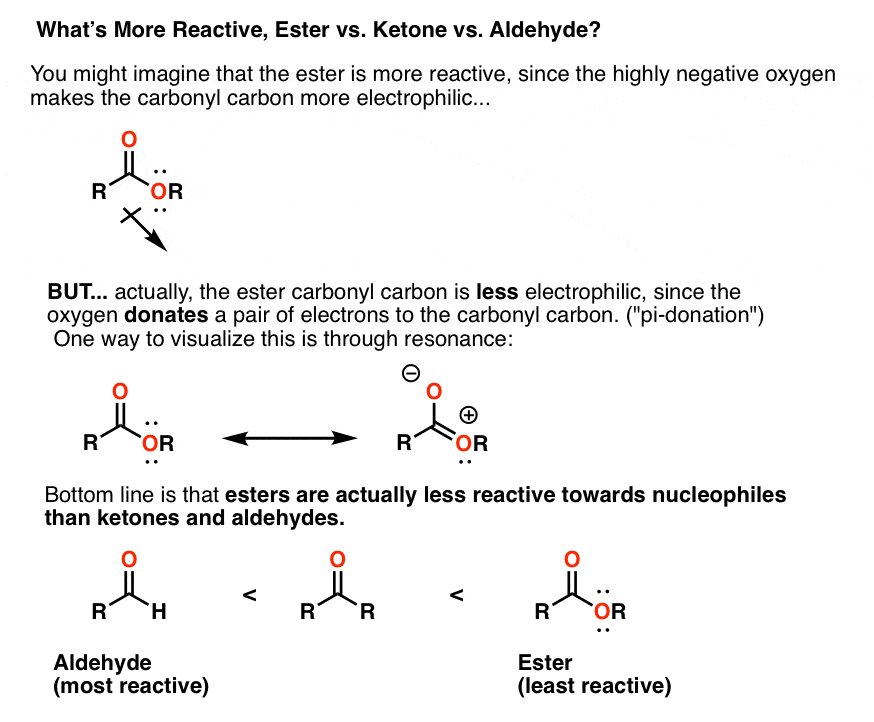
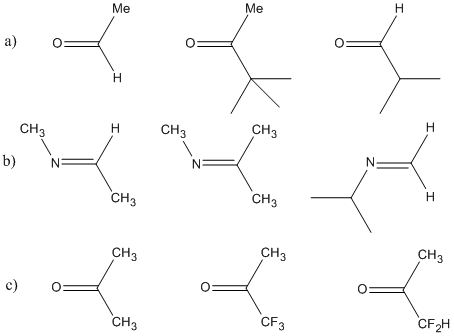
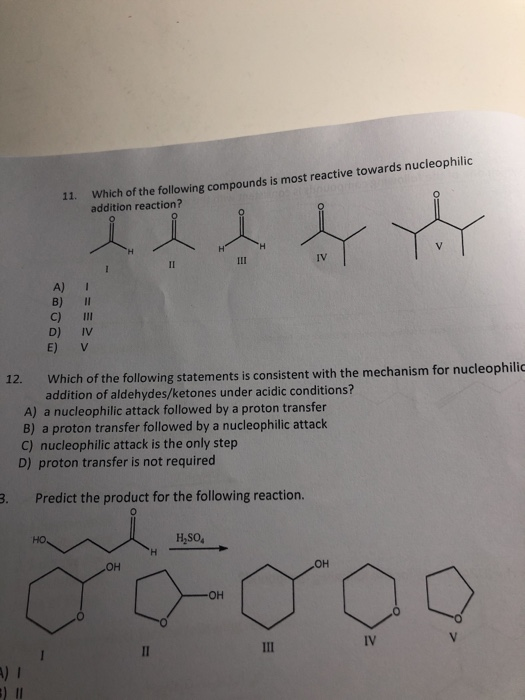
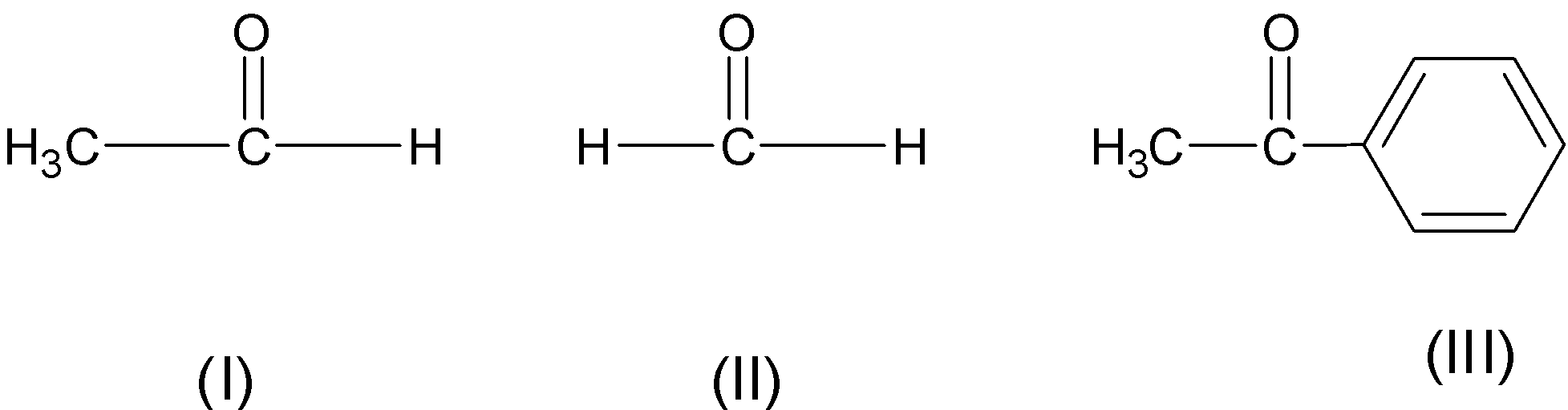
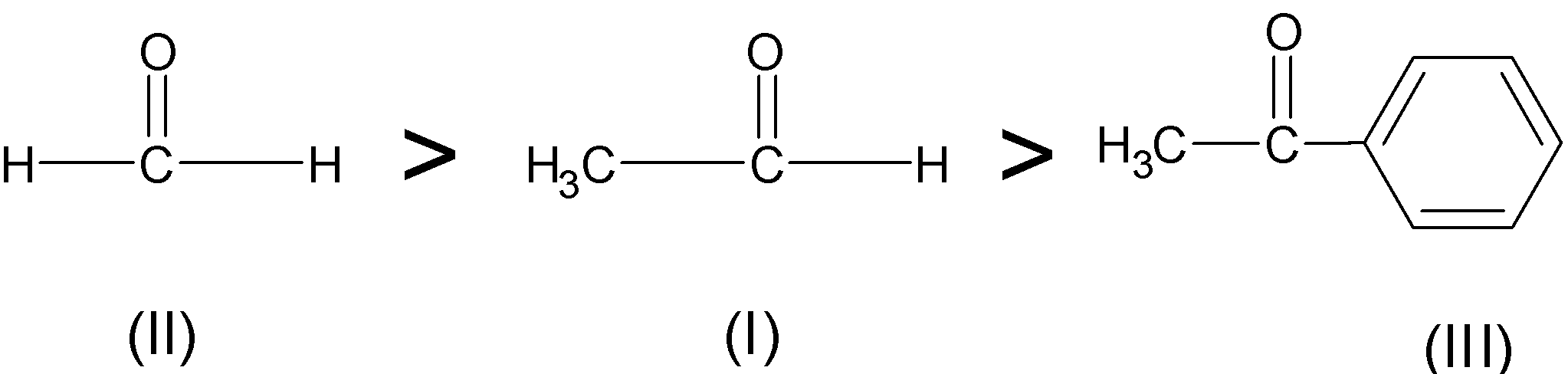
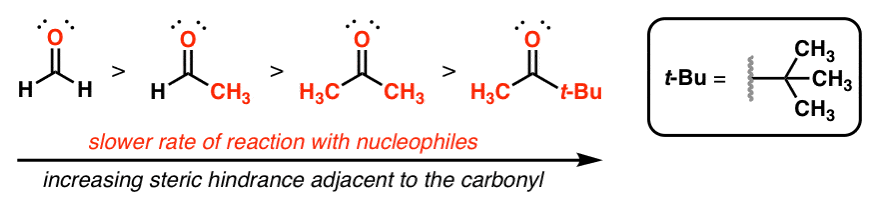
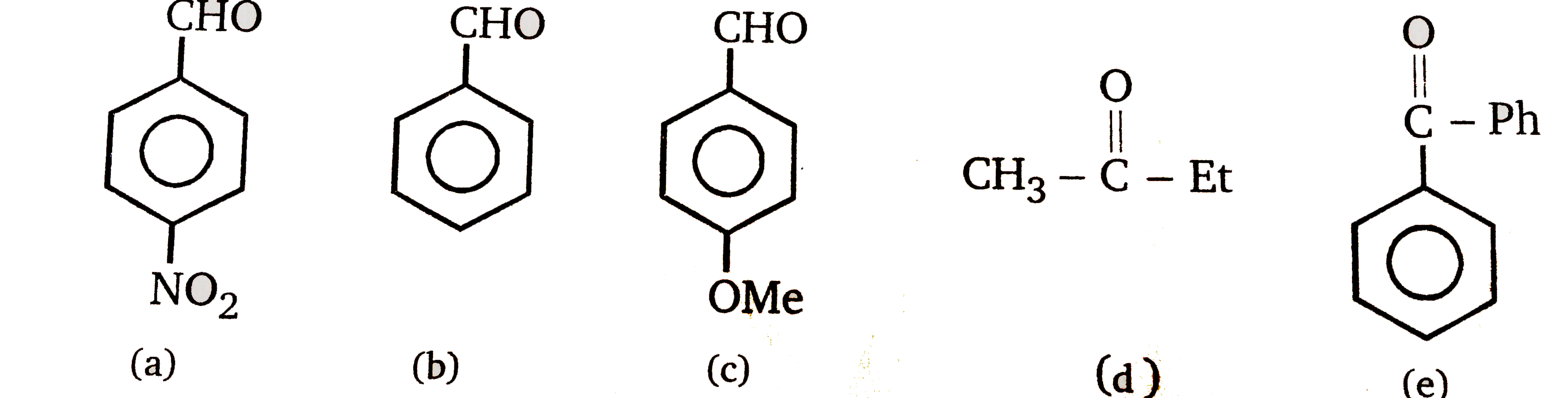order of reactivity of carbonyl compounds towards nucleophilic addition
|
Carbonyl Reactivity
reactivity of the following carbonyl compounds towards nucleophilic attack. Most reactive. Least reactive. OH. O. NR. NR2. O. O. Carbonyls have a "resonance |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
8.4. Arrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions. carbonyl group towards nucleophilic addition ... |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
compounds in increasing order of their reactivity in nucleophilic addition reactions. ... carbonyl oxygen activates the carbonyl group towards nucleophilic ... |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
compounds usually undergo conjugate nucleophilic additions named Michael additions. The nucleophilic activation of the a |
|
Lech201.pdf
than haloalkane and therefore they are less reactive towards nucleophilic substitution reaction 10.16 Arrange the compounds of each set in order of ... |
|
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
The carbon-oxygen double bond of the carbonyl group is extremely polarized in the direction of the highly electronegative oxygen. |
|
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
The carbon-oxygen double bond of the carbonyl group is extremely polarized in the direction of the highly electronegative oxygen. |
|
QUESTION BANK UNIT 12 ALDEHYDES KETONES
(b) Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction : CH3COCH3 HCHO |
|
Stereoselective Nucleophilic Additions to Aldehydes and Synthesis
25 May 2012 Stereoinduction models for carbonyl addition reactions ... 57. In order to investigate the inherent mode of reactivity for α-chloro aldehydes. |
|
Study material XII - Part 2.docx
Assertion : Carbonyl compounds take part in nucleophilic addition Arrange the following in the increasing order of their reactivity towards nucleophilic ... |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Carbonyl compounds are of utmost importance to organic chemistry. reactive than ketones in nucleophilic addition reactions due to steric and electronic. |
|
Reactions of Carbonyl Compounds
(b) Nucleophilic addition at the carbonyl group followed by protonation: compound will be much more highly reactive toward such nucleophiles. Case (b). |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
Since aldehydes and ketones both possess the carbonyl functional group they undergo similar chemical reactions. 1. Nucleophilic addition reactions. |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
nucleophilic addition to carbonyl compounds; however other nucleophiles can undergo the same reaction. carbon more reactive toward the alkyl group. |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
towards nucleophilic addition reactions. The nucleophilic activation of the ab-unsaturated carbonyl compounds is a ... The experimental order of. |
|
Carbonyl Reactivity
reactivity of the following carbonyl compounds towards nucleophilic attack. Most reactive. Least reactive Carbonyl Additions: Synthesis and Reactivity. |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
towards nucleophilic addition reactions. The nucleophilic activation of the ab-unsaturated carbonyl compounds is a ... The experimental order of. |
|
CHEMISTRY CL-12 MCQS TERM (2021-2022)
Arrange the following compounds in-decreasing order of their boiling less reactive than halo alkanes towards nucleophilic substitution due to resonance. |
|
Organic Chemistry 5e (Bruice) Chapter 17: Carbonyl Compounds II
18) List the following carbonyl compounds in order of decreasing reactivity toward nucleophiles: ester acid chloride |
|
Haloalkanes and Haloarenes Haloalkanes and Haloarenes
Table 10.4: Nucleophilic Substitution of Alkyl Halides (R–X). Reagent. Nucleophile Predict the order of reactivity of the following compounds in S. |
|
THE CHEMISTRY OF THE CARBONYL GROUP - University of Oxford
1) Nucleophilic addition to C=O A) Nucleophiles and electrophiles B) Reversible addition (hydrates and hemiacetals) C) Irreversible addition (reduction and Grignard addition) 2) Nucleophilic substitution of C=O A) acetals B) imines oxmies and hydrazones 3) Nucleophilic substitution at C=O A) tetrahedral intermediates in substitution- |
|
Arrange the following compounds in increasing order of their reactivity
There are two major types of carbonyl compounds: Type 1: The acyl group is attached to a group that does not have any lone pairs and cannot act as a leaving group O CC RHRR Type 2: The acyl group is attached to a group L: which has a lone pair and is either a good leaving group or can be protonated to make it a good leaving group CRL |
|
Lecture 5: Nucleophilic Addition Reactions of Carboxylic Acid
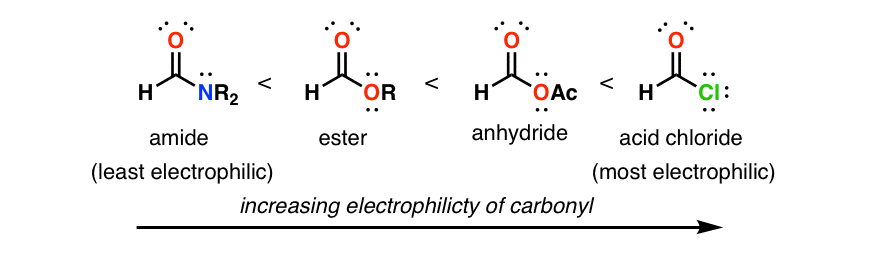
Relative Reactivity of Carboxylic Acid Derivatives Towards Nucleophilic Attack There are many types of carboxylic acid derivatives The order of reactivity towards nucleophilic addition-elimination of a few is as follows: Cl O O OO O O N O O O >>> > acid chloride acid anhydride ester amide carboxylate |
|
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS GRIGNARD REACTION Carbonyl-containing compounds such as aldehydes ketones carboxylic acid and acid anhydrides possess an electophilic carbon center which can be attacked by various nucleophiles (Figure 1) |
|
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl
The order of reactivity for the carboxylic acid derivatives is extremely important in organizing the large amount of information and the large number of reactions in this chapter The reactivity order is as follows: Acid chloride > anhydride > thioester > ester > amide |
|
16: Addition and Substitution Reactions of Carbonyl Compounds
We illustrate the basic mechanistic features of nucleophilic addition and substitution reactions on carbonyl compounds using the nucleophile hydroxide ion that we write either as HO-or -OH (Figure 16 006) Figure 16 006 HO-in HOH (16 2A) Water is generally the solvent for reactions of the hydroxide nucleophile -OH 3 |
Why does the Order of reactivities of carbonyl compounds increase in nucleophilic addition reactions?
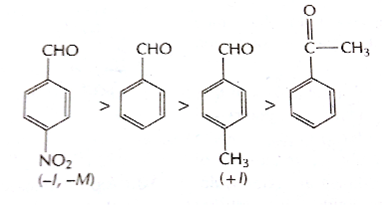
- The electron density at the carbonyl carbon increases with the increase in the +I effect. As a result, the chances of attack by a nucleophile decrease. Hence, the increasing order of the reactivities of the given carbonyl compounds in nucleophilic addition reactions is:
Is carboxylic acid a nucleophile?
- NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS GRIGNARD REACTION Carbonyl-containing compounds, such as aldehydes, ketones, carboxylic acid and acid anhydrides, possess an electophilic carbon center, which can be attacked by various nucleophiles (Figure 1).
What happens if a nucleophile attacks a carbonyl group?
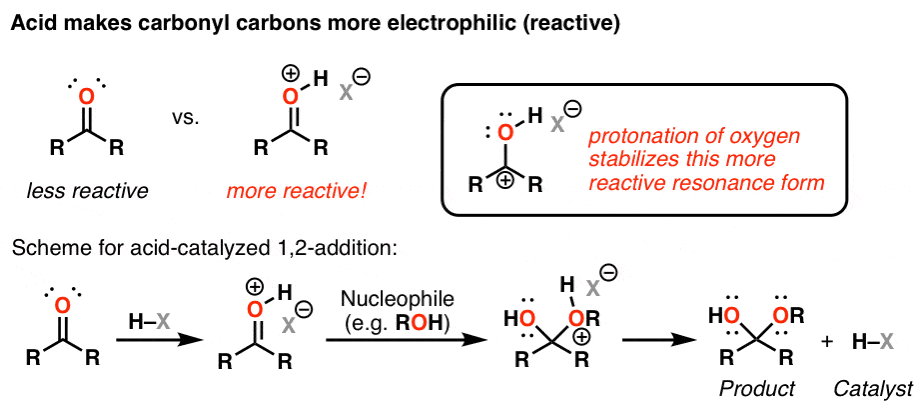
- However, when weak nucleophiles are used, the carbonyl group must be activated with the help of an acid catalyst for the nucleophilic addition reaction to proceed. The carbonyl group has a coplanar structure and its carbon is sp 2 hybridized. However, the attack of the nucleophile on the C=O group results in the breakage of the pi bond.
What is the least reactive compound among the following carbonyl compounds?
- The least reactive compound among the following carbonyl compounds towards nucleophilic addition reactions is Reactivity of a carbonyl compound towards nucleophilic addition reactions depends on its e- density. e? density at carbonyl carbon increases with increase in the +I effect. This +I effect is the least in aldehydes.
|
Nucleophilic Addition Reactions of Carboxylic Acid Derivatives
Relative Reactivity of Carboxylic Acid Derivatives Towards Nucleophilic Attack We need to be able to rationalise this order of reactivity In essence the more δ+ charge on the carbonyl carbon, the more electrophilic is the group and more reactive it is towards nucleophilic addition |
|
Nucleophilic addition to the carbonyl group
Molecular orbitals explain the reactivity of the carbonyl group We are now You met nucleophilic addition to a carbonyl group on pp 115 and 121 The polarization of the antibonding π* orbital towards carbon is also ketones Having introduced you to the sequence of events that makes up a nucleophilic attack at C=O |
|
Reactions of Carbonyl Compounds
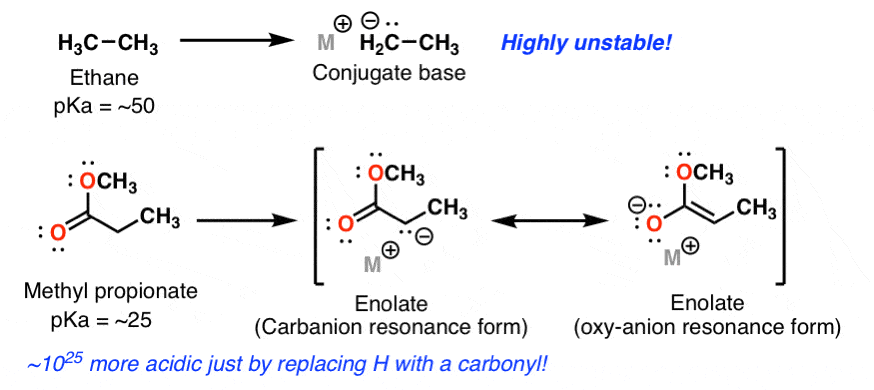
In many reactions at carbonyl groups, a key step is addition of a nucleophile, For acetaldehyde, the half-life of the exchange reaction is on the order of 1 transfer in activating the carbonyl group toward nucleophilic attack, (2) the reactivity |
|
Addition, Condensation and Substitution Reactions of Carbonyl
of carbonyl compounds toward addition of nucleophiles Several factors The reactivity of carbonyl compounds toward hydration parallels the order indicated in |
|
Chemical reactivity of carbonyl compounds - International Journal of
The observed reactivity order is shown below: into the reactivity scale towards nucleophiles: additions of alcohols to aldehydes and ketones take place The |
|
Reactions involving carbonyl compounds - NPTEL
I Nucleophilic addition reactions Carbonyl group of aldehyde, ketone, carboxylic acid, ester etc , a The C-O bond is polarized towards the more electronegative oxygen Bonding in In order to get product in good yields, it is necessary to |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
The reactions of aldehydes and ketones can be divided into two main categories: nucleophilic addition to carbonyl compounds; however, other nucleophiles can undergo the carbon more reactive toward the alkyl group The product of one full equivalent of base is required in order to form the the ionized product in |
|
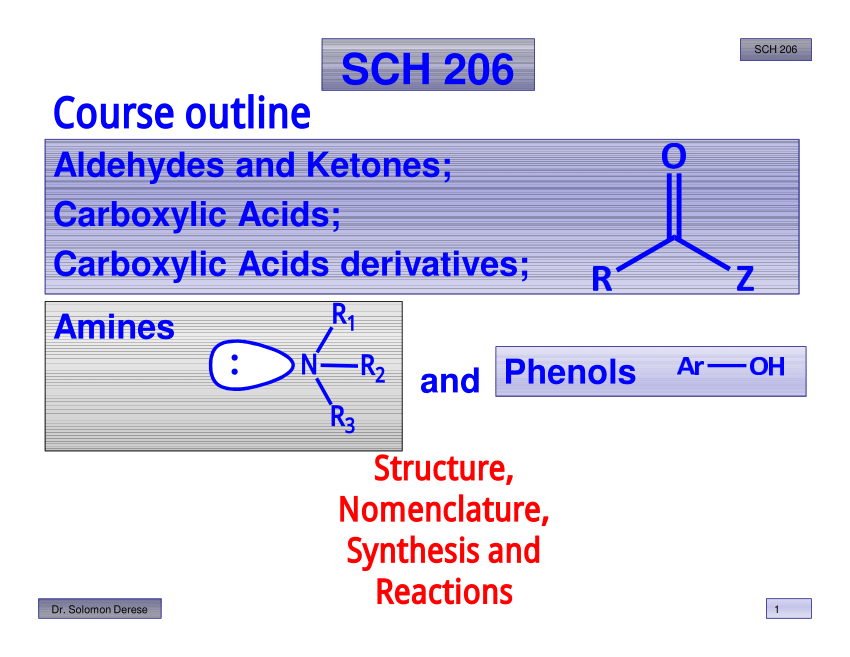
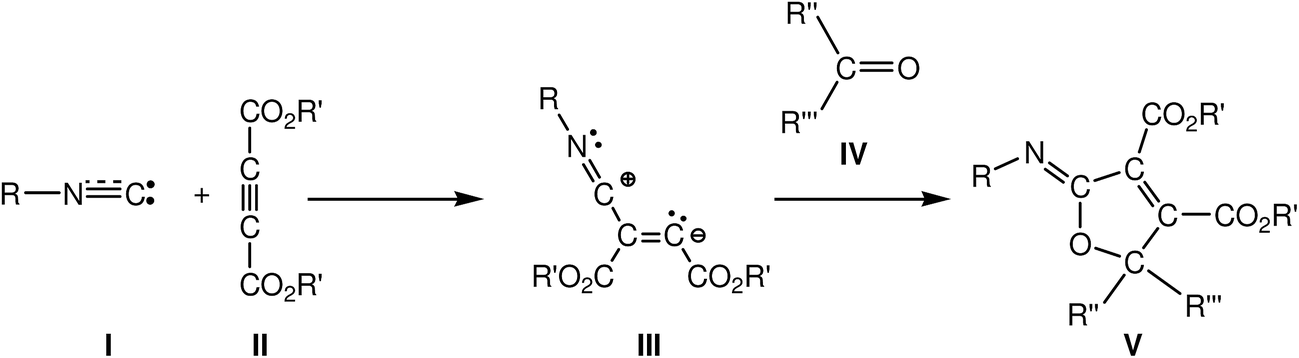
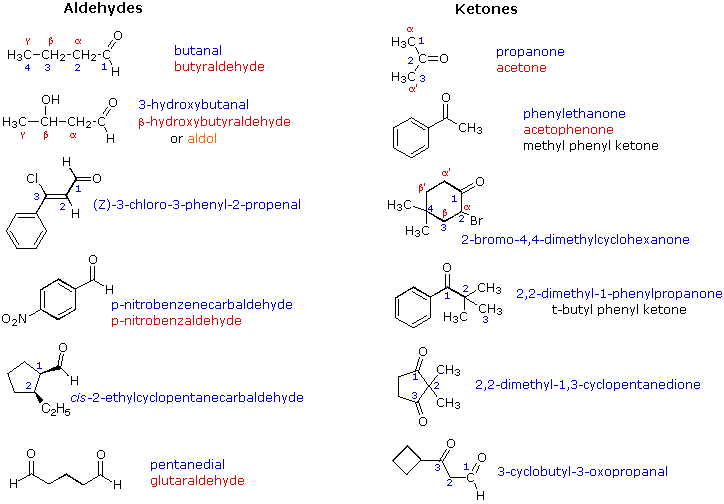
SCH 206 Course outline
carbonyl group less electrophilic The order of reactivity of carboxylic acid derivatives toward nucleophilic acyl substitution can be explained on the basis of the |