molar mass of co2 in kg/mol
|
Characterization of mixed CO2 + TBPB hydrates for refrigeration
13 déc. 2011 mCO2liq Mass of CO2 dissolved [kg] mh Hydrate mass [kg] mliq Aqueous solution mass [kg]. OH. M. 2. Molar mass of water [kg.mol-1]. |
|
Salphen-Co(III) complexes catalyzed copolymerization of epoxides
26 nov. 2019 produce polycarbonates with Mn up to 10 kg mol?1 and carbonate linkage up ... affords a high molar mass polycarbonate with Mn 25 kg.mol?1. |
|
Monomodal Ultrahigh-Molar-Mass Polycarbonate Homopolymers
21 nov. 2019 and eventually obtained at best molar mass of CO2-based polycarbonates in the range of 300 × 103 g/mol29 although ... (kg/mol). |
|
Problem Set 1
To find the mass of carbon present in the CO2 in grams we need only to multiply the moles of carbon by the molar mass of carbon. Solve. (a) 27 ×109 t CO2 ×. |
|
Moles = mass molar mass
kg molar mass g mol-1 or kg mol-1. Example. Calculate the number of moles of oxygen molecules in 4g a) 2.4 dm3 of carbon dioxide CO2 at rtp. |
|
Volumetric properties of carbon dioxide + acrylic acid binary in the
critical CO2 can solubilize low molecular weight polar molecules such as ethanol acetone |
|
CHEM1001 2006-J-4 June 2006 22/01(a) • The complete
4 juin 2006 mass = number of moles × molar mass = 51.5 × 32.00 = 1650 g = 1.65 kg. 3 mol of CO2 and 4 mol of H2O are produced for every 1 mol of propane ... |
|
Switchable Catalysis Improves the Properties of CO2-Derived
20 févr. 2020 industrial CO2 capture technologies.316. Carbon dioxide/epoxide ROCOP can also deliver high molar mass polycarbonates (Mn = 10?100 kg/mol. |
|
Industrial application of supercritial carbon dioxide soluble polymers
20 févr. 2019 Carbon capture has become a major global process carbon dioxide (CO2) can ... groups of molar masses between 2 and 3.5 kg mol-1 (Table 3 |
|
Calculations with Chemical Reactions - George Brown College
The molar mass of carbon monoxide is 28 01 g/mol and the molar mass of iron is 55 85 g/mol Fe2O3(s) + 3 CO(g) ? 2 Fe(s) + 3 CO2(g) Solution 1: Using |
|
All 13 - CHAPTER 14
Properties The molar masses of CO2 O2 and N2 are 44 0 32 0 and 28 0 kg/kmol respectively (Table A-1) The constant- volume specific heats of these gases |
|
CO2 - molar and relative molecular mass - ??? ??????
Relative molecular weight of CO2: 44 0095; Molar mass of CO2: 44 0095 g/mol (0 04401 kg/mol) ; Mr[CO2] = Ar[C] + Ar[O] * 2 = 12 0107 + 15 9994 * 2 = 44 0095 |
|
Of 2 The molecular weights of carbon and oxygen are 1201 g/mol
The molecular weights of carbon and oxygen are 12 01 g/mol and 16 00 g/mol respectively What is the specific volume in m3/kg of carbon dioxide (CO2) if |
|
Mole & Molar Mass - Montgomery College
12 00 g C-12 = 1 mol C-12 atoms = 6 022 × 1023 atoms • The number of particles in 1 mole is called Avogadro's Number (6 0221421 x 1023) 6 022 ×10 23 atoms 1 |
|
NCERT Textu al Exercise (Solved) - Studymate
(ii) Molecular mass of CO2 = 12 01 amu + 2 ×16 00 amu = 44 01 amu (iii) Molecular mass of Ans Molar mass of methanol (CH3OH) = 32 g/mol = 0 032 kg/mol |
|
Calculating the Molar Volume of Carbon Dioxide
= m2 – m1 + ma = 82 237 g – 82 151 g + 0 198 g = 0 284 g (Eq 4) (5) Calculate the number of moles of CO2 in the sample n using Equation 5 n = (m |
|
1642 Model: The carbon dioxide in the cube is an ideal gas Solve
Model: The carbon dioxide in the cube is an ideal gas Solve: Using the ideal gas equation and The molar mass of CO2 is 44 g/mol or 0 044 kg/mol Thus |
|
103: Molar Mass - Chemistry LibreTexts
4 août 2022 · The molecular mass of carbon dioxide is 44 01amu The molar mass of any compound is the mass in grams of one mole of that compound One mole of |
|
Molar mass - Cableizer
Molar mass is a physical property defined as the mass of a given substance divided by the amount of substance The base SI unit for molar mass is kg/mol |
What is the molar mass of carbon dioxide in kg mol?
Explanation: The molar mass of CO2 is 0.044 kg/mol.How many moles of CO2 are in 1 kg of CO2?
That means that 1000 g (1 kg) is 23 moles of CO2.What is the mass of carbon in 1 kg of CO2?
Carbon has an atomic mass of 12 and oxygen has an atomic mass of 16. Therefore CO2 has an atomic mass of 44. This means that one kilogram (kg) of carbon will produce = 3.67 kg of CO2. When people discuss carbon flowing through the carbon cycle this difference creates some confusion.- The molecular mass of carbon dioxide CO2 is 44 g/mole.
|
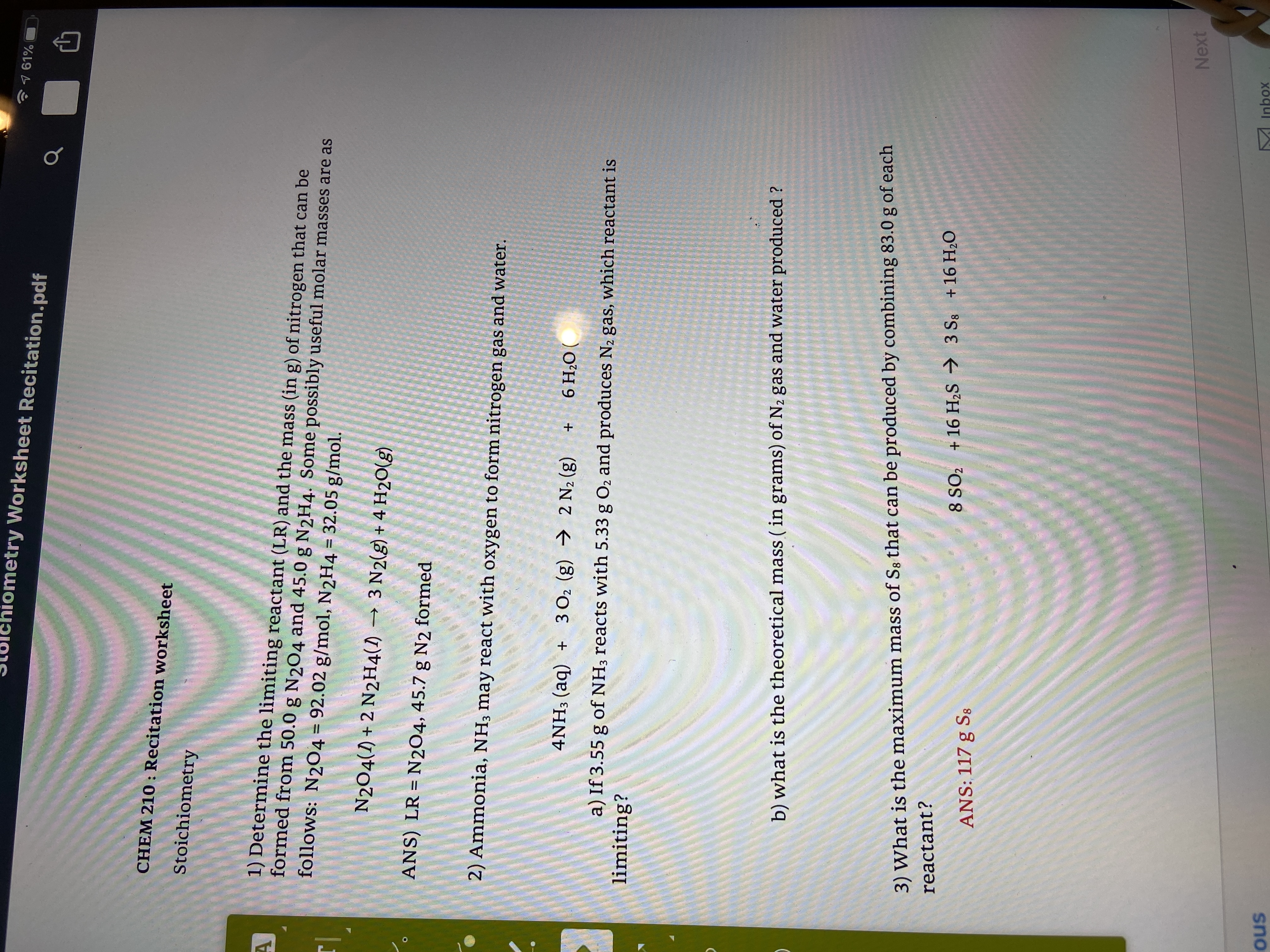
Problem Set 1
For this we need the molar masses of charcoal (carbon, 12 011 g/mol), propane (C3H8, 44 097 g/mol), and carbon dioxide (CO2, 44 01 g/mol) |
|
How much volume does 1 kg of CO2 occupy at room temperature
CO2 has a molecular weight of 44 g/mol 1 kg CO2 = 1000 g × (1 mol/44 g) = 22 7 mol CO2 V=nRT/P, V=(22 7)(0 0821)(300)/1 = 559 L CO2 at 27°C (300K), 1 atm This is a little more than half a cubic meter approximately equal to the volume of two bathtubs or the trunk of a large car |
|
Calculating the Molar Volume of Carbon Dioxide
Determine the molar volume of carbon dioxide by measuring the mass and vol- (5) Calculate the number of moles of CO2 in the sample, n, using Equation 5 |
|
Moles = mass molar mass - Knockhardy
kg molar mass g mol-1 or kg mol-1 Example Calculate the number of moles of oxygen molecules in 4g a) 2 4 dm3 of carbon dioxide, CO2 at rtp b) 120 cm3 of |
|
CHAPTER 5 GASES - ResearchGate
calculating the moles of CO2, we can use this fact as a conversion factor to convert Remember, M must be in units of kg/mol 2 N g 1 kg 2(14 01 g/mol) 28 02 |
|
Mole & Molar Mass - Montgomery College
atoms of Zn (in kg) (c) 2 33 x 10 20 atoms of oxygen (in ng) 4 How many molecules are in 2 50 moles of CO2? 5 Calculate the mass , in grams, of 0 433 mol of |
|
Concentrations and Other Units of Measure Unit conversion
CO2: MW = 1x12 + 2x16 = 12 + 32 = 44 grams per mole Mass in grams = Molecular weight x Number of moles where Density = 997 g/L (Think: 1 kg per liter) |
|
NCERT Solutions for Chapter 1 Some Basic Concept - Studymate
(ii) Molecular mass of CO2 = 12 01 amu + 2 ×16 00 amu = 44 01 amu (iii) Molecular mass of Ans Molar mass of methanol (CH3OH) = 32 g/mol = 0 032 kg/mol |
|
Calculations with Chemical Reactions - George Brown College
Molar mass– the mass of 1 mole of a substance (molecule or atom) 2 Fe(s) + 3 CO2(g) Solution 1: Using the Step-by-Step Proportion Technique uqn: mass |
|
(Solutions to Chapter 5 problems) Chem151 [Kua] 58 The question
5 30 The ideal gas equation, PV = nRT, can be used to calculate moles using P-V-T data we can determine the average molecular speed for both bulbs Cl2: urms = 1- 3- -1 -1 mol kg p(CO2) = (3 515 x 10-4)(1 atm) = 3 515 x 10-4 atm; |













![WS - Molar Mass - [PDF Document] WS - Molar Mass - [PDF Document]](https://docplayer.net/docs-images/46/21528553/images/page_4.jpg)

:max_bytes(150000):strip_icc()/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)













/sucrose-molecule-488635475-58c071f63df78c353cc53625.jpg)