out of lyophilic and lyophobic sols which one can be easily converted into a gel and why
|
Unit-1 Setting.pmd
To prepare (a) lyophilic sol; and (b) lyophobic sol. Theory (vi) Out of lyophilic and lyophobic sols which one can be easily converted into a gel and why? |
|
B. Sc. II-Sem Colloidal state (1) The foundation of colloidal chemistry
Lyophobic sols can be easily coagulated by the addition of small quantity of an electrolyte. •. When a lyophilic sol is added to any lyophobic sol it becomes |
|
Surface Chemistr face Chemistr face Chemistry
In this Unit you will be studying some important features of surface chemistry such as adsorption |
|
Colloids
nature could be converted into a colloid by subdividing it into particles The property of lyophilic sols to prevent the precipitation of a lyophobic sol is ... |
|
E.J.W. Verwey and J.Th.G. Overbeek. Theory of the Stability of
have maintained their individuality in that they hold together. I. § 1. دری. LYOPHOBIC AND LYOPHILIC COLLOIDS. 3 a positively charged AgI sol can easily be ... |
|
UNIT 20: COLLOIDS AND mCROMOLECULES
It has been observed that lyophobic sols in contrast to lyophilic sols |
|
Note: The yellow coloured questions are from the reduced portion of
Lyophobic sols are readily coagulated as compare to lyophilic sols. 1. What Name one method by which coagulation of lyophobic sol can be carried out. 5 ... |
|
اﻟﺟﺎﻣﻌﺔ اﻟﺗﮐﻧوﻟوﺟﯾﺔ ﻗﺳم اﻟﮭﻧدﺳﺔ اﻟﮐﯾﻣﯾﺎوﯾﺔ اﻟﺛﺎﻧﯾ
nature could be converted into a colloid by subdividing it into The lyophilic sol used to protect a lyophobic sol from precipitation is referred to as. |
|
Unacademy
For example inert gases |
|
Physical Chemistry
On evaporating the sol the dispersed phase obtained can be easily reconverted into the solution by simply agitating it with the dispersion medium. Gums |
|
Unit-1 Setting.pmd
(v) How can you convert a colloidal dispersion of sulphur into a true solution? (vi) Out of lyophilic and lyophobic sols which one can be easily converted into |
|
Preparation of one lyophilic and one lyophobic sol.docx
Chemistry Practical Class 12 Preparation of One. Lyophilic Q7: Out of lyophilic and lyophobic sols which could be quickly converted into a gel and why? |
|
Surface Chemistry Viva Questions With Answers
Question.37. Out of lyophilic and lyophobic sols which can be easily converted into a gel and why? Answer. Lyophilic sols can be |
|
Lech105.pdf
catalysis and colloids including emulsions and gels. temperature may pass into chemisorption as the temperature is ... More easily liquefiable. |
|
B. Sc. II-Sem Colloidal state (1) The foundation of colloidal chemistry
Lyophilic and lyophobic colloidal solutions (or sols) are generally nature become these can be precipitated and directly converted into colloidal state. |
|
Colloids: Thomas Graham (1861) studied the ability of dissolved
converted into a colloid by subdividing it into particles of colloidal size. Lyophilic and Lyophobic Sols or Colloids: Sols are of two types –. |
|
Note: The yellow coloured questions are from the reduced portion of
Name the process by which alcohols convert directly into Name one method by which coagulation of lyophobic sol can be carried out. 5. Out of KI or K2SO4 ... |
|
Surface chemistry
catalysis and colloids including emulsions and gels. temperature may pass into chemisorption as the temperature is ... More easily liquefiable. |
|
UNIT 20: COLLOIDS AND mCROMOLECULES
Coarse suspensions can be easily filtered using ordinary filter papers so as Solid Emulsions and Gels ... preparation of lyophilic and lyophobic sols. |
|
UNIT 20: COLLOIDS AND mCROMOLECULES
Coarse suspensions can be easily filtered using ordinary filter papers so as Solid Emulsions and Gels ... preparation of lyophilic and lyophobic sols. |
|
OLLOIDS - NCERT
medium colloidal sols are divided into two categories namely lyophilic (solvent attracting) and lyophobic (solvent repelling) If water is the dispersion medium the terms used are hydrophilic and hydrophobic Egg albumin starch and gum are lyophilic sols Freshly prepared ferric hydroxide aluminium hydroxide and |
|
12 Chemistry English 2020 21 Electrochemistry Solution
to NaOH solution The lyophilic sols are directly formed by mixing and shaking the substance with a suitable liquid Lyophobic sols cannot be prepared by direct mixing and shaking Special methods are employed to prepare these • Egg : One • Sodium chloride : 5g • Ferric chloride : 2g • Aluminium chloride : 2g • Starch/gum : 500 mg |
|
8 COLLOIDS - The National Institute of Open Schooling (NIOS)
lyophilic colloidal solution has been formed then dispersed phase and dispersion medium can be separated easily Once separated these can again be formed by remixing the two phases These sols are quite stable If water is used as dispersion medium then it is termed as hydrophilic colloid |
|
BSc (Honours) Part-I Paper-I Topic: Protection of Colloid
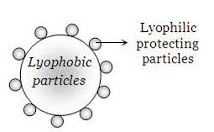
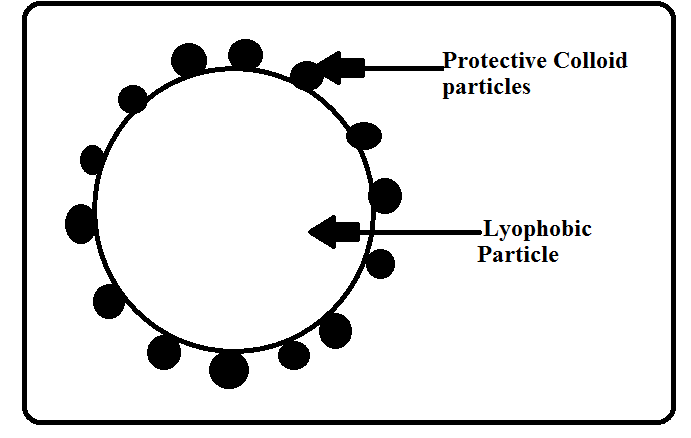
Lyophilic sols are more stable than lyophobic sols Lyophobic sols can be easily coagulated by the addition of small quantity of an electrolyte When a lyophilic sol is added to any lyophobic sol it becomes less sensitive towards electrolytes Thus lyophilic colloids can prevent the coagulation of any lyophobic sol |
|
NCERT Solutions for Class 12 Chemistry Chapter 5 Surface
Such sols are called lyophobic sols These sols are irreversible in nature For example: sols of metals Now two things determine the stability of hydrophilic sols - the salvation of a colloidal particle and the presence of a gas At the same time the presence of a charge determines the stability of a hydrophobic sol |
What is the difference between lyophilic sols and lyophobic sols?
- These are formed by organic substances like gum, starch, protein etc. These are reversible and more stable. 20. Lyophobic sols : There is very little interaction between dispersed phase and sulphides etc. These are irreversible and less stable. 21. The stability of lyophilic sols is due to their greater hydration in the solution. 22.
How do solvation effects contribute to the stability of lyophilic systems?
- (ii) Solvation effects contribute much towards the stability of lyophilic systems. For example, gelatin has a sufficiently strong affinity for water. It is only because of the solvation effects that even the addition of electrolytes in small amounts does not cause any flocculation of hydrophilic sols.
Can lyophilic colloids prevent the coagulation of a lyophobic Sol?
- Thus, lyophilic colloids can prevent the coagulation of any lyophobic sol. “The phenomenon of preventing the coagulation of a lyophobic sol due to the addition of some lyophilic colloid is called sol protection or protection of colloids.”
What are the types of colloidal sols?
- Further, a heterogeneous system of a solid asdispersed phase and a liquid as dispersion medium is called a sol. Dependingupon the nature of interaction between the dispersed phase and the dispersionmedium, colloidal sols are divided into two categories, namely, lyophilic (solventattracting) and lyophobic (solvent repelling).
|
Unit 1(Colloids) - NCERT
the solvation of colloidal particles by the solvent whereas lyophobic sols are stabilised by (vi) Out of lyophilic and lyophobic sols, which one can be easily converted into a gel and why? (vii) Differentiate between a gel and a sol (viii) What |
|
Surface Chemistry - SelfStudys
gel (ii) NH3 is adsorbed by charcoal Examples : (i) Water vapours absorbed by anhydrous example, when a solid substance is broken into two pieces It can be easily reversed by heating or decreasing the pressure (a) Lyophilic and (b) Lyophobic sols can be precipitated and directly converted into colloidal state |
|
Physical Chemistry
Gels Preparation of Gels Emulsions Determination of type of Emulsion Preparation of into colloidal solution by simply shaking it with the dispersion medium Colloidal Lyophilic Lyophobic sols 1 Preparation Can be easily prepared by direct mixing insufficient to convert hydroxide completely into chloride) |
|
UNIT -5 SURFACE CHEMISTRY
Name catalyst used in the conversion of alcohols into gasoline (petrol) Sol 9 Name the dispersed phase in gel Liquid 10 Give an example for oil in What happens when an electrolyte is added to lyophobic sol? gas will be adsorbed readily on the surface of 1 gram of activated charcoal Gets easily coagulated 10 |
|
B Sc II-Sem Colloidal state (1) The foundation of colloidal chemistry
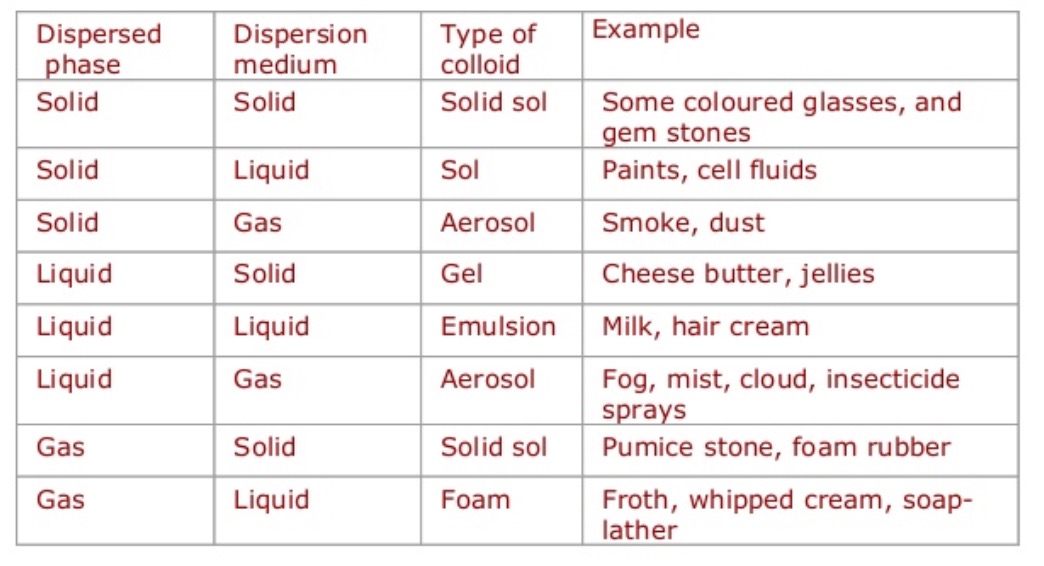
(1) Phases of colloids: We know that a colloidal solution is of heterogeneous nature Gels Cheese, butter, boot polish, jelly, curd Solid Solid Solid sols ( coloured Lyophilic and lyophobic colloidal solutions (or sols) are generally prepared by become these can be precipitated and directly converted into colloidal state |
|
Colloids
nature, could be converted into a colloid by subdividing it into particles of WHAT ARE GELS ? Some features of lyophilic and lyophobic sols are listed below ethanol but on transference to water, the molecules precipitate out to form colloidal particles all the electrolyte present in the sol can be removed easily Sol |
|
E:\Study Material\Physical Chem - Parishram
Charcoal, silica, gel, alumina gel are good adsorbents because they have It convert alcohol directly into gasoline by dehydrating them to give a mixture of These solutions are easily formed and the lyophilic colloids are reversible in nature Coagulation of lyophobic sols can be carried out by the following methods |
|
Class: BSc II- Semester Paper Name: Physical - JNMPG COLLEGE
Gels Cheese, butter, boot polish, jelly, curd Liquid Liquid Emulsions dispersed phase and the dispersion medium are called lyophobic colloids ” (iii) They are Property Lyophilic sols (Suspensoid) Lyophobic sols (Emulsoid) Surface Freshly prepared ferric hydroxide can be converted into colloidal state by shaking it |