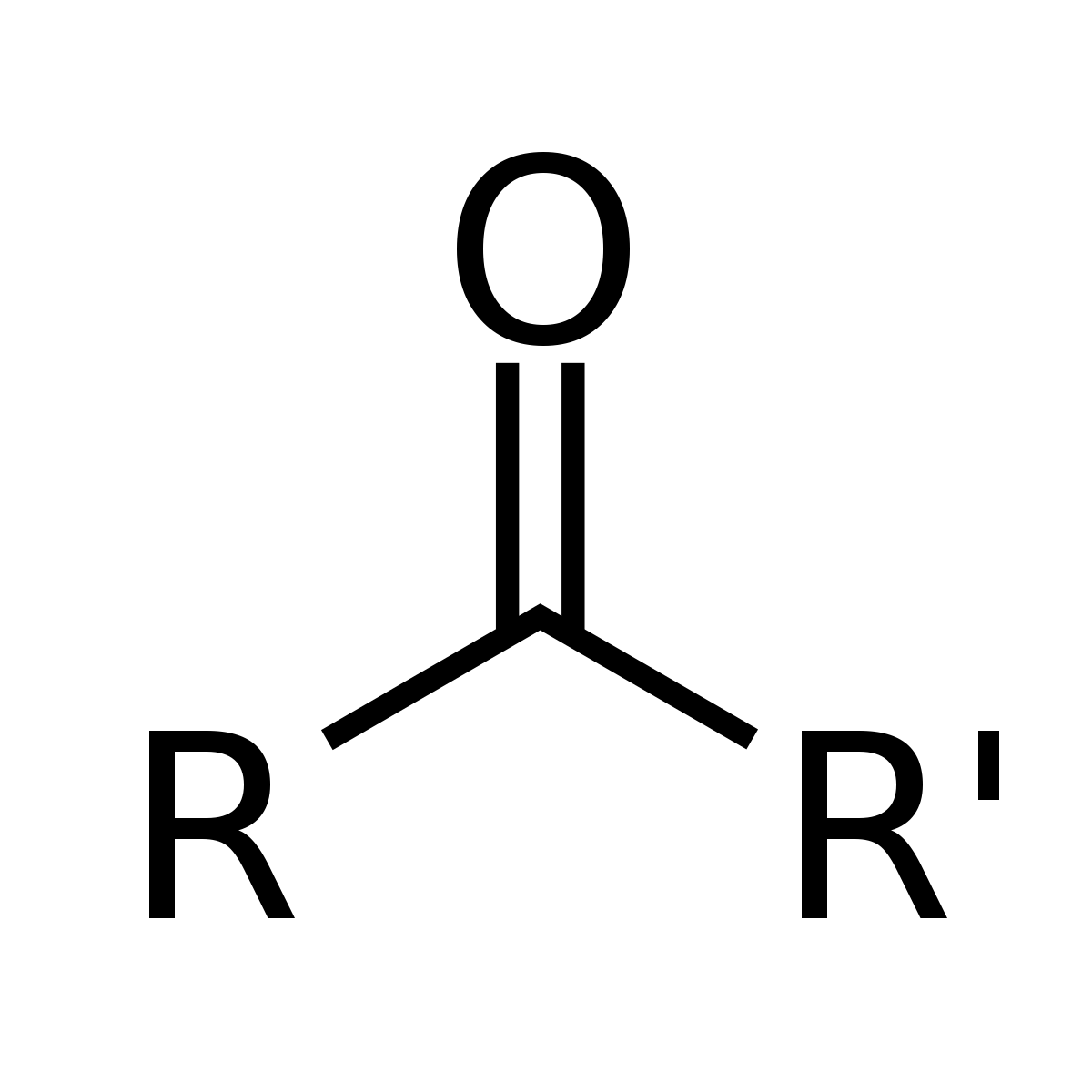
name a few tests for aldehyde and ketones
|
Testsforfunctionalgroups - inorganiccompounds
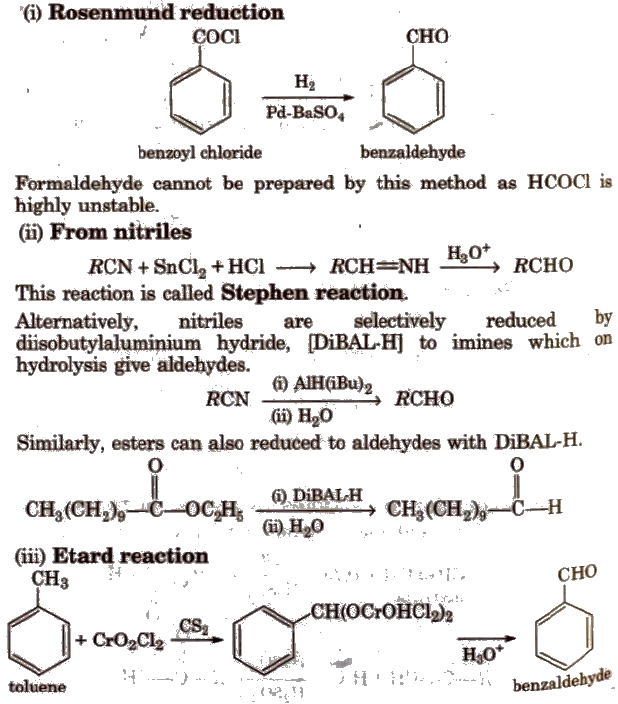
Reaction takes place as follows : Both the above reactions are used as tests for unsaturation. These two carbonyl compounds (aldehydes and ketones) are. |
|
Chemical tests to distinguish carbonyl compounds
You should recall how to name aldehydes and ketones: A few drops of the carbonyl compound are put in a test tube with about 5cm3 of Brady's reagent. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
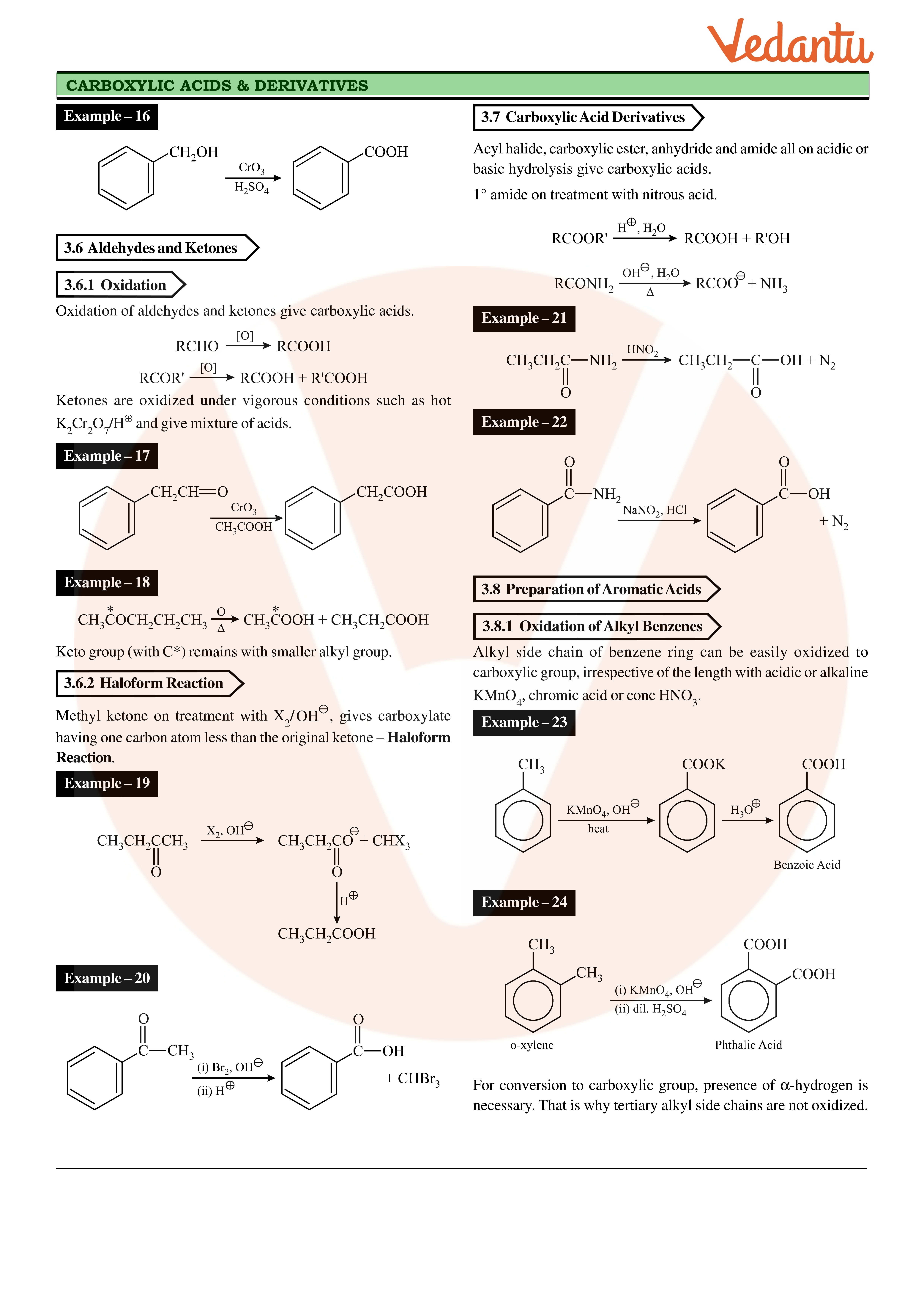
Table 12.1: Common and IUPAC Names of Some Aldehydes and Ketones. Structure (ii) Fehling's test: Fehling reagent comprises of two solutions. |
|
Experiment 13 Qualitative Tests for Carbonyls; Unknown Carbonyl
Part I Chemical Tests. Test 1: 24-Dinitrophenylhydrazone Test. Most aldehydes and ketones will react with dinitrophenylhydrazine within a few. |
|
Shaken not stirred: a schools test for aldehydes and ketones
Test for ketones with a minimum quantity of 50 mg of PhNHNH3Cl/NaOAc . 3H2O in water (8 mL) at RT for a few seconds. Entry. Name. Structure. Test 1. |
|
Identifying an Unknown Compound by Solubility Functional Group
test solution with pH paper. Water. Small polar organic compounds such as alcohols |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
Table 12.1: Common and IUPAC Names of Some Aldehydes and Ketones. Structure. Common name (i) Tollens' test: On warming an aldehyde with freshly prepared. |
|
Lab 14: Qualitative Organic Analysis
ketones. Tollen's test. C-23. Tests for the presence of aldehydes. Amides. Alkaline hydrolysis narrow down the list to only a few candidates. |
|
Chemical tests to distinguish carbonyl compounds - WordPresscom
1: Detecting an aldehyde or ketone Aldehydes and ketones react with 24-dinitrophenylhydrazine (24-DNP or 24-DNPH) to form an orange or yellow precipitate |
|
Tests for Aldehydes and Ketones - Sodium Bisulphite(NaHSO3) 24
9 oct 2019 · Tests for Aldehydes and Ketones - In this experiment you will explore how to confirm the presence of aldehydes and ketones functional group |
|
Tests for Aldehydic and Ketonic Groups - PraxiLabs
Classify organic compounds containing carbonyl groups into aldehydes and ketones Compare between aldehydes and ketones in terms of chemical structures |
|
Identification of an Unknown – Alcohols Aldehydes and Ketones
The results of these classification tests will allow the unknown to be classified as an alcohol an aldehyde or a ketone Additional structural information can |
|
INORGANICCOMPOUNDS - NCERT
Reaction takes place as follows : Both the above reactions are used as tests for unsaturation These two carbonyl compounds (aldehydes and ketones) are |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids - NCERT
Table 12 1: Common and IUPAC Names of Some Aldehydes and Ketones Structure (ii) Fehling's test: Fehling reagent comprises of two solutions |
|
Test for Aldehydes and Ketones - Vedantu
Procedure and Observation: · 1 In ethanol dissolve the given organic compound · 2 Add the 24-dinitrophenyl hydrazine alcoholic reagent to this solution · 3 |
|
Identification of Aldehydes and Ketones
If no reaction occurs warm the test tube in water bath for few minutes (note that excessive heating will cause the appearance of a false positive test by |
|
12 ALDEHYDES AND KETONES - eLearnPunjab
Tests for the distinction between aldehydes and ketones The IUPAC names of aldehydes are derived from the names of alkanes |
|
The Carbonyl Group Nomenclature of Aldehydes and Ketones
(The carbon to which the aldehyde group is attached is carbon “1”) 10 Examples: Naming Aldehydes and Ketones • Name the following compounds: O |
What are the tests of aldehydes and ketones?
Tollens Test
The Tollens' test is a reaction that is used to distinguish aldehydes from ketones, as aldehydes are able to be oxidized into a carboxylic acid while ketones cannot. Tollens' reagent, which is a mixture of silver nitrate and ammonia, oxidizes the aldehyde to a carboxylic acid.What are the three tests to differentiate between aldehydes and ketones?
The main difference between aldehydes and ketones is their chemical structure. Some tests which help them distinct from each other are Schiff's test, Tollen's test, Fehling's test, Sodium hydroxide test, etc.What are 2 tests performed in this experiment that is used to distinguish aldehyde from ketone?
Most ketones, however, are not oxidized by Tollens' reagent, so no solid silver will form. In this lab, you will identify several unknown aldehydes and ketones by performing the DNPH test, the Tollens test, and the iodoform test.- Test for aldehydes
Fehling's solution (a mixture of solutions of copper sulphate and sodium hydroxide) is a blue solution which when gently heated with an aldehyde forms a red precipitate of copper(I) oxide. Ketones do not undergo a visible change.
|
Identification of an Unknown – Alcohols, Aldehydes, and Ketones
Iodoform Reaction: The iodoform test indicates the presence of an aldehyde or ketone in which one of the groups directly attached to the carbonyl carbon is a methyl group Such a ketone is called a methyl ketone In the iodoform test, the unknown is allowed to react with a mixture of excess iodine and excess hydroxide |
|
Chemical tests to distinguish carbonyl compounds - WordPresscom
You should recall how to name aldehydes and ketones: - longest chain A few drops of the carbonyl compound are put in a test tube with about 5cm3 of Brady's |
|
Aldehydes and Ketones
Aldehydes and ketones both contain the carbonyl functional group, which imparts ammonia solution used in the Tollens test or Cu2+ in alkaline solution used in few more drops of NH4OH solution, if needed, until the precipitate completely dissolves Name one aldehyde or ketone that is used widely by consumers |
|
Iodoform test of aldehydes and ketones - Squarespace
The side effects of some ketones are oiled, which will not be Tollen test for Aldehydes Aldehyde standards Cyclohexanone and Benzaldehyde on the shelf and label them with four compound names, or codes, and the name of the first test |
|
Identification of Aldehydes and Ketones
aldehydes and ketones contain the carbonyl group, C=O that largely If no reaction occurs, warm the test tube in water bath for few minutes (note that |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
Table 12 1: Common and IUPAC Names of Some Aldehydes and Ketones Structure Common (i) Tollens' test: On warming an aldehyde with freshly prepared |
|
7-Aldehydes, Ketones, C Acids
Aldehydes and ketones are molecules that contain a carbonyl group, which is an oxygen atom with If the test solution turns green when chromic acid is added use a few crystals ) Shake Name each of the following compounds a b c d |
|
Carbonyl Unknowns
2,4-dinitrophenylhydrazine test: positive for aldehydes or ketones • Schiff's is illustrative of some of the reactions that we learn in class It is useful Name: Lab Report Requirements: No procedure or yield information required Fill out the |
|
Experiment 6 Reactions of Carbonyl Compounds: Qualitative
laboratory tests that distinguish aldehydes from ketones are based on the fact that the Remove any silver mirror deposits with a few drops of 8M nitric acid, The Purpald® reagent (the name of the reagent is a registered trademark of the |
|
A schools test for aldehydes and ketones M John Plater - CORE
A schools test for aldehydes and ketones in water at rt using test tubes has been 3H2O reacts satisfactorily with both aldehydes and ketones at rt within a few Name Structure Test 1 PhNHNH3Cl (rt in water) Test 2 PhNHNH3Cl/ NaOAc |