative properties includes: PARTICLES IN THE SOLUTION and completely ... 3. Pabove solution < Pabove pure solvent. Pressure P(atm). |
|
SECTION 16.1 PROPERTIES OF SOLUTIONS (pages 471–477)
Feb 17 2012 Name three factors that influence the rate at which a solute dissolves in a solvent. ... SECTION 16.3 COLLIGATIVE PROPERTIES OF. SOLUTIONS ... |
|
Using the Principle of Colligative Properties in Food Science
When increasing the amount of sugar in the solution does the boiling point increase or decrease? Given these senso- rial changes |
|
163 Colligative Properties of Solutions
In this section you will discover how a solute can change the freezing point of a solution Guide for Reading Key Concepts • What are three colligative |
|
Colligative Properties
Properties of solutions that depend on the number of molecules present and not on the kind of molecules are called colligative properties |
|
Colligative properties of solutions
Colligative properties of solutions (diffusion osmosis) are vital for the organism Osmosis stipulates distribution of water and nutrients among different |
|
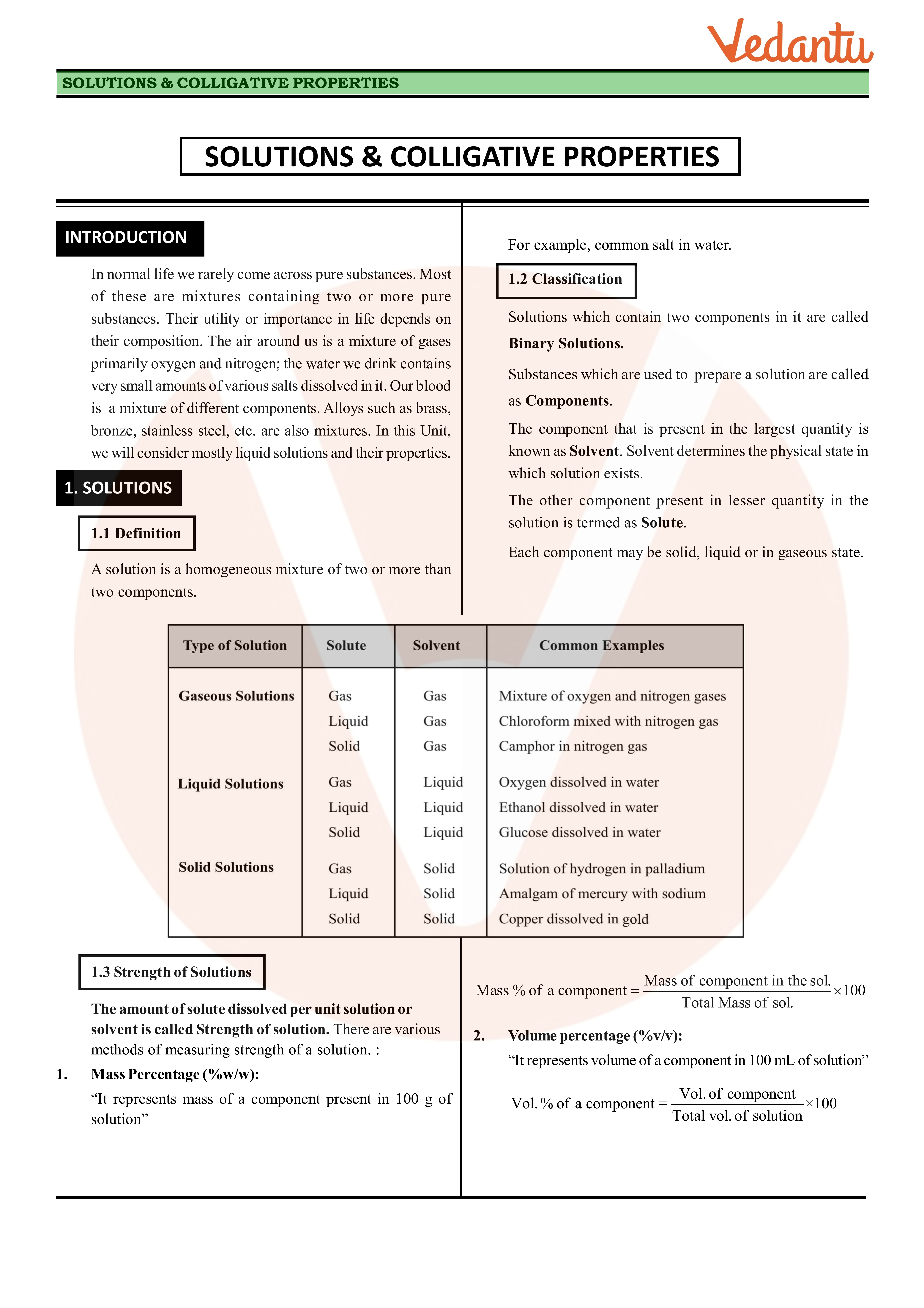
SOLUTIONS & COLLIGATIVE PROPERTIES - GGPS Bokaro
SOLUTIONS COLLIGATIVE PROPERTIES 3 Mass by volume percentage ( w/v): “It represents mass of solute in grams present in 100 mL of solution” |
|
Solutions and Colligative Properties - Saptarshi Classes
Formation/Preparation: The homogeneous solution is formed due to force of attraction between the molecules or particles of solute and solvent Heterogeneous |
|
Colligative Properties - Definition Types Examples Raoults Law
There are different types of colligative properties of a solution which include vapour pressure lowering boiling point elevation freezing point depression |
|
SOLUTIONS AND COLLIGATIVE PROPERTIES - MasterJEE Classes
COLLIGATIVE PROPERTIES 1 INTRODUCTION (a) Solutions are homogenous mixtures of two or more substances (b) We call what is being dissolved in the |
|
Experiment on Colligative properties
The list of colligative properties includes: a) lowering vapor pressure above a solution; b) freezing temperature depression; c) boiling temperature elevation; |
What are 3 colligative properties of a solution?
Colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.What are the three most common colligative properties?
These colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.Are there 4 colligative properties?
There are four colligative properties: vapor pressure lowering, boiling point ele- vation, freezing point depression, and osmotic pressure.- Examples of colligative properties include vapor pressure lowering, freezing point depression, osmotic pressure, and boiling point elevation.
|
Experiment 1: Colligative Properties - ULM
measuring the freezing point depression of a solution of this solute in a solvent as compared to the freezing point of the pure solvent Background: Colligative properties are properties of a solvent, such as freezing point along with all three graphs This should give you about a 6 or 7 page lab report, not all pages will be |
|
CHAPTER 13
whereas one formula unit of calcium chloride gives three ions in solution Compounds in Cl– Ions in Aqueous Solutions and Colligative Properties 419 SECTION 1 Sample Problem A Write the equation for the dissolution of aluminum |
|
Solutions - NCERT
studying the properties of the solutions, like vapour pressure and colligative properties We will begin with types of Henry was the first to give a quantitative relation break some of the hydrogen bonds between them Due to weakening of |
|
SOLUTIONS - Foothill High School
17 fév 2012 · SECTION 16 1 PROPERTIES OF SOLUTIONS (pages 471–477) This section Name three factors that influence the rate at which a solute dissolves in a solvent a b c 3 SECTION 16 3 COLLIGATIVE PROPERTIES OF |
|
?mIL= 259*1402%45=0
Chapters 13 &14 – Solutions, Net Ionic Equations, and Colligative Properties Vocabulary Using a table of solubility rules, be able to write net ionic equations Unit 12 Bell Work Three ways to increase the rate of dissolving are: 1 2 3 |
|
ColHgafive Properties: Freezing-Point Depression and Molar Mass
To become familiar with colligative properties and to use them to determine the molar mass states that the freezing point and boiling point of a solution differ Dom those List three colligative properties and suggest a rationale for the choice |






























/glass-saucepan-on-a-gas-burner-with-boiling-water-dor961844-58fced4e3df78ca159b1f51c.jpg)


