physical and chemical properties of aldehydes and ketones
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
correlate physical properties and chemical reactions of aldehydes ketones and carboxylic acids |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
to those of C=C double bonds. 6. Properties of Aldehydes and Ketones. • Aldehydes and ketones are polar molecules because the C=O bond has a dipole moment:. |
|
Aldehydes and Ketones
IN THIS AND several of the following chapters we study the physical and chemical properties of compounds containing the carbonyl group |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
correlate physical properties and chemical reactions of aldehydes ketones and carboxylic acids |
|
MATHEMATICS
Physical properties-Chemical reactions: Cleavage of C-O bond and electrophilic substitution of aromatic ethers. ALDEHYDES AND KETONES. |
|
The Carbonyl Group
Learn the important physical properties of the aldehydes and ketones. • Learn the major chemical reaction of aldehydes and ketones and learn how to predict. |
|
The Carbonyl Group Nomenclature of Aldehydes and Ketones
Learn the IUPAC system for naming aldehydes and ketones. •. Learn the important physical properties of the aldehydes and ketones. •. Learn the major chemical |
|
TELANGANA STATE BOARD OF INTERMEDIATE EDUCATION
physical properties and Chemical reactions Aldehydes and. Ketones 12.9 Nomenclature and structure of carbonyl group. 12.10Preparation of aldehydes and |
|
Botany-I: Intermediate First Year
substituted benzenes; Physical properties of aldehydes and ketones; Chemical reactions of aldehydes and ketones-nucleophilic addition reduction |
|
Chapter 4 Aldehydes and Ketones - Angelo State University
Physical Properties of Aldehydes and Ketones Name Molecular weight Boiling point Solubility in water butane 58 g/mol 0°C Insoluble propanal 58 g/mol 49°C Soluble acetone 58 g/mol 56°C Soluble 1-propanol 60 g/mol 97°C Soluble Boiling Points: Alcohols Aldehydes/Ketones Ethers Alkanes Water Solubility: Alcohols Aldehydes/Ketones Ethers Alkanes 20 |
|
27 ALDEHYDES KETONES AND CARBOXYLIC ACIDS
They differ in that the carbonyl carbon in ketones is bound to two carbon atoms (RCOR’) while that in aldehydes is bound to at least one hydrogen (H2CO and RCHO) Thus aldehydes always place the carbonyl group on a terminal (end) carbon while the carbonyl group in ketones is always internal |
|
10 Aldehydes and Ketones - Elsevier
Section 10 3 Physical Properties of Aldehydes and Ketones Table 10 1 Comparisons Of Physical Properties 10 3 PHYSICAL PROPERTIES OF ALDEHYDES AND KETONES CO CH 3 CH 3 2-propanone (2 9 D) (acetone) CO CH CH or ?+ ? Table 10 1 Comparisons of Physical Properties Property CH 3 CH 2 CH 2 CH 3 CH 3 CH 2 CHO CH 3 CH 2 CH 2 OH Dipole moment Butane |
|
AldehydesAldehydesAldehydes Ketonestonestones
235 Aldehydes Ketones and Carboxylic Acids The physical properties of aldehydes and ketones are described as follows Methanal is a gas at room temperature Ethanal is a volatile liquid Other aldehydes and ketones are liquid or solid at room temperature The boiling points of aldehydes and ketones are higher than |
|
27 ALDEHYDES KETONES AND CARBOXYLIC ACIDS
The physical properties and chemical reactions ofaldehydes and ketones are a direct consequence of this polarisation The dipole-dipole attraction between the molecules of aldehydes and ketonesresults in their higher boiling points as compared to the hydrocarbons of similarmolecular weight |
|
Searches related to physical and chemical properties of aldehydes and ketones filetype:pdf
Many ketones and aldehydes are fragrant Predict the products of the oxidation of the following aldehydes Acetaldehyde Benzaldehyde 2 Ketones resist oxidation by most oxidizing agents 3 Oxidation is used as a chemical test to distinguish between an aldehyde and a ketone B Reduction Aldehyde Ketone |
What is the difference between aldehydes and ketones?
- Aldehydes have at least one hydrogen atom bonded to the carbonyl group, theother group may be either a hydrogen or an alkyl (or aryl) group. In ketones,the carbonyl group is bonded to two alkyl or aryl groups. The two groupsbonded to a ketone may be similar or different resulting in a symmetrical oran unsymmatrical ketone, respectively.
Do aldehydes and ketones undergo nucleophilic addition reactions?
- Since aldehydes and ketones both possess the carbonyl functionalgroup, they undergo similar chemical reactions. Contrary to electrophilic addition reactions observed in alkenes, thealdehydes and ketones undergo nucleophilic addition reactions.
Which reaction gives acetaldehyde and ketone?
- By ozonolysis of alkenes: As we know, ozonolysis of alkenesfollowed by reaction with zinc dust and water gives aldehydes,ketones or a mixture of both depending on the substitutionpattern of the alkene (Unit 9, Class XI). By hydration of alkynes: Addition of water to ethyne in thepresence of H2SO4 and HgSO4 gives acetaldehyde.
How are aldehydes oxidised to carboxylic acids?
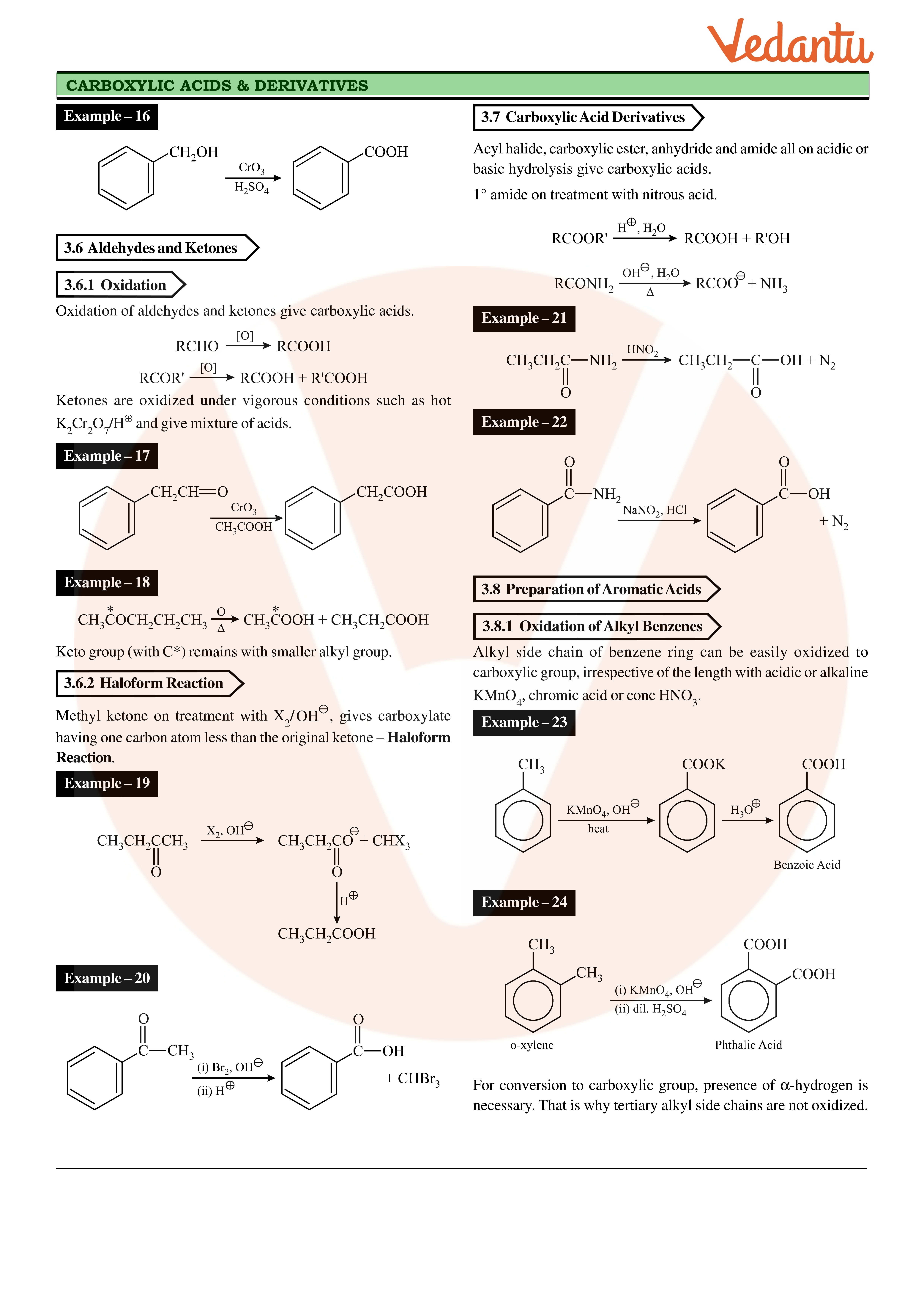
- Aldehydes are easilyoxidised to carboxylic acids by mild oxidising reagents such as Tollens’ reagent andFehling’s reagent. These oxidation reactions are used to distinguish aldehydes fromketones.
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Aldehydes and ketones are hydrogen bond acceptors; this makes them have considerable solubilities in water Ketones such as acetone are good solvents because they dissolve both aqueous and organic compounds Recall that acetone is a polar, aprotic solvent Reactions with acids: – The carbonyl oxygen is weakly basic |
|
Aldehydes and Ketones
Because of the polarity of the carbonyl group, aldehydes and ketones are polar com- pounds and interact in the liquid state by dipole–dipole interactions As a result, aldehydes and ketones have higher boiling points than those of nonpolar compounds with compa- rable molecular weight |
|
ALDEHYDES AND KETONES
Carbonyl carbon of both aldehyde and ketones is sp2 – hybridised, One of the three sp2 PHYSICAL PROPERTIES OF ALDEHYDES AND KETONE 1 |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
correlate physical properties and chemical reactions of aldehydes, ketones and carboxylic acids, with their structures; • explain the mechanism of a few selected |
|
27 ALDEHYDES, KETONES AND CARBOXYLIC ACIDS - NIOS
group makes the oxygen atom nucleophilic and basic while the carbon atom becomes electrophilic The physical properties and chemical reactions of aldehydes |
|
Organic Chemistry - UCR Chemistry - University of California
13 6 Spectrometric Properties of Carbonyl Compounds However, since most other chemical reactions of aldehydes and ketones are pKa values of these molecules are important physical properties that not only characterize them as |
|
Alcohols, Ethers, Aldehydes, and Ketones
ether, thiol, sulfide, disulfide, aldehyde, and ketone families, and describe how Properties of Alcohols common leaving groups used in organic chemistry |
|
PHYSICOCHEMICAL PROPERTIES OF ORGANIC MEDICINAL
Aldehyde and ketones are "carbonyl" derivatives of the hydrocarbons A carbonyl is defined as an organic functional group consisting of a carbon atom involved |
|
Chapter 14 Aldehydes, Ketones, and Chiral Molecules
Physical Properties of Aldehydes and Ketones The carbonyl and chemical properties of the two forms called enantiomers are identical These two forms or |
|
Chemistry Notes for class 12 Chapter 12 Aldehydes, Ketones and
Physical Properties of Aldehydes and Ketones 1 Methanal (HCHO) is a gas at room temperature and its 40 aqueous solution is known as formalin |