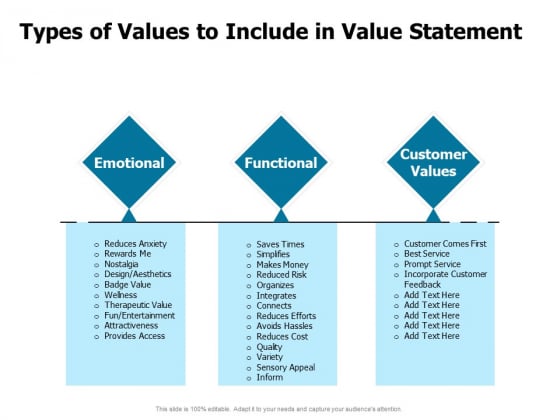
pka values
|
Table of Acids with Ka and pKa Values* CLAS
%20Base%20Strength/Table%20of%20Acids%20w%20Kas%20and%20pKas.pdf |
|
Determination of pKa values of active pharmaceutical ingredients
In a potentiometric titration a sample is titrated with acid or base using a pH electrode to monitor the course of titration. The pKa value is calculated from |
|
PKa Values of Guanine in Water: Density Functional Theory
2 The ionization constants (pKa values) of the normal DNA bases are roughly. 2-3 pH units from physiological pH predicting that the ionized forms might exist |
|
PKa Data Compiled by R. Williams pKa Values INDEX Inorganic 2
Inorganic. 2. Phenazine. 24. Phosphates. 3. Pyridine. 25. Carboxylic acids. 4 8. Pyrazine. 26. Aliphatic. 4 |
|
Biphasic pKa Values
14 janv. 2019 Abstract: A novel approach – termed here as biphasic pKa values – is presented for quantifying the acidity/basicity of lipophilic compounds. |
|
Comparison of Predicted pKa Values for Some Amino-Acids
27 nov. 2018 The ACD/Labs pKa prediction method [11] enables to calculate single pKa values for all possible dissociation centers when the rest of the ... |
|
Measurement of Nucleobase p K a Values in Model
13 févr. 2004 Measurement of Nucleobase pKa Values in Model. Mononucleotides Shows RNA-RNA Duplexes To Be More. Stable than DNA-DNA Duplexes. |
|
Comparison of Nine Programs Predicting p K a Values of
4 déc. 2009 Knowledge of the possible ionization states of a pharmaceutical substance embodied in the pKa values. (logarithm of the acid dissociation ... |
|
Table of Acids with Ka and pKa Values* CLAS - UC Santa Barbara
%20Base%20Strength/Table%20of%20Acids%20w%20Kas%20and%20pKas.pdf |
|
PKa Values INDEX - Organic Chemistry Data
pKa Data Compiled by R Williams pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4 8 Pyrazine 26 Aliphatic 4 8 Aromatic 7 8 Quinoline 27 Phenols 9 Quinazoline 27 Alcohols and oxygen acids 10 11 Quinoxaline 27 Amino Acids 12 Special Nitrogen Compounds 28 Peptides 13 Hydroxylamines 28 Nitrogen Compounds 14 |
|
Functional group pka - Indiana University Bloomington
3 Carboxylic acid pKa = 4?5 4 Ammonium ion pKa = 9?10 5 Phenol pKa = 10 6 Thiol pKa = 10 7 Alcohol pKa = 16?18 8 Water pKa = 15 7 9 Amide pKa = 18 10 Alpha proton of ketone/aldehyde pKa = 20 11 Alpha proton of ester pKa = 25 12 Terminal alkyne pKa = 25 13 |
|
Acids and Bases - University of Washington
The pKa - a single scale for reporting and comparing the ionization capacity of acids and bases Since every base has its conjugate acid it is possible to compare all acids and bases on a single scale The scale commonly used is the acidity constant of the conjugate acid of any conjugate pair expressed in logarithmic units |
|
Searches related to pka values filetype:pdf
values for molecules vs ions A molecule which loses a proton ionizes: HA + H 2 O ? H 3 O+ + A-A low pK a value therefore denotes good aqueous solubility An ion which loses a proton however de-ionizes: HB+ + H 2 O ? H 3 O + + B and so a high pK a value denotes good aqueous solubility There is no reason why pK a values |
How do you calculate pKa value?
- pKa = -log[Ka] Acid dissociation constants, or pKa values, are essential for understanding many fundamental reactions in chemistry. These values reveal the deprotonation state of a molecule in a particular solvent. There is great interest in using theoretical methods to calculate the pKa values for many different types of molecules. Calulation of pKa. Let us consider a weak acid HA which ionizes in the aqueous solution as: HA + H 2 O ? H 3 O + (aq) + A – (aq)
How to determine pKa values?
- Find the dissociation constant with the formula Ka = (H+)²/ ( (HA) – (H+)), where Ka is the dissociation constant, and (HA) is the concentration of the acid before dissociation. In chemistry, the pKa value is really a way of measuring acidity. It is the negative logarithm from the equivalence constant once the solvent is water.
Which has the lower value of PKA?
- The pKa value of 2-chloroacetic acid is 2.8 and the pKa value of 2-fluoroacetic acid is 2.6. Thus, the 2-fluoroacetic acid has lower pKa value. So, the 2-fluoroacetic acid is strong acid and 2-chloroacetic acid is a weak acid. Weak acid has a strong conjugate base.
Does lower pKa value mean stronger acid?
- pKa is the negative log of the acid dissociation constant or Ka value. A lower pKa value indicates a stronger acid. That is, the lower value indicates the acid more fully dissociates in water. In addition to using pKa to gauge the strength of an acid, it may be used to select buffers.
|
Table of Acids with Ka and pKa Values* CLAS - UCSB CLAS
Table of Acids with Ka and pKa Values* CLAS Acid HA A- Ka pKa Acid Strength Conjugate Base Strength Hydroiodic HI I- Hydrobromic HBr Br- |
|
PKa Data Compiled by R Williams pKa Values INDEX Inorganic 2
pKa Data Compiled by R Williams pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4, 8 Pyrazine 26 Aliphatic |
|
Discussion of pH and pKa Values
You must know the pKa value for the conjugate acid you will be using However, the above equation has additional information that you should understand Most |
|
PKa Data Compiled by R Williams page-1 pKa Values INDEX
pKa Data Compiled by R Williams page-1 pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4, 8 Pyrazine 26 |
|
PH and Buffers - CSUN
Weak Acids and pKa Similar to pH, the value of Ka can also be represented as pKa pKa is a constant for each conjugate acid and its conjugate base pair |
|
Determination of pKa values of active pharmaceutical ingredients
Moreover, pKa values can also be predicted by com- putational methods on the basis of molecular structure 2 pKa determination 2 1 Potentiometric titration In |
|
Pka basescdx
pKa 35 7 lithium diisopropyl amide LDA pKa 35 amide H3C OH pKa 15-16 Note; The pKa values associated with bases is normally meant to refer to the true |

















![PDF] Moral Values: Moral Values v 2 (Ethics) PDF] Moral Values: Moral Values v 2 (Ethics)](https://0.academia-photos.com/attachment_thumbnails/33134253/mini_magick20180819-25196-1o86smq.png?1534711075)







![PDF] LIBRARY The Real Madrid Way How Values Created the Most Succ PDF] LIBRARY The Real Madrid Way How Values Created the Most Succ](https://joheryo.weebly.com/uploads/1/2/5/7/125715312/415418081.png)