name three colligative properties of solutions
|
16.3 Colligative Properties of Solutions
In this section you will discover how a solute can change the freezing point of a solution. Guide for Reading. Key Concepts. • What are three colligative. |
|
SECTION 16.1 PROPERTIES OF SOLUTIONS (pages 471–477)
17-Feb-2012 Name three factors that influence the rate at which a solute dissolves in a solvent. ... SECTION 16.3 COLLIGATIVE PROPERTIES OF. SOLUTIONS ... |
|
1) Find the molarity of all ions in a solution that contains 0.165 moles
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES 3) Find the molarity of each ion present after mixing 35 ml of 0.42 M K?SO4 with 27 ml of 0.17 M K3PO4. |
|
All Bottled up: The Perfect Ecosystem
They also learn about three colligative properties of solutions. Introduction. 1. Before the introductory activity make a “temperature-. |
|
Concentration of Solutions and Molarity
To calculate the molarity of a solution divide the moles of solute by the volume of the Three important colligative properties of solutions are. |
|
2nd Semester Chem FINAL EXAM GUIDELINE & REVIEW
3-5 Write the formula for each of the following: a. sulfur trioxide ____SO3___ Name the three colligative properties of solutions: _vapor. |
|
Solutions
Identify Name three colligative properties of solutions. 29. Explain Why does a solution have a lower vapor pressure than the pure solvent of that solution? |
|
Bound Water in Plant Sap and Some Effects of Temperature and
of these three colligative properties are interrelated through the applica- bility of the gas laws to solutions. Few if any |
|
The Science of a Sundae: Using the Principle of Colligative
students will be able to (1) state what colligative properties are and give examples (2) explain what happens as a concentrated sugar and water solution is |
|
Chemistry Colligative Properties Of Solutions Section Review
Colligative Properties Of Solutions Section Review and numerous book collections from exam STEP 3 Develop the strategies that will give you the edge on. |
|
Solutions and Colligative Properties - Saptarshi Classes
Concentration of solutions: It is defined as the amount of solute dissolved in specific amount of solvent Dilute solutions: The solutions containing relatively |
|
Colligative Properties - Definition Types Examples Raoults Law
The four colligative properties that can be exhibited by a solution are given below: Boiling point elevation; Freezing point depression; Relative lowering of |
|
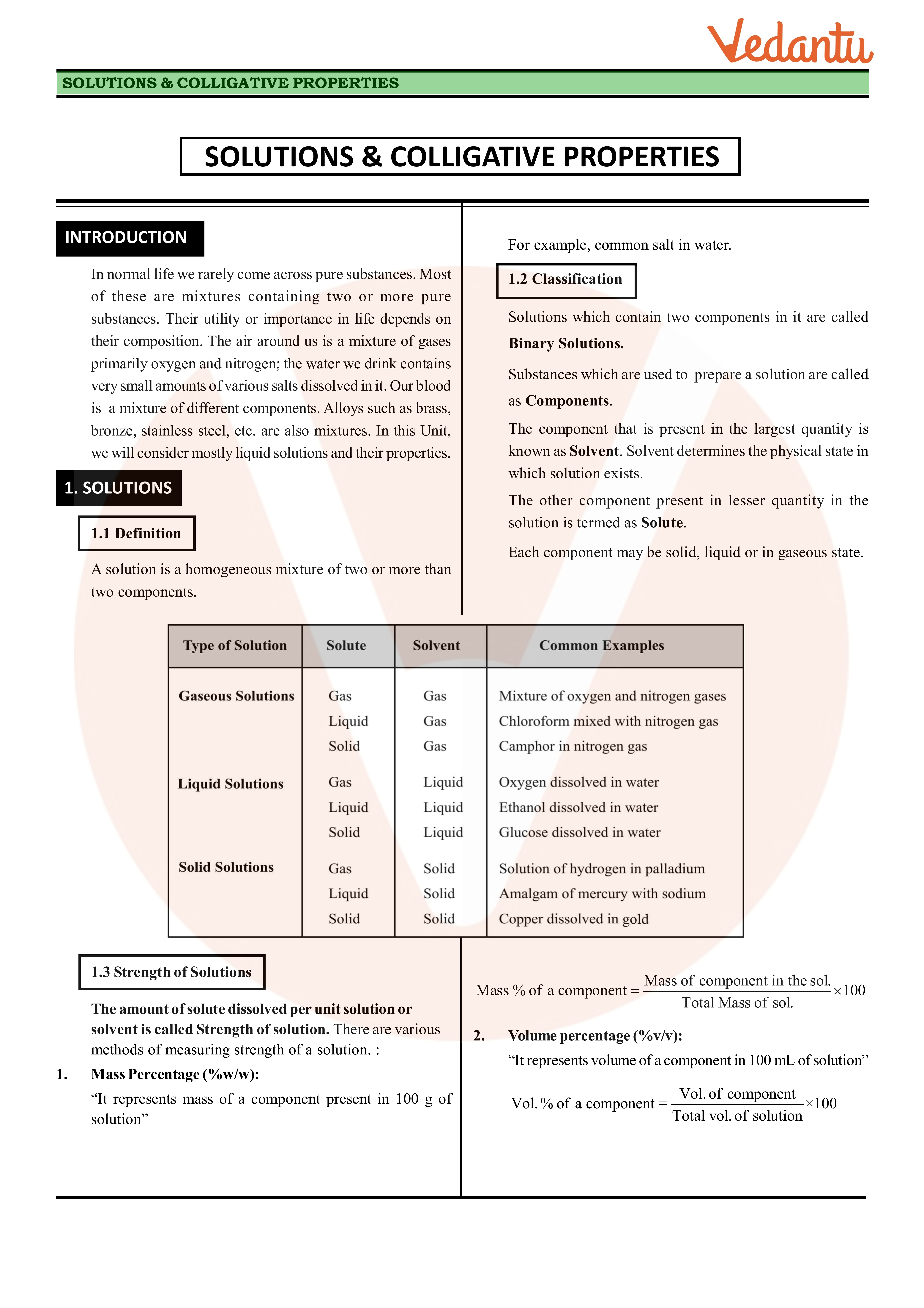
SOLUTIONS & COLLIGATIVE PROPERTIES - GGPS Bokaro
SOLUTIONS COLLIGATIVE PROPERTIES 3 Mass by volume percentage ( w/v): “It represents mass of solute in grams present in 100 mL of solution” |
|
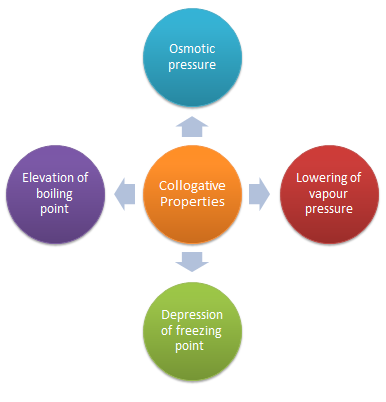
Colligative Properties
Properties of solutions that depend on the number of molecules present and not on the kind of molecules are called colligative properties |
|
Chapter 13: Physical Properties of Solutions
Colligative Properties terminology: • Solution: a homogeneous mixture • Solute: the component that is dissolved in solvent -- usually in smaller amount |
|
Leep502pdf - NCERT
takes place when small amount of 'A' is added to the solution Colligative properties depend on ______ (i) the Name and explain the phenomenon |
|
Colligative properties of solutions
Colligative properties of solutions (diffusion osmosis) are vital for the The list of key terms parameters characteristics which the student |
|
Properties of solutions
Colligative properties depend only on number of particles of the solute and do not depend on its identity Page 14 Classification Colligative properties are: |
What are 3 colligative properties of a solution?
Colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.What are the 4 colligative properties of solutions?
These colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure. This small set of properties is of central importance to many natural phenomena and technological applications, as will be described in this module.What are the names of the 4 colligative properties?
There are four colligative properties: vapor pressure lowering, boiling point ele- vation, freezing point depression, and osmotic pressure.- Examples of colligative properties include vapor pressure lowering, freezing point depression, osmotic pressure, and boiling point elevation.
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
If we consider dilute solutions then ∆P ≅ (nsolute/nsolvent)P°solvent Therefore, the lowering of the vapor pressure in a dilute solution of a non-volatile solute is |
|
SOLUTIONS - Foothill High School
17 fév 2012 · SECTION 16 1 PROPERTIES OF SOLUTIONS (pages 471–477) This section Name three factors that influence the rate at which a solute dissolves in a solvent a b c 3 SECTION 16 3 COLLIGATIVE PROPERTIES OF |
|
SOLUTIONS
Give examples of three colligative properties 2 How many particles in solution are produced by each formula unit of potassium carbonate, K2CO3? 3 How may |
|
Unit 2pmd - NCERT
(iii) the physical properties of the solute particles dissolved in solution (iv) the nature of solvent We have three aqueous solutions of NaCl labelled as 'A', 'B' and 'C' with Relative lowering of vapour pressure is a colligative property because Give an example of a material used for making semipermeable membrane for |
|
Solutions - NCERT
studying the properties of the solutions, like vapour pressure and colligative properties We will begin with Henry was the first to give a quantitative relation |
|
Chapter 15: Solutions
15 1 What are solutions? 453 There are three common ways to increase the collisions Colligative properties include vapor pressure lowering, boiling point |
|
?mIL= 259*1402%45=0
Chapters 13 &14 – Solutions, Net Ionic Equations, and Colligative Properties Vocabulary Understand and be able to give examples of the Tyndall effect Be able to describe Three ways to increase the rate of dissolving are: 1 2 3 |