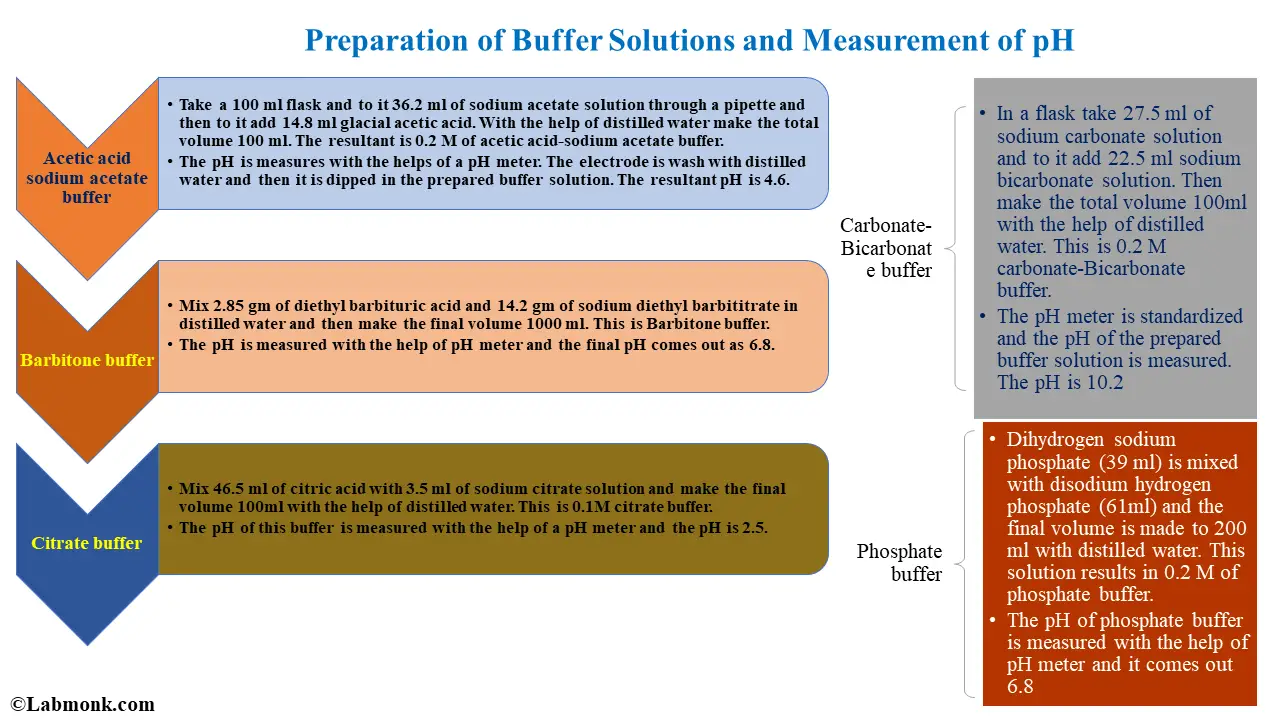
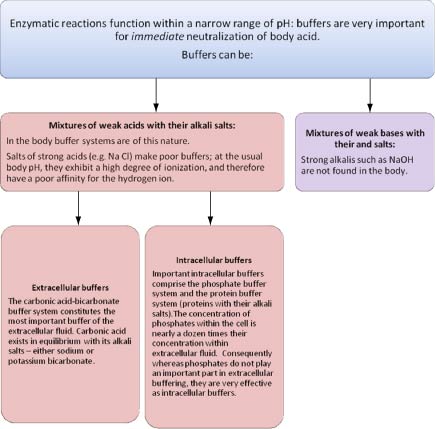
phosphate buffer ph range
|
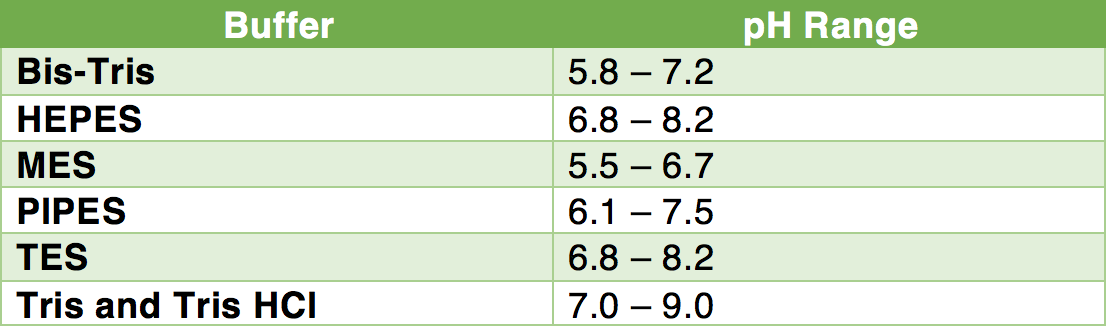
Buffer pKa and pH Range Values
Potassium Phosphate Monobasic – H. 2. KO. 4. P. Potassium Phosphate |
|
Calbiochem
Figure 5: Effect of Temperature on pH of Phosphate Buffer. Page 18. Table 2: pK Phosphate Buffer; pH range 5.8 to 8.0. (a) 0.1 M Sodium phosphate monobasic ... |
|
A Guide to HPLC and LC-MS Buffer Selection
In this case a blend of phosphate and acetate buffer will allow continuous variation of the mobile phase from. 2<pH<8. Once you find the desired pH |
|
Phosphate buffer preparation of 0.1 M potassium phosphate buffer at
By varying the amount of each salt a range of buffers can be prepared that buffer well between pH 5.8 and pH 8.0 (please see the tables below). Phosphates have |
|
TCEP•HCl
pH range. ... 7 demonstrated that TCEP can be used for this purpose: Add TCEP at a final concentration of 3.8-4.0mM to 10mg/mL IgG in 0.1M phosphate buffer (pH ... |
|
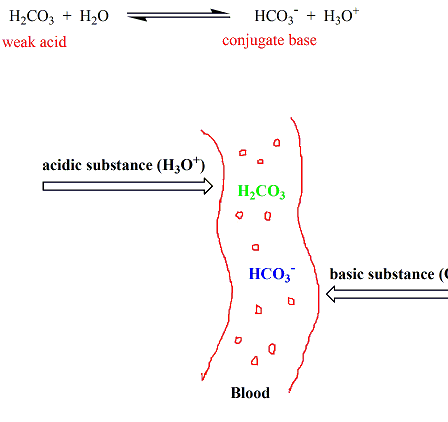
EFFECTS OF pH AND THE COMPONENTS OF BICARBONATE
EFFECT OF CONCENTRATION OF POTASSIUM PHOSPHATE BUFFER SOLUTION AT PH 6.9 ON THE. NITROGEN FRACTIONS OF POTATO DISCS AT 230 C. DURING 72 HOURS*. CONCEN |
|
Abstract Phosphate buffered formulations show higher subvisible
Formulation optimization (e.g. choice of buffer |
|
Ni-NTA Purification System
Aug 13 2015 pH of the 5X Native Purification Buffer to create 1X Native Purification Buffer ... 20 mM Sodium Phosphate Buffer |
|
The solubility of sickle and non-sickle hemoglobins in concentrated
Packed cells were hemolyzed in 5 volumes of 5 mM potassium phosphate buffer pH 7.4 |
|
Phosphate Buffered Saline System (PBS1) - Datasheet
of pH of a buffer when diluted with an equivalent volume of water is 0.08.2 Therefore |
|
Buffers
The phosphate buffer system has a pK a of 6.86. Hence it provides effective buffering in the pH range of 6.4 to 7.4. The bicarbonate buffer system plays an. |
|
A Guide to HPLC and LC-MS Buffer Selection
With a slight extension of the buffering range from ±1 pH units from the pKa phosphate and acetate can cover the entire pH range of 2<pH<8 normally used |
|
Phosphate buffer preparation of 0.1 M potassium phosphate buffer at
phosphate and dibasic monohydrogen phosphate. By varying the amount of each salt a range of buffers can be prepared that buffer well between pH 5.8 and pH |
|
THE INFLUENCE OF NEUTRAL SALTS ON THE pH OF
It was decided to study first the effects of addition of neutral salt to the phosphate buffer system because it is the one used extensively in biological |
|
Phosphate Buffer pH 7.2 Product Information Page
Phosphate Buffer pH 7.2 is used for the preparation of microbiological dilution blanks. Product Summary and Explanation. The formula for phosphate buffer |
|
The Effect of Formaldehyde Fixation on RNA
Incubation in phosphate buffer. (pH 9) resulted in an electrophoretic migration pattern running just slower than native RNA. In the total RNA system the low. |
|
The solubility of sickle and non-sickle hemoglobins in concentrated
Packed cells were hemolyzed in 5 volumes of 5 mM potassium phosphate buffer pH 7.4 |
|
Buffer pKa and pH Range Values
Buffer pKa and pH Range Values. For preparation of. Buffers in the pH. Buffers. pKa range. Hydrochloric Acid - HCl Potassium Phosphate Tribasic – K3PO4. |
|
Potassium Phosphate Buffer pH 6.0 1X
Potassium Phosphate Buffer pH 6.0 1X. Product Code: TL1102. Disclaimer : User must ensure suitability of the product(s) in their application prior to use. |
|
The effect of phosphate buffer on improving the performance of
2 ???. 2018 ?. 6 Usually the one-stage. ATAD is conducted without pH control |
|
Phosphate buffer - University of Nebraska–Lincoln
phosphate buffer Information from cshprotocols org:Gomori buffers the most commonly used phosphate buffers consist of a mixture of monobasic dihydrogenphosphate and dibasic monohydrogen phosphate By varying the amount of each salt a range of buffers canbe prepared that buffer well between pH 5 8 and pH 8 0 (please see the tables below) |
|
Due to the variety of factors that influence pH it is
influences pH The dilution value for phosphate defined as the change of pH of a buffer when diluted with an equivalent volume of water is 0 08 (2) Therefore a 25 mM phosphate buffer prepared with half of the volumes of P8709 and P8584 indicated on the table for a specific pH would be approx |
|
How to prepare Phosphate buffer solution pH 74 - ResearchGate
buffers from pH 2 8 to 4 8 There are additional buffer choices if acidic analytes would be not be ionizable at lower pH For more details on buffers refer to Table 1 Buffer v/v ( ) pKa at 25 °C Max pH range UV cutoff (nm) Trifluoroacetic acid (TFA) 0 1 0 3 210 0 05 0 3 210 0 01 0 3 210 Phosphate pK1 2 1 1 1 –3 1 |
How to prepare a phosphate buffer with pH 7.4?
- In preparing Phosphate buffer solution with a pH = 7.4. Add 250.0 mL of 0.2 M potassium dihydrogen phosphate R to 393.4 mL of 0.1 M sodium hydroxide.
How do you prepare phosphate buffer pH 6.8?
- Phosphate Buffer pH 6.8, 0.2 M Mixed: Dissolve 13.872 g of potassium dihydrogen phosphate and 35.084 g of disodium hydrogen phosphate in sufficient water to produce 1000 ml. Store in a cold place.
What is the pH range of buffer?
- Buffer range is the pH range over which the buffer is most effective. This range is usually between 0.1 and 10. In practice, when the [A – ]/[HA] ratio is less than 0.1 or when the concentration of one component is more than 10 times as concentrated as the other, buffering capacity is weak.
|
Buffers
The phosphate buffer system has a pK a of 6 86 Hence, it provides effective buffering in the pH range of 6 4 to 7 4 The bicarbonate buffer system plays an |
|
413 BUFFER SOLUTIONS - ResearchGate
Phosphate buffer solution pH 7 0 R3 Dissolve 5 g of potassium dihydrogen phosphate R and 11 g of dipotassium hydrogen phosphate R in 900 ml of water R Adjust to pH 7 0 (2 2 3) with dilute phosphoric acid R or dilute sodium hydroxide solution R Dilute to 1000 ml with water R and mix |
|
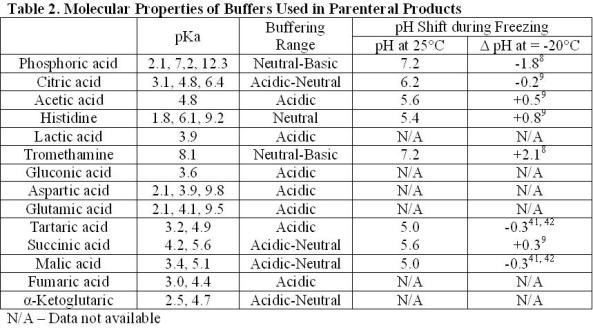
Development of Buffer Systems for pH Control and Evaluation of pH
of rapid drifting of pH downward during fennentation To evaluate the effective- ness of citric or phosphoric acids as components of phosphate-bicarbonate buffer |
|
Appendix
3 Sodium phosphate buffer pH 5 7–8 0 Stock solutions A: 0 2 M solution of monobasic sodium phosphate, NaH2PO4 (27 8 g in 1 L |
|
Buffering agents and Buffers - INTERCHIM
I e the pH should be determined at the final temperature, in presence of salts [i e phosphate pH change with salts concentration] near the pKa of the buffering |

















/462888083-56a09bba5f9b58eba4b207ff.jpg)