pka of h2s
|
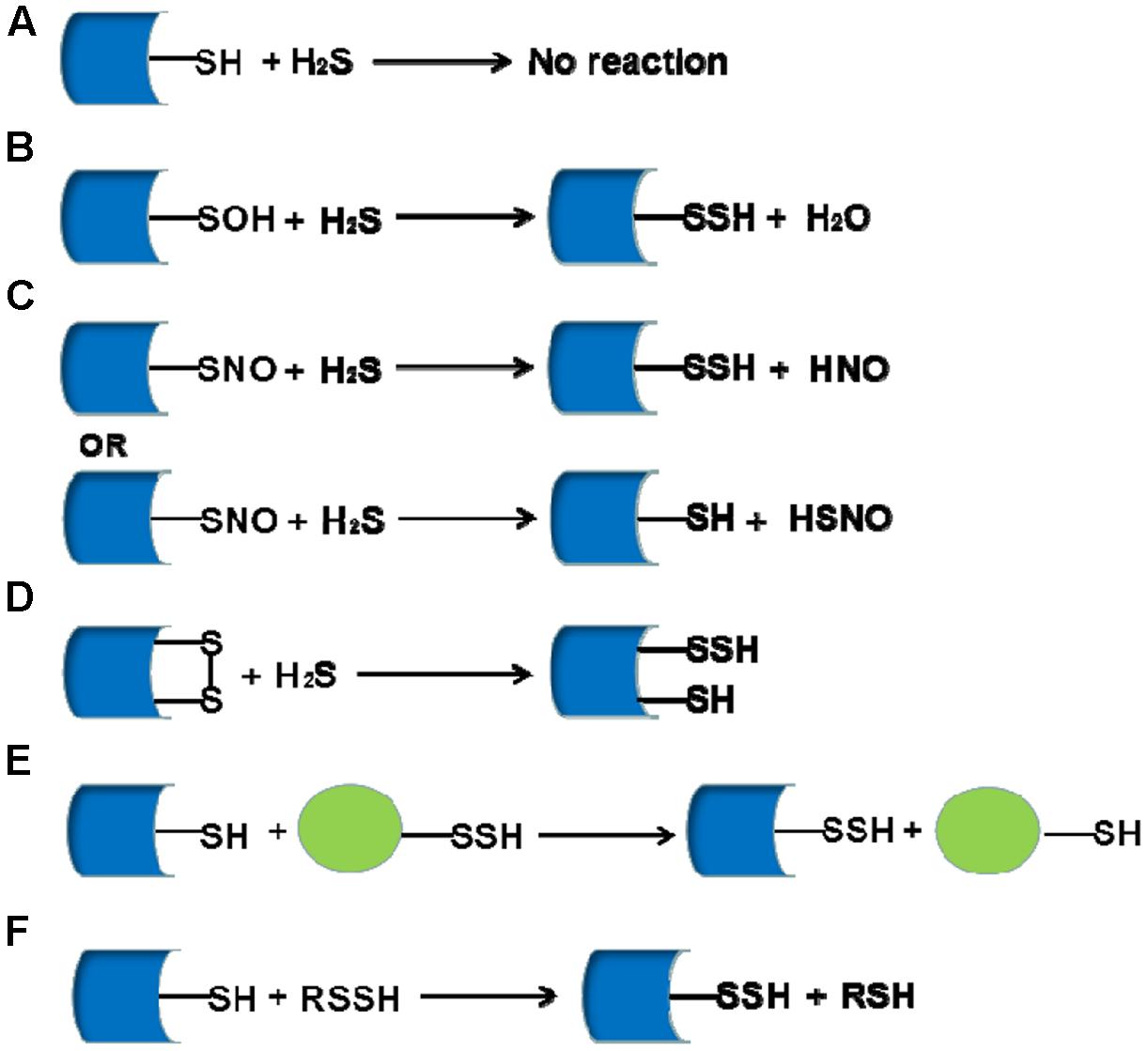
Reaction of Hydrogen Sulfide with Disulfide and Sulfenic Acid to
16 thg 6 2015 At neutral pH |
|
KỲ THI NĂNG KHIẾU LẦN IV NĂM HỌC 2019 - 2020 MÔN: Hóa học
Cho: pKa: H2S 700 ; 12 |
|
A practical guide to working with H2S at the interface of chemistry
11 thg 5 2016 Hydrogen sulfide (H2S) is the most recently accepted endogenously produced gasotransmitter and is ... H2S is a weak acid with a first pKa of 7.0 |
|
Kinetic study of hydrogen sulfide absorption in aqueous chlorine
14 thg 11 2013 H2S is a weak diacid in equilibrium with its conjugated base |
|
ĐỀ THI NĂNG KHIẾU LỚP 10
5 thg 5 2022 pHx=2 |
|
Synthesis and Hydrogen Sulfide Releasing Properties of
28 thg 6 2021 pKa of. H2S is 7.0 |
|
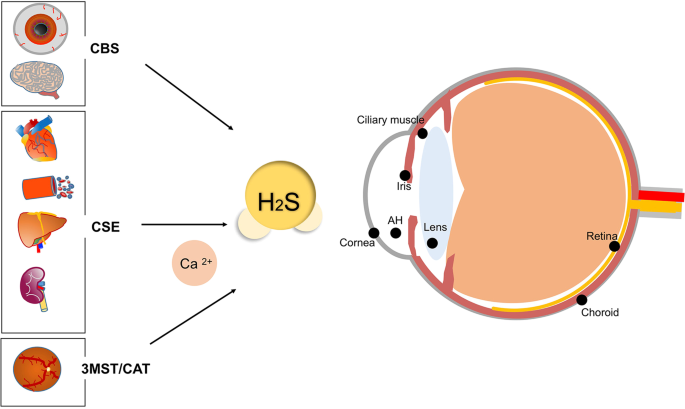
H2S catalysed by CBS regulates testosterone synthesis through
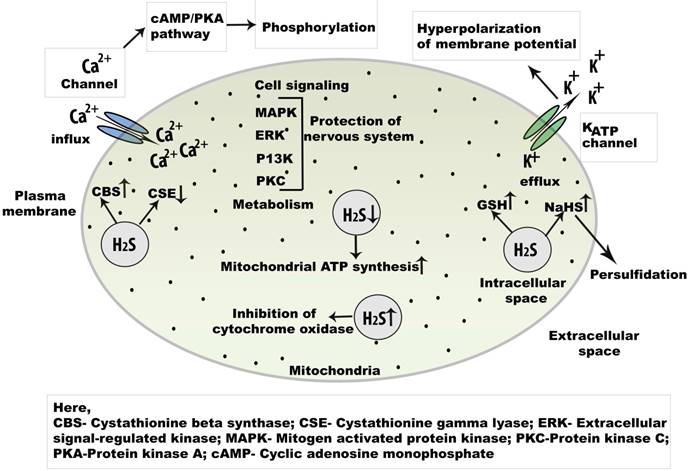
12 thg 2 2021 cAMP/PKA |
| 3461. WANG et Al. course of treatment ... |
|
D.H. Ripin D.A. Evans pKas of Inorganic and Oxo-Acids Chem 206
H2S. -1.7. 15.7. H3O. +. H2O. -1.3. HNO3. 3.29. HNO2. 4.72. HN3. 9.24. NH4Cl. 3.17. -0.98 pKa's of CH bonds at Nitrile Heteroaromatic |
|
Factors Governing the Protonation State of Cysteines in Proteins: An
to reproduce the pKa of H2S the hydration free energy of Zn2+ |
|
The new low value for the second dissociation constant for H2S: Its
In this equation pKa is the dilute solution pKa value with. [B-] and [HB] the indicated equilibrium molarities in the basic solutions. All the nonidealities |
|
Reaction of Hydrogen Sulfide with Disulfide and Sulfenic Acid to
16-Jun-2015 At neutral pH the species H2S |
|
Table of Acids with Ka and pKa Values* CLAS
%20Base%20Strength/Table%20of%20Acids%20w%20Kas%20and%20pKas.pdf |
|
A practical guide to working with H2S at the interface of chemistry
11-May-2016 Basic properties of biological hydrogen sulfide (H2S) ... H2S is a weak acid with a first pKa of 7.0 and a second pKa |
|
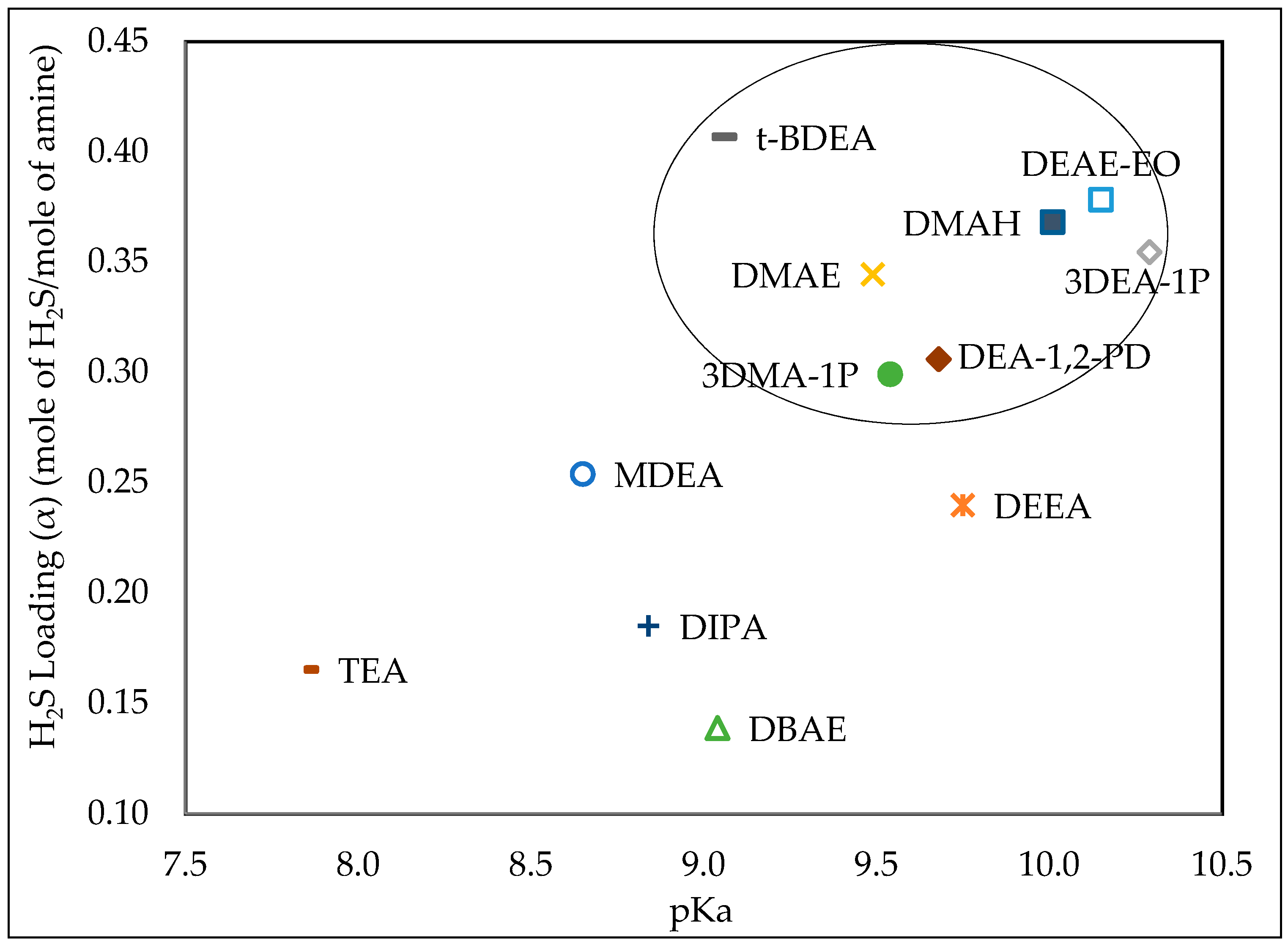
Study of Various Aqueous and Non-Aqueous Amine Blends for
15-Mar-2019 Maximum achieved H2S loadings as the function of pKa gas partial pressure |
|
Biosynthesis Quantification and Genetic Diseases of the Smallest
01-Jul-2021 Smallest Signaling Thiol Metabolite: Hydrogen Sulfide ... Deprotonation of H2S to form HS? has a pKa of 6.9–7.1. |
|
Spectroscopic investigation of the reaction of metallo
Keywords: Hydrogen Sulfide Protoporphyrin IX |
|
Kinetic study of hydrogen sulfide absorption in aqueous chlorine
(pKA3 = 13.94 at 293°K) at pH used in chemical scrubbing (pH ? 11-12). During H2S chemical scrubbing in chlorine solutions |
|
Acids and Bases: Molecular Structure and Acidity
listed in even the most extensive tables of pKa values. How can we know the pKa of a Recall that weaker bases have stronger conjugate acids so H2S. |
|
Hydrogen sulfide upregulates renal AQPâ•2 protein expression and
PKA inhibitor H89 but not the vasopressin 2 receptor (V2R) antagonist tolvaptan. Inhibition of endogenous H2S production impaired urine concentration in |
|
Photochemical reductive homologation of hydrogen cyanide using
15-May-2018 of either HCN 1 itself or hydrogen sulfide (H2S) as stoichiometric ... (HS? |
|
Table of Acids with Ka and pKa Values* CLAS - UC Santa Barbara
%20Base%20Strength/Table%20of%20Acids%20w%20Kas%20and%20pKas.pdf |
|
What Chemical interaction occurs when hydrogen peroxide is added to
H2SO4 HBr HI I Br HSO4 TsOH HNO3 HF O H O H O H H O H O H H O H O O O H NH H2CO3 HN 3 O H H H2S HCl Cl H F N NO3 SH TsO- HCO3 N O O-10-9-8-3 6-2 4-1 7-1 3 4 7 4 8 3 2 sulfuric acid hydroiodic acid hydrobromic acid |
|
Hydrogen Sulfide in Aqueous Solvents - NIST
H2S with T/K corresponding to this equation and the values of x H2S from experimental measurements made since 1930 are plotted in fig 1 Values in the range 273 2 to 283 2 are better fitted by the equation: in x H2S =-14 761 + 2507 2/(T/K) 6x H2S = ± 0 000030 The average value of x H2S for a partial pressure of 1 013 bar at 298 15 K is 0 |
|
4 Acid Base Chemistry - University of Texas at Austin
H2S - HÆ+ HS K= 10 1 -7 1 pK = 7 1 (HS) 2 The fundamental relationship with weak acids is that the extent of of the reaction or the amount of acid that dissociates is directly related to the pH When pH < pK little acid dissociates As pH increases more acid must dissociate to maintain equilibrium |
|
PKa Table: Effect of electronegativity and resonance
Acid pKaConjugate Base HCl-7 Cl-2 CH3CH2OH HO-2 H2O H OH O O O OH O phenol phenoxide e g sodium phenoxide O Na+ HO-H HO- CH3CH2O H H C H3 CH 2O eth anol CH3HO- ethnoxid H2N-H a mmonia H2N- aide e g sodium ethoxide |
|
Searches related to pka of h2s filetype:pdf
pKa values of the binary hydrides of the nonmetals HI-9 3 H 2Te 2 6 HBr-8 7 H 2Se 3 7 AsH 3 23 GeH 4 25 HCl-6 3 H 2S 6 89 PH 3 27 SiH 4 ~ 35 HF 3 15 H 2O 15 74 NH 3 39 CH 4 ~ 44 Acidic and Basic Oxides The oxides that one uses to form acids and bases in aqueous solution often have reactivity that re?ects their acidic or basic character |
What is the pKa of H2O2?
- As André Macherius mentioned H2O2 dissociates forming HO2- and H+. The pKa is 11.75 according to Wikipedia. This is of some importance as the base form is more reactive e.g. in several of the hydrolysis reactions. For this reason commercial H2O2 solutions are usually added some acid (phosphoric) to increase the stability.
What are the pKa values?
- pKa Data Compiled by R. Williams page-1 pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4, 8 Pyrazine 26 Aliphatic 4, 8 Aromatic 7, 8 Quinoline 27 Phenols 9 Quinazoline 27 Alcohols and oxygen acids 10, 11 Quinoxaline 27 Amino Acids 12 Special Nitrogen Compounds 28 Peptides 13 Hydroxylamines 28
How many pKa's does sulfuric acid have?
- If you look at a molecule like Sulfuric acid, H2SO4, a molecule with more than one acidic hydrogen, it's going to have two pKa's, one for each proton. In these cases, you can avoid confusion if you talk about the pKa of the proton instead of the pKa of the molecule.
Is the pKa of an acid dependent on the base?
- The pKa of the acid isn't dependent on the pKb or strength of the base. The equilibrium constant, Keq, which roughly measures how how readily the reactants will become the products, will. Think of it like two children who want the same toy. Suzy has the toy. Billy wants to take it.
|
Examen du 9 janvier 2007 2 heures - lutesupmcfr
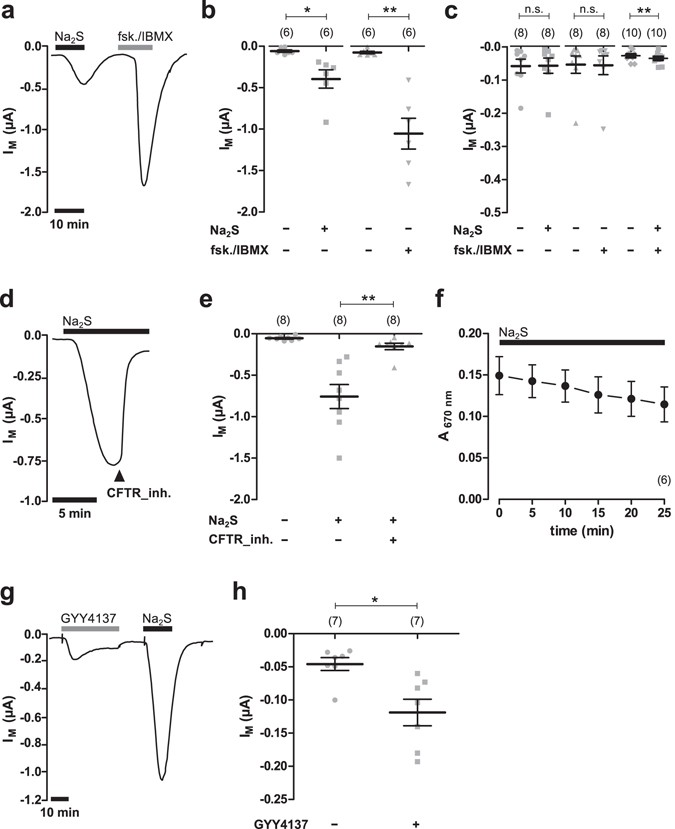
solution soit égal au pKa du couple acido-basique NH4 + /NH3 ? H2S est un diacide faible dont les constantes d'acidité sont : pKA(H2S/HS - ) = 7,0 et pKA( |
|
Le diagramme de prédominance dun diacide
Pour chaque couple acido-basique : H2S/HS- et HS-/S2-, on trace α0 = f pH ( ) et α1 = f pH ( ) (point d'inflexion à pH=pKa) Toute verticale permet d'obtenir les |
|
Calcul du pH dun mélange dacides et de bases - Free
Calculer la constante d'équilibre de chacune des réaction envisagées Données : • pKA (CH3CO2H / CH3COO-) = 4,8 ; • pour H2S :pKA1 = 7,0 ; pKA2 = 13,0 ; |
|
H2S RESUMEE TOXICO - Association Toxicologie-Chimie
Dans l'eau, H2S à pH neutre (pH 7) se dissocie à égalité en anion hydrogénosulfure (H-S-, pKa=7,04) et dianion sulfure (S2-, pKa=11,96) et peut s' oxyder en |
|
Table of Acids with Ka and pKa Values* CLAS - UCSB CLAS
pKa Acid Strength Conjugate Base Strength Hydroiodic HI I- Hydrobromic 8/9 3 x 10-8 7 10/7 03 Hydrosulfuric H2S HS- 1 0 x 10-7/9 1 x 10- 8 7/7 04 |
|
Chimie - e3a-Polytech
Le sulfure d'hydrogène H2S est à température ambiante un gaz très soluble dans l'eau et à l'odeur pKa (H2A / HA–) = 3,6 et pKa (HA– / A2–) = 10,5 À titre de |
|
Université Paris Descartes Outils chimiques pour létude - Thesesfr
Ce premier pKa proche du pH biologique est un avantage pratique, car on peut remplacer H2S gazeux par sa forme saline NaSH plus facile à manipuler, dès lors |


![PDF] A novel and cost-effective hydrogen sulfide removal PDF] A novel and cost-effective hydrogen sulfide removal](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/asset/b49607d6-f3a0-4fd7-87f0-3db496e9816d/gr1.jpg)


![PDF] Caustic Scrubber Designs for H 2 S Removal from Refinery Gas PDF] Caustic Scrubber Designs for H 2 S Removal from Refinery Gas](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid\u003d402\u0026t\u003dl)

![pKa Tables - [PDF Document] pKa Tables - [PDF Document]](https://cyberleninka.org/viewer_images/1226370/f/1.png)









![Fluorescent Probes for Hydrogen Sulfide Detection - [PDF Document] Fluorescent Probes for Hydrogen Sulfide Detection - [PDF Document]](https://img.yumpu.com/16604369/1/500x640/tables-pk-pks-log-kf-et-potentielpdf-jy-magna.jpg)














![Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus](https://ars.els-cdn.com/content/image/1-s2.0-S2090123220300606-gr2.jpg)

![PDF] Hydrogen sulfide in cell signaling signal transduction PDF] Hydrogen sulfide in cell signaling signal transduction](https://www.dovepress.com/cr_data/article_fulltext/s249000/249605/img/DMSO_A_249605_O_F0004g.jpg)
![Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus](https://upload.wikimedia.org/wikipedia/commons/f/f9/AmineTreating.png)





![Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus](https://faseb.onlinelibrary.wiley.com/cms/asset/d0a826cf-09b8-4137-b2bc-6b397557622c/fsb2033012005-fig-0005-m.jpg)










![pKa Tables - [PDF Document] pKa Tables - [PDF Document]](https://media.springernature.com/lw685/springer-static/image/art%3A10.1186%2Fs12967-018-1753-7/MediaObjects/12967_2018_1753_Fig1_HTML.png)


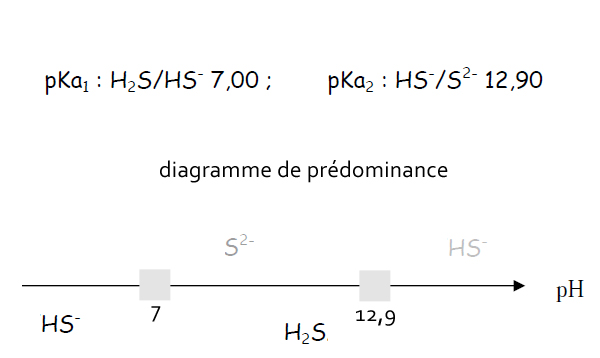
![PDF] Epithelial Electrolyte Transport Physiology and the PDF] Epithelial Electrolyte Transport Physiology and the](https://europepmc.org/articles/PMC6818192/bin/ijmsv16p1386g003.jpg)


![PDF] Study of Various Aqueous and Non-Aqueous Amine Blends for PDF] Study of Various Aqueous and Non-Aqueous Amine Blends for](https://ars.els-cdn.com/content/image/1-s2.0-S2090123220300606-gr1.jpg)


![Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus Full text] Hydrogen Sulfide Adipose Tissue and Diabetes Mellitus](https://aiche.onlinelibrary.wiley.com/cms/asset/cae711f0-ff41-4b88-9794-4fc57a04542a/ep13304-fig-0001-m.jpg)