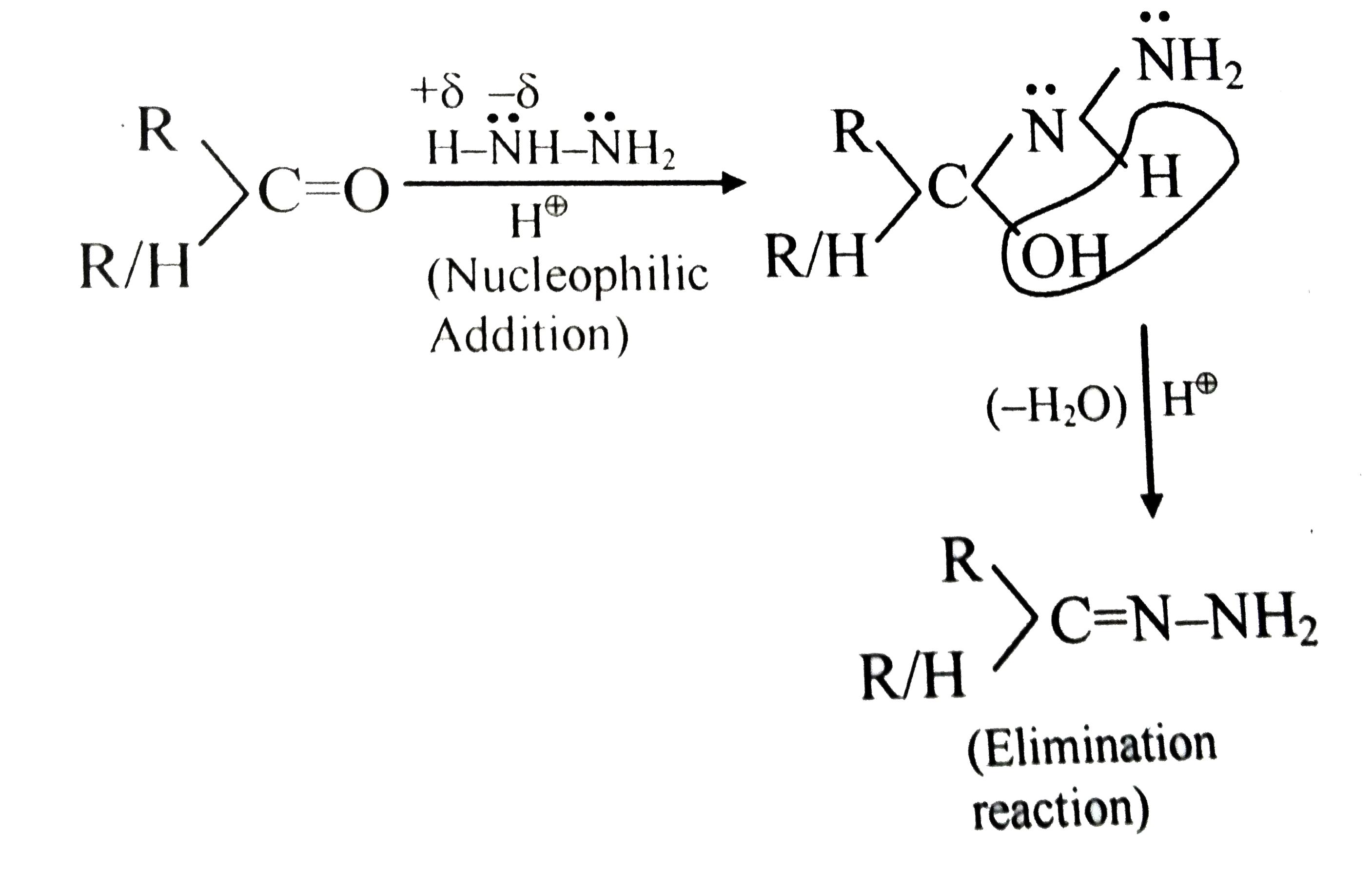
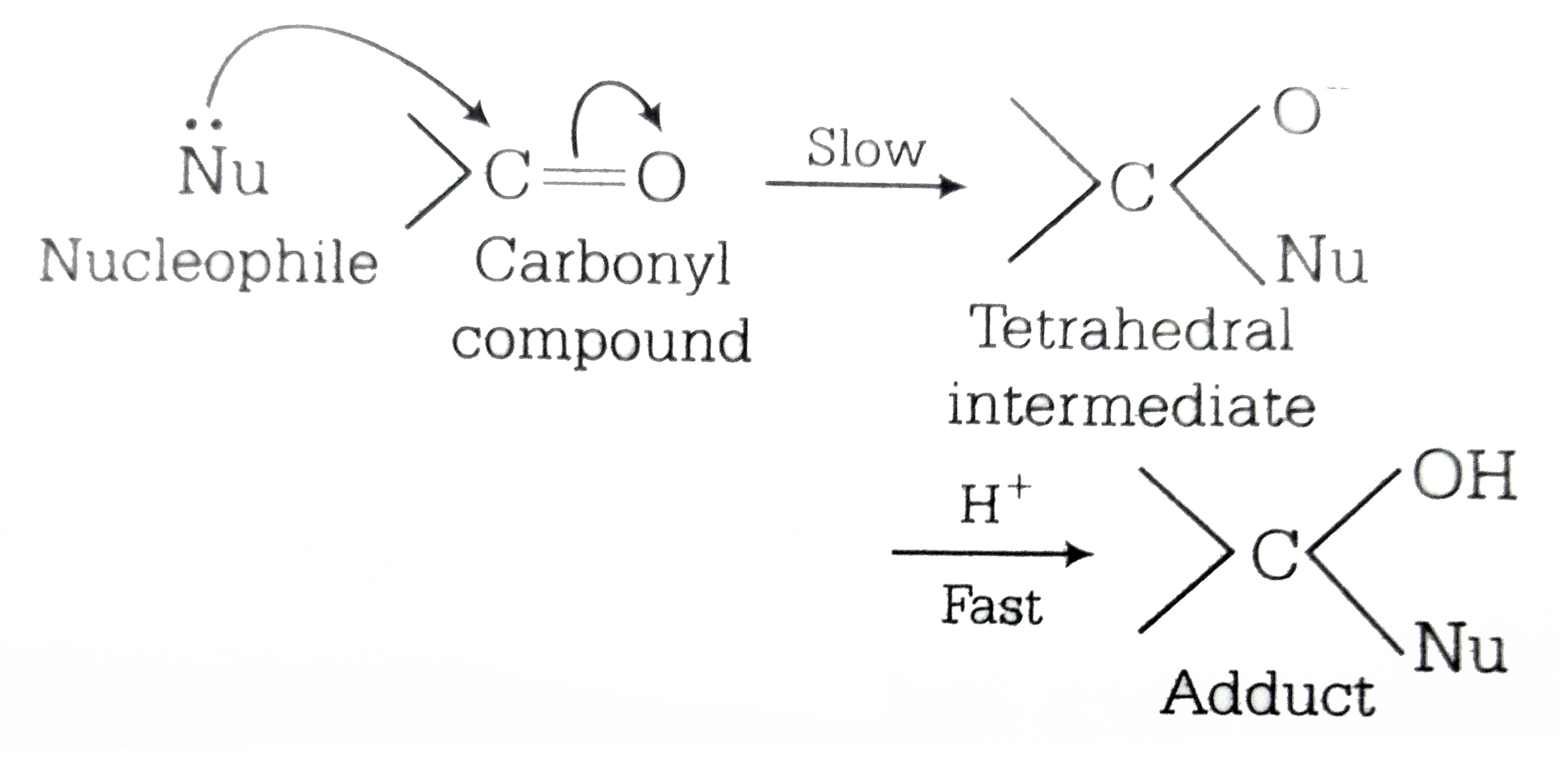
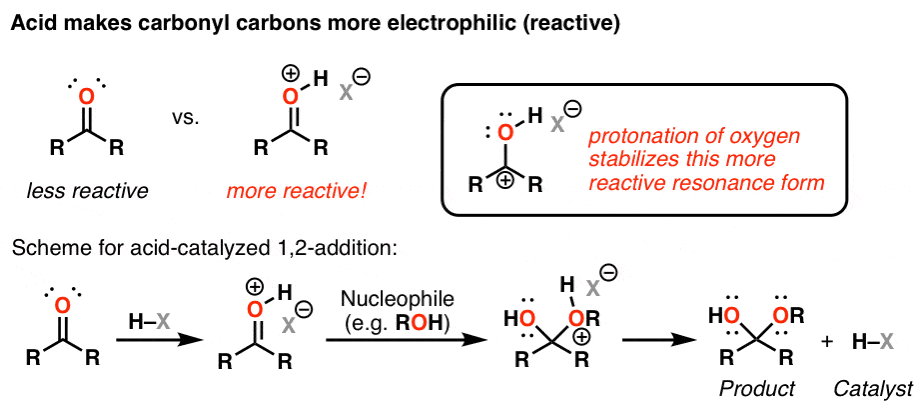
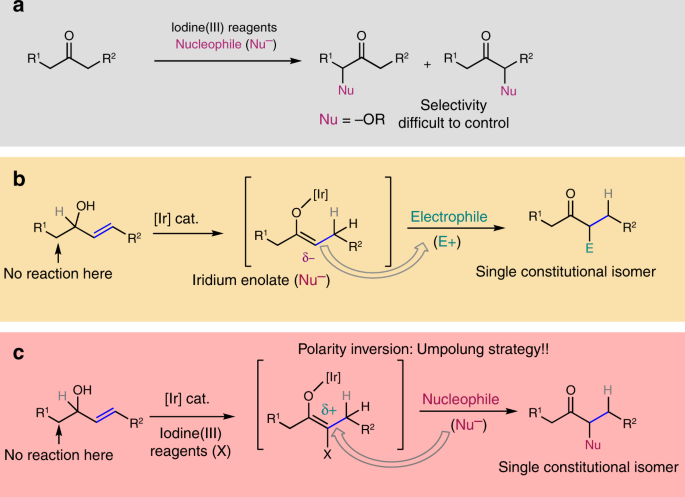
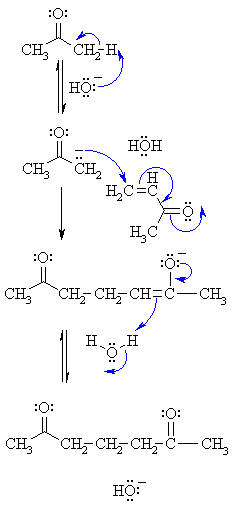
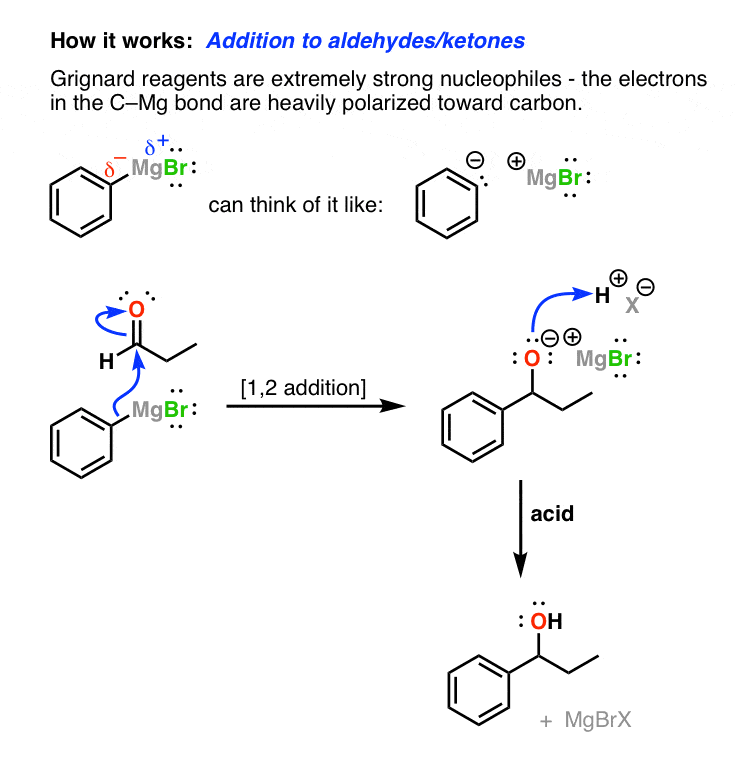
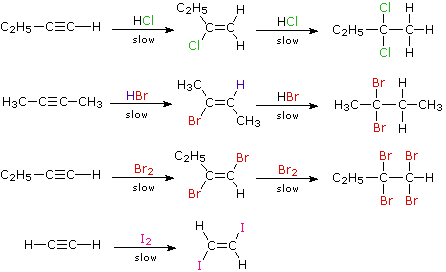
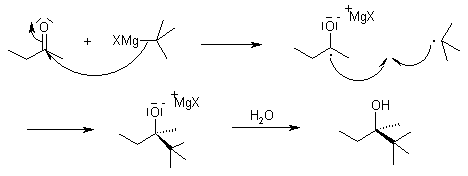nucleophilic addition reagents
|
Unusual Nucleophilic Addition of Grignard Reagents in the
10 août 2018 Herein we describe a reaction pathway in a Grignard reagent-based synthesis of substituted pyrimidines. A general synthesis of ?-keto-2-methyl- ... |
|
Unusual Nucleophilic Addition of Grignard Reagents in the
10 août 2018 Herein we describe a reaction pathway in a Grignard reagent-based synthesis of substituted pyrimidines. A general synthesis of ?-keto-2-methyl- ... |
|
Nucleophilic Addition of Organometallic Reagents to Cinchona
30 mai 2007 Nucleophilic Addition of Organometallic Reagents to Cinchona. Alkaloids: Simple Access to Diverse Architectures**. |
|
Stereoselectivity of the nucleophilic addition of organocopper
of the Nucleophilic Addition of Organocopper. Reagents to Chiral /3-Unsaturated Carbonyl Compounds. Ab. Initio Molecular Orbital Studies of Steric and |
|
Stereoselectivity of the nucleophilic addition of organocopper
of the Nucleophilic Addition of Organocopper. Reagents to Chiral /3-Unsaturated Carbonyl Compounds. Ab. Initio Molecular Orbital Studies of Steric and |
|
COMMUNICATION Unprecedented Nucleophilic Additions of Highly
research.[6] Building on our recent findings on the nucleophilic addition of Grignard and organolithium reagents to carbonyl. |
|
Nucleophilic addition of organochromium reagents to carbonyl
2) The development of the allylic chromium reagent (Nozaki-Hiyama reaction). 3) Nickel-catalyzed addition of alkenyl- aryl- |
|
Synthesis of Primary Amines v/a Nucleophilic Addition of
lithium reagents or LiBH4 to afford a wide variety of primary amines in good to nucleophilic additions of organometallic reagents to resin-immobilized ... |
|
Enantioselective Cu-Catalyzed Nucleophilic Addition of Fluorinated
17 fév. 2022 Enantioselective Cu-Catalyzed Nucleophilic Addition of Fluorinated. Reagents: C?C Bond Formation for the Synthesis of Chiral Vicinal. |
|
Oxidative nucleophilic addition of organovanadium reagents to
via carbonyl addition reactions constitutes one of the important vanadium compound as a key reagent for oxidative nucleophilic addition. |
|
16: Addition and Substitution Reactions of Carbonyl Compounds
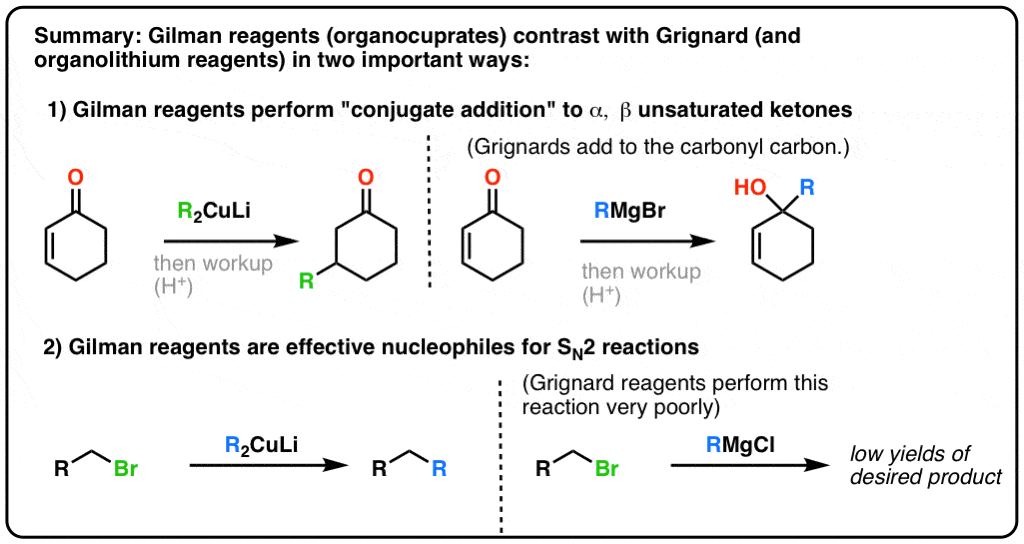
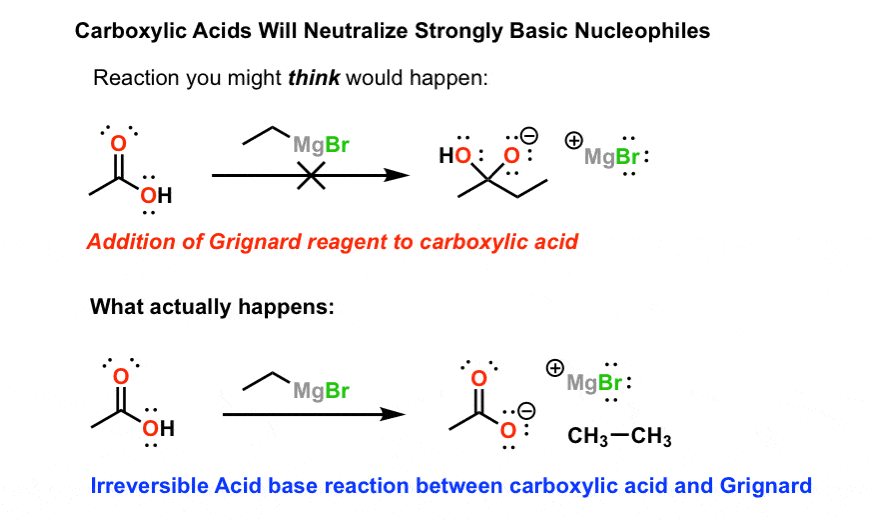
Nucleophilic addition of hydroxide ion followed by elimination of Z occurs with all compounds of the structure R-C(=O)-Z except carboxylic acids (Z = OH) Carboxylic acids rapidly react with HO-to form negatively charged carboxylate ions that are unreactive to C=O addition of nucleophiles such as HO- For example the product that would |
|
107: Nucleophilic Addition Reactions - Chemistry LibreTexts
Nucleophilic addition and abstraction • In the case of reductive elimination and migratory insertion we have seen how reaction can occur between two ligands of the coordination sphere therefore causing reaction between two organicfragmentsresulting inanindependentorganicproductonce eliminated |
|
Lecture 4: Nucleophilic Addition Reactions of Aldehydes and
Organolithium Reagents and Grignard Reagents The addition of carbon nucleophiles into aldehydes and ketones provides one of the mosteffective ways of forming new C?C bonds as well as alcohols Many types of carbonnucleophiles have been developed for this purpose |
|
Section J – Aldehydes and ketones J3 UCLEOPHILIC ADDITION
Charged nucleophiles undergo nucleophilic addition with an aldehyde or ketone to give a charged intermediate which has to be treated with acid to give the ?nal product Neutral nucleophiles require acid catalysis and fur-ther reactions can take place after nucleophilic addition Related topics Nucleophilic addition – charged nucleophiles (J4) |
|
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS GRIGNARD REACTION Carbonyl-containing compounds such as aldehydes ketones carboxylic acid and acid anhydrides possess an electophilic carbon center which can be attacked by various nucleophiles (Figure 1) |
|
Searches related to nucleophilic addition reagents filetype:pdf
Mechanistic study of the nucleophilic addition reaction of diethylamine 1 with 1-cyano-2-phenyl vinyl methanesulfonate 2 Analysis of the stationary points involved in the reaction between 1-cyano-2-phenyl vinyl methanesulfonate and diethylamine in acetonitrile as solvent indicates that this nucleophilic addition |
|
Reactions of Ketones and Aldehydes Nucleophilic Addition
Reaction of Organometallic Reagents with Aldehydes and Ketones Treatment of an aldehyde or ketone with a Grignard reagent organolithium and sodium |
|
Aldehydes and Ketones: Nucleophilic Addition to the Carbonyl
A Wittig reagent is prepared from the reaction of an alkyl halide with triphenylphosphine (Ph3P:) to give a phosphonium salt The protons on the carbon adjacent |
|
Chapter 19 Aldehydes and Ketones: Nucleophilic Addition Reactions
In nucleophilic addition reactions The partially positive carbonyl carbon undergoes attack by electron-rich reagents or nucleophiles (Nu:) |
|
Chapter 19 Aldehydes and Ketones: Nucleophilic Addition Reactions
1) Aldehydes are easily oxidized ketones are not 2) Aldehydes are more reactive in nucleophilic additions than ketones Page 6 |
|
Chapter 16 Aldehydes and Ketones: Nucleophilic Addition Reactions
Nucleophilic addition of the equivalent of a carbon anion or carbanion A carbon–magnesium bond is strongly polarized so a Grignard reagent reacts for all |
|
J3 NUCLEOPHILIC ADDITION - Organic Chemistry Second Edition
Grignard reagents (RMgX) and organolithium reagents (RLi) are used as the source of carbanions The reaction mechanism involves nucleophilic addi- tion of the |
|
Chapter 19 Aldehydes and Ketones: Nucleophilic Addition Reactions
Nucleophilic Addition Reactions McMurry strongly polarized so a Grignard reagent reacts for Mechanism of Addition of Grignard Reagents |
|
Nucleophilic Addition and Substitution A variety of reagents can add
Reagents like H-OH and H-OR(alcohols) can add across the C=O bond of aldehydes and ketones The acid catalyzed mechanism is very similar to the addition to |
|
Aldehydes and Ketones: Nucleophilic Addition to C=O - LNCT
- This very common type of reaction is called a nucleophilic addition ? Relative Reactivity of Carbonyl Compounds to Nucleophilic Addition - In general |
|
UNIT 6 NUCLEOPHILIC ADDITION TO CARBONYL COMPOUNDS
The carbon carrying partial positive charge called electrophilic or cationoid centre This can be attacked by nucleophilic reagents The oxygen carrying |
What reagents are used in nucleophilic reaction of alkenes?
- The overall reaction is the addition of HX to the double bond: The HX reagent can be water, an alcohol ( ROH), a thiol ( RSH), an amine ( RNH 2), or hydrogen cyanide ( HCN) or other carbon acids (i.e., compounds with acidic C ? H bonds). However, nucleophilic addition of these reagents to simple alkenes rarely is encountered.
What is nucleophilic addition reaction?
- Nucleophilic addition reaction is a chemical reaction in which a nucleophile (electron-rich species) combines and forms a sigma bond with an electron-deficient molecule to form a new compound.
What is the best reagent for nucleophilic substitution?
- Every reagent ever in our class and what they do Terms in this set (27) NaCl H2O Good nucleophile suggests nucleophilic substitution, solvent suggests SN1 reaction.
Is carboxylic acid a nucleophile?
- NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS GRIGNARD REACTION Carbonyl-containing compounds, such as aldehydes, ketones, carboxylic acid and acid anhydrides, possess an electophilic carbon center, which can be attacked by various nucleophiles (Figure 1).
|
Lecture 4: Nucleophilic Addition Reactions of Aldehydes and Ketones
The addition of carbon nucleophiles into aldehydes and ketones provides one of the most effective ways of forming new C−C bonds, as well as alcohols Many types of carbon nucleophiles have been developed for this purpose Organolithium reagents and Grignard reagents are two of the most important and widely used |
|
Nucleophilic addition to the carbonyl group
We will frequently use a device like this, showing a reaction scheme with a mechanism for the same reaction looping round underneath The reagents and |
|
Chapter 16 Aldehydes and Ketones: Nucleophilic Addition Reactions
19 7 Nucleophilic Addition of Grignard Reagents and Hydride Reagents: Alcohol Formation ▫ Treatment of aldehydes or ketones with Grignard reagents |
|
Reactions of Ketones and Aldehydes Nucleophilic Addition
Nucleophilic Addition of Hydride Reagents: Alcohol Formation Treating an aldehyde or a ketone with NaBH 4 or LiAlH 4 , followed by water or some other |
|
Aldehydes and Ketones: Nucleophilic Addition to the Carbonyl
Addition of Grignard Reagents (Chapters 14 5-14 6) 120 17 6: Principles of Nucleophilic Addition: Hydration of Aldehydes and Ketones Water can reversibly |
|
Chapter 19 Aldehydes and Ketones: Nucleophilic Addition Reactions
19 ○ Treatment of aldehydes or ketones with Grignard reagents yields an alcohol ○ Nucleophilic addition of the equivalent of a carbon anion, or carbanion |
|
Worksheet – Nucleophilic Addition and Substitution A variety of
Reagents, like H-OH and H-OR(alcohols) can add across the C=O bond of aldehydes and ketones The acid catalyzed mechanism is very similar to the addition to |
|
16: Addition and Substitution Reactions of Carbonyl Compounds
Magnesium, Lithium and Zinc Reagents Addition Addition of Organometallic Reagents In nucleophilic acyl addition reactions, the nucleophile binds to the |