mesomeric effect pdf
|
Mesomeric effect
11 mars 2021 The net electron flow from or to the substituent is determined also by the inductive effect. The mesomeric effect as a result of p-orbital ... |
|
BSc Chemistry
There are four types of electronic effects mainly viz. inductive effect |
|
Mesomeric effect & its application.pdf
Mesomenic effect: effect. It is a mosomeric permament offer an extends in the molecule as. The structure ... Cannonical structure is called resonance. |
|
NOTE ON THE INDUCTIVE AND MESOMERIC EFFECTS IN
The purely inductive effect is obtaiiled if we put the resonance integral between these two2 atoms p |
|
Questions and Answers – Mesomeric Effect 1. In mesomeric effect
Answer: d. Explanation: In the mesomeric effect the electrons are transferred from multiple bonds to an atom |
|
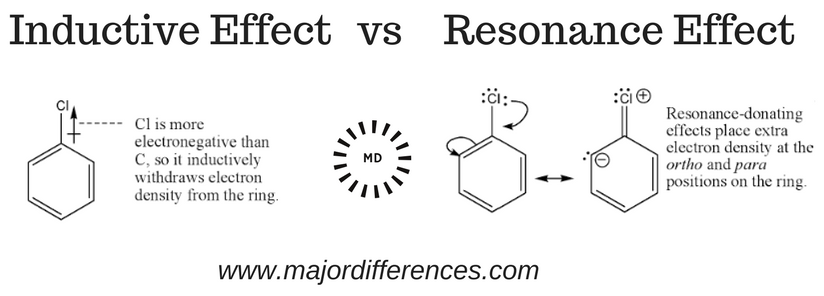
Inductive effect is defined as - permanent displacement of shared
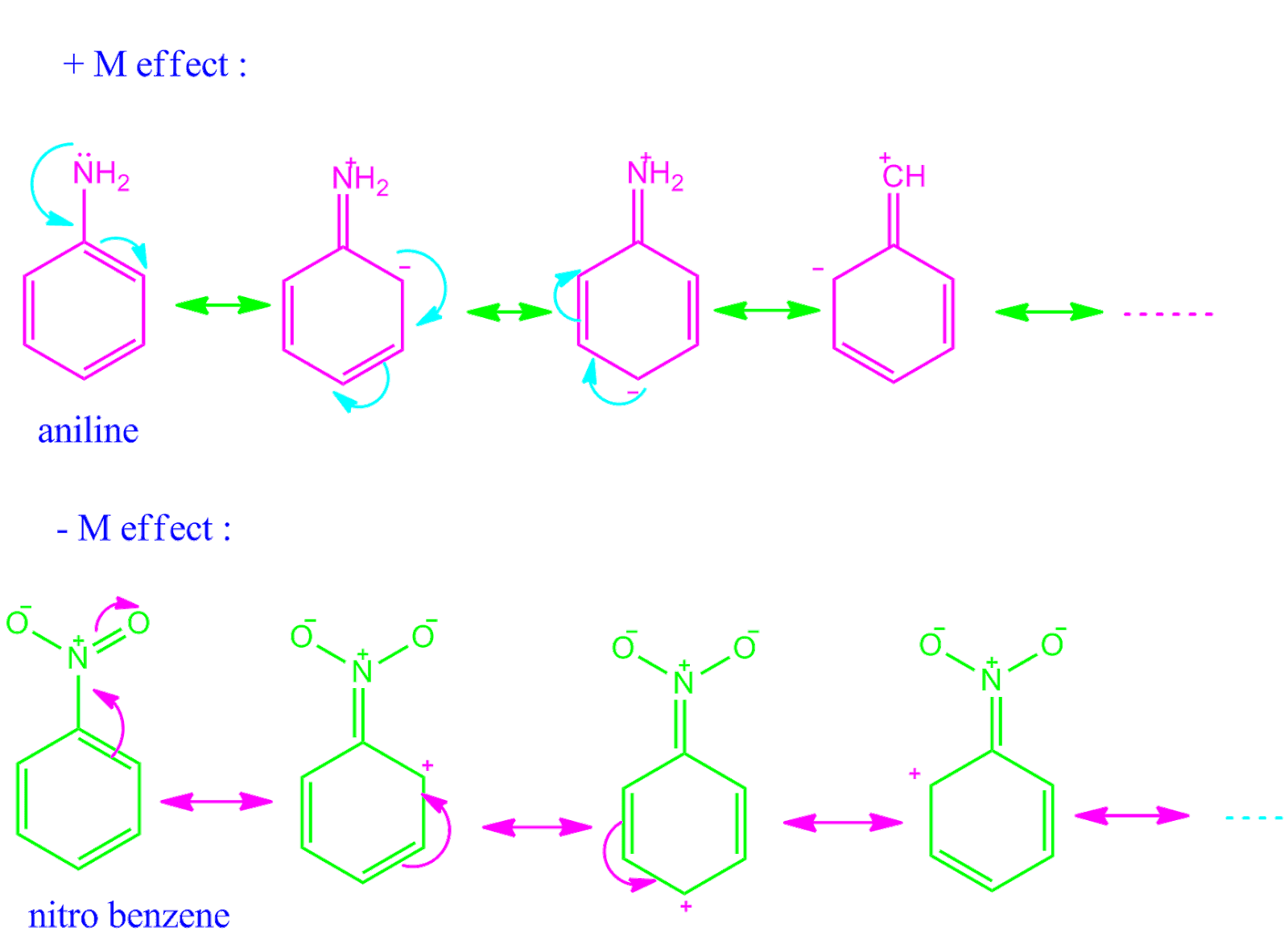
The flow of electrons from one part of a conjugated ? system to the other caused by phenomenon of resonance is called resonance effect or mesomeric effect. -M |
|
Block – 2: Organic reaction mechanism
?To know about electronic effect and its types. ?inductive effects. ?Classify a group into +I or –I group. ?Understand mesomeric or resonance effect. ? |
|
Davydov Splitting Resonance Effect and Phonon Dynamics in CVD
When excitation laser energy is very close to the allowed excitonic energy state the resonance effect occurs and influences the Raman scattering intensity of |
|
Factors affecting vibrational frequencies and IR Spectroscopy of
4. The Shape of Absorption Bands. 5. Factors Influencing Vibrational Frequencies. 5.1 Effect of Bond Order. 5.2 Resonance and Inductive Electronic Effect. |
|
Resonance Effect for Neural Spike Time Reliability
of neurons to a coherent input (Gerstner et al. 1996). However in many situations |
|
Mesomeric effect - LS College Muzaffarpur
11 mar 2021 · The mesomeric effect in chemistry is a property of substituents or functional groups in a chemical compound It is defined as the polarity |
|
Mesomeric effect & its applicationpdf
Mesomeric effect and the side of the Conjugated system is effect It is a mesomenic ermament effect and extends in the molecule |
|
(PDF) Resonance and the Mesomeric Effect - ResearchGate
9 oct 2022 · PDF Resonance Effect: The polarity induced in a molecule by the interaction of a lone pair of electrons with a pi bond or the interaction |
|
BSc Chemistry - e-PG Pathshala
MODULE 1: Revisit to Electronic Effects: Inductive and Mesomeric Effects Mesomeric Effects TABLE OF CONTENT 1 Learning outcomes 2 Introduction |
|
Questions and Answers – Mesomeric Effect - CUTM Courseware
The mesomeric effect is a permanent effect and operates in compounds containing at least one double bond 3 The phenomenon in which 2 or more structures |
|
1 Basic Concepts - Wiley-VCH
The first is the inductive effect and the second is the mesomeric effect For a student who knows and understands these two concepts well it will be easier |
|
Block – 2: Organic reaction mechanism - UOU
Of these electromeric effect is temporary while rest is permanent and is exhibited as the dipole moment of the molecule 1 Inductive effect: |
|
1The Group which shows +M effect is 2) -NO 3) -OH 4 - KEA
-E effect involves complete transfer of pi bond present in carboxyl group by attacking reagents CN- SO it is 2) –E EFFECT 1)Mesomeric effect |
|
NOTE ON THE INDUCTIVE AND MESOMERIC EFFECTS IN
In 1949 the present author (7) published a series of diagrams of electroilic charge distributions in moilosubstituted benzene derivatives where as in phenol |
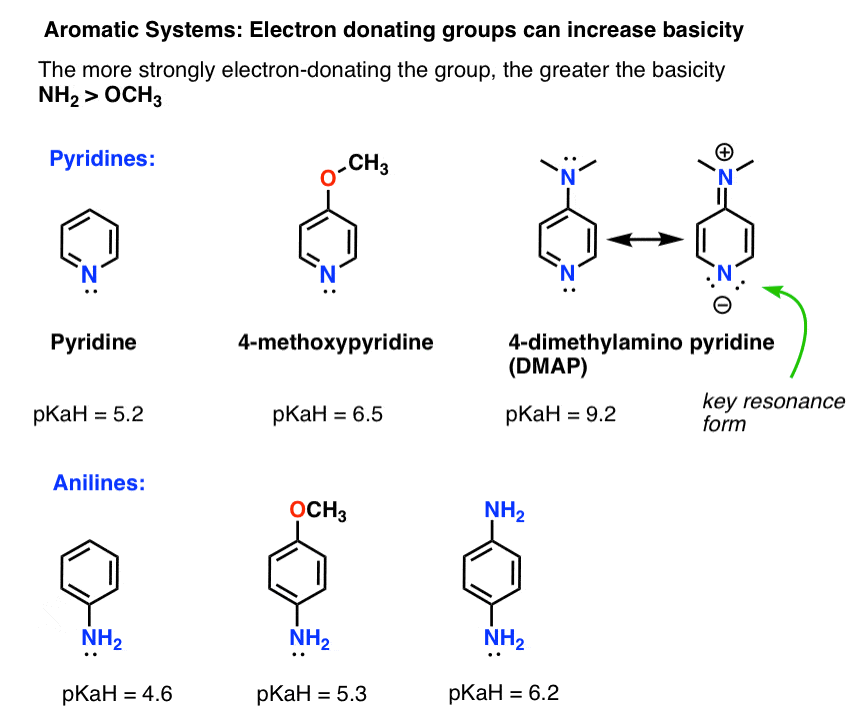
What is mesomeric effect in chemistry?
What is Mesomeric Effect? The polarity developed between atoms of a conjugated system by the electron transfer or pi–bond electron transfer is known as the Mesomeric effect.What is mesomeric effect types?
In chemistry, the mesomeric effect (or resonance effect) is a property of substituents or functional groups in a chemical compound. It is defined as the polarity produced in the molecule by the interaction of two pi bonds or between a pi bond and lone pair of electrons present on an adjacent atom.How many types of mesomeric effect are there?
If a substituent with a double bond or nonbonding electrons is directly attached to a conjugated system, the electron density and, as a result, the chemical shift will change. Further, there are two types of mesomeric effects: +M effect and -M effect according to types of mesomeric effects notes.- The main difference between resonance and the mesomeric effect is that the resonance effect describes how a molecule's lone pair of electrons and bond pair of electrons determine its chemical structure, whereas the mesomeric effect describes how a molecule's chemical structure is stabilized by using a functional group.
|
PDF :8 - BSc Chemistry
There are four types of electronic effects mainly, viz inductive effect, mesomeric ( or resonance) effect, electromeric effect and hyperconjugative effect Of these, |
|
CONCEPTS IN ORGANIC CHEMISTRY - KEA
phenomenon of resonance is called resonance effect or mesomeric effect -M or - R effect : When the electron displacement is towards the group e g :-NO 2 , |
|
General Organic Chemistry
(a) Steric inhibition of resonance (b) Ortho effect 1 4 Inductive effects: In a covalent bond between two different atoms, the electrons in the bond |
|
Block – 2: Organic reaction mechanism - UOU
There are four types of electronic effects mainly, viz inductive effect, mesomeric ( or resonance) effect, electromeric effect and hyperconjugative effect Of these |
|
Resonence or Mesomerism - Magadh University
Mesomeric effect or Resonence effect In case of conjugated system (having alternate single and double bond) the electrons can flow from one part of the system |
|
Application of resonance effect pdf - f-static
Application of resonance effect pdf Negative resonance or mesomeric effect (- M or -R): It is shown substituents or groups that excrete electrons by the |
|
Resonance and mesomeric effect pdf
structures Resonance in chemistry helps in understanding the stability of the compound along with energy states Definition - The resonant effect can be defined |
|
Inductive and mesomeric effects - Vrije Universiteit Brussel - VUB
21 sept 2016 · Concepts: inductive and mesomeric effects, aromaticity, - Substrates: carbon chains, organic rings, inorganic rings, - An excursion into |
|
Mesomeric Effect - AN College Patna
The mesomeric effect (M effect) refers to the polarity produced in a molecule as a result transmitted along a chain in a similar way as are inductive effects |