 1-10-times-tables-chart.pdf
1-10-times-tables-chart.pdf
6 x 1 = 6. 6 x 2 = 12. 6 x 3 = 18. 6 x 4 = 24. 6 x 5 = 30. 6 x 6 = 36. 6 x 7 = 42. 6 x 8 = 48. 6 x 9 = 54. 6 x 10 = 60. 6 x 11 = 66. 6 x 12 = 72. 7 x 1 = 7.
 Multiplication Tables and Charts
Multiplication Tables and Charts
Multiplication Tables and Charts. 1 x 1 = 1. 1 x 2 = 2. 1 x 3 = 3. 1 x 4 = 4. 1 x 5 = 5. 1 x 6 = 6. 1 x 7 = 7. 1 x 8 = 8. 1 x 9 = 9. 1 x 10 = 10. 1 x 11 = 11. 1
 Multiplication Tables
Multiplication Tables
1 x 0 = 0. 1 x 1 = 1. 1 x 2 = 2. 1 x 3 = 3. 1 x 4 = 4. 1 x 5 = 5. 1 x 6 = 6. 1 x 7 = 7. 1 x 8 = 8. 1 x 9 = 9. 1 x 10 = 10. 1 x 11 = 11. 1 x 12 = 12.
 List MF19
List MF19
variance 1 then
 MULTIPLICATION TABLES 1 TO 10 (MATH KIDS AND CHAOS)
MULTIPLICATION TABLES 1 TO 10 (MATH KIDS AND CHAOS)
Thanks for downloading this math printable. I really hope you find it useful! Please be aware that this product is for personal use only and is protected by.
 STATISTICS
STATISTICS
Let us now recall the various ways of presenting the data through some examples. Example 1 : Consider the marks obtained by 10 students in a mathematics test as.
 Lab manual XII (setting on 25-06-09) 1_10.pmd
Lab manual XII (setting on 25-06-09) 1_10.pmd
Mathematics. 111. OBSERVATION. The image of point N. 1 in the mirror (the line y log 10 e e y x. = 10 log. 1 log log 10 e e x x. ⇒. = = 0.434294 (using log ...
 Physical Science: Tables & Formulas
Physical Science: Tables & Formulas
Page 1. Page 1 of 10. Physical Science: Tables & Formulas. SI Base Units. Base Quantity. Unit Name. Unit Symbol. Amount of substance mole. Mol. Electric current.
 kelm401.pdf
kelm401.pdf
1 8765432. Fig. 5. Domain. Codomain. Page 23. Mathematics. 23. DEMONSTRATION. 1 10. 5. Prepare the table of ordered pairs for sin x sin 2x
 PISA RELEASED ITEMS - MATHEMATICS
PISA RELEASED ITEMS - MATHEMATICS
Code 1: 10 AM or 10:00. No credit. Code 0: Other responses. Code 9: Missing Code 9: Missing. Page 78. ReleasedPISAItems_Maths.doc. Page 78. M521: Table Tennis ...
 List MF19
List MF19
List MF19. List of formulae and statistical tables. Cambridge International AS & A Level. Mathematics (9709) and Further Mathematics (9231).
 PISA RELEASED ITEMS - MATHEMATICS
PISA RELEASED ITEMS - MATHEMATICS
Code 1: 168.3 cm (unit already given). No credit. Code 0: Other responses. 10 11 12 13 14 15 16 17 18 19 20. 190.
 Physical Science: Tables & Formulas
Physical Science: Tables & Formulas
Page 1 of 10. Physical Science: Tables & Formulas. SI Base Units. Base Quantity. Unit Name. Unit Symbol. Amount of substance mole. Mol. Electric current.
 kelm401.pdf
kelm401.pdf
mathematics the role of laboratory is helpful in understanding the an opportunity to the students to do certain calculations using tables calculators
 Data Handling Data Handling Data Handling Data Handling
Data Handling Data Handling Data Handling Data Handling
MATHEMATICS. 192. EXERCISE 9.1. 1. In a Mathematics test the following marks were obtained by 40 students. Arrange these marks in a table using tally marks
 STATISTICS
STATISTICS
Example 1 : Consider the marks obtained by 10 students in a mathematics test as 1. Total. 30. Table 14.1 is called an ungrouped frequency distribution ...
 Mathematics programmes of study: key stages 1 and 2 - GOV.UK
Mathematics programmes of study: key stages 1 and 2 - GOV.UK
The principal focus of mathematics teaching in key stage 1 is to ensure that pupils 2 5 and 10 multiplication tables and connect them to each other.
 Lab manual XII (setting on 25-06-09) 1_10.pmd
Lab manual XII (setting on 25-06-09) 1_10.pmd
between the various branches of mathematics (g) correct Laboratory Manual. 3. l ... 1. In Fig. 1
 PISA 2018 Results: Combined Executive Summaries
PISA 2018 Results: Combined Executive Summaries
In 20 education systems including those of 15 OECD countries
 F9 formulae sheet and maths tables
F9 formulae sheet and maths tables
F9 formulae sheet and maths tables Present value of 1 i.e. (1 + r)–n ... 10%. 1. 0·990 0·980 0·971 0·962 0·952 0·943 0·935 0·926 0·917 0·909. 1.
Page 1 of 10
Physical Science: Tables & Formulas
SI Base Units
Base Quantity Unit Name Unit Symbol
Amount of substance mole Mol
Electric current ampere A
Length meter M
Luminous intensity candela Cd
Mass kilogram Kg
Time second S
Temperature Kelvin K
SI Derived Units
Derived Quantity Name (Symbol) Expression in terms of other SI unitsExpression in terms
of SI base unitsArea Square meter (m2)
Volume Cubic meter (m3)
Speed/velocity Meter per second (m/s)
Acceleration Meter per second squared (m/s2)
Frequency Hertz (Hz) s-1
Force Newton (N) m . kg . s-2
Pressure, stress Pascal (Pa) N.m2 m-1 . kg . s-2
Energy, work, quantity of heat Joule (J) N. m m2 . kg . s-2Power Watt (W) J/s m2 . kg . s-3
Electric charge Coulomb (C) -- s . A
Electric potential difference Volt (V) W/A m2·kg·s-3·A-1 Electric resistance ȍ V/A m2·kg·s-3·A-2 Prefixes used to designate multiples of a base unit Prefix Symbol Meaning Multiple of base unit Scientific Notation tera T trillion 1, 000, 000, 000, 000 1012 giga G billion 1, 000, 000, 000 109 mega M Million 1, 000, 000 106 kilo k Thousand 1, 000 103 centi c One hundredth 1/100 or .01 10-2 milli m One thousandth 1/1000 or .001 10-3 micro u One millionth 1/1000000 or .000001 10-6 Nano n One billionth 1/1000000000 or .000000001 10-9 pico p One trillionth 1/1000000000000 or.000000000001 10-12In general, when converting from base units (m, l, g, etc) or derived units (m2,m3, m/s, Hz, N, J, V, etc) to a
multiple greater (kilo, mega, giga, or tera) than the base or derived unit- then divide by the factor. For
example: 10m = 10/1000km = 1/100 km = .01km.Page 2 of 10
When converting from base units or derived units to a multiple smaller (centi, milli, micro, nano) than the
base or derived unit- then multiply by the factor. For example: 10m = 10 x 100cm = 1000cm.Subatomic Particles
Particle Charge Mass Location
Proton +1 1 nucleus
Neutron 0 1 nucleus
Electron -1 0 Outside the nucleus
Common Cations
Ion Name (symbol) Ion Charge
Lithium (Li) 1+
Sodium (Na) 1+
Potassium (K) 1+
Rubidium (Rb) 1+
Cesium (Cs) 1+
Beryllium (Be) 2+
Magnesium (Mg) 2+
Calcium (Ca) 2+
Strontium (Sr) 2+
Barium (Ba) 2+
Aluminum (Al) 3+
Common Anions
Element Name (symbol) Ion Name (symbol) Ion ChargeFluorine Fluoride 1-
Chlorine Chloride 1-
Bromine Bromide 1-
Iodine Iodide 1-
Oxygen Oxide 2-
Sulfur Sulfide 2-
Nitrogen Nitride 3-
Common Polyatomic Ions
Ion Name Ion Formula Ion Name Ion Formula
Carbonate CO32- Nitrite NO2-
Chlorate ClO3- Phosphate PO43-
Cyanide CN- Phosphite PO33-
Hydroxide OH- Sulfate SO42-
Nitrate NO3- Sulfite SO32-
Page 3 of 10
Prefixes for Naming Covalent Compounds
Number of Atoms Prefix Number of Atoms Prefix
1 Mono 6 Hexa
2 Di 7 Hepta
3 Tri 8 Octa
4 Tetra 9 Nona
5 penta 10 deca
Types of Chemical Reactions
Type of reaction Generalized formula Specific Example Combustion HC + O2 AE H2O + CO2 2C2H6 + 7O2 AE 6H2O + 4CO2Synthesis A + B AE AB 2Na + Cl2 AE 2NaCl
Decomposition AB AE A + B 2H2O AE 2H2 + O2 Single Replacement A + BC AE AC + B 2Al + 3CuCl2 AE 3Cu + 2AlCl3 Double Replacement AX + BY AE AY + BX Pb(NO3)2 + K2CrO4 AE PbCrO4 + 2KNO3Principle)
Condition Effect
Temperature Increasing temperature favors the reaction that absorbs energy (endothermic) Pressure Increasing pressure favors the reaction that produces less gas. Concentration Increasing conc. of one substance favors reaction that produces less of that substanceCommon Acids
Acid Formula Strength
Hydrochloric (muriatic) acid HCl strong
Nitric acid HNO3 strong
Sulfuric acid H2SO4 strong
Acetic acid CH3COOH weak
Citric acid C6H8O7 weak
Formic HCOOH weak
Common Bases
Base Formula Strength
Potassium hydroxide (potash) KOH strong
Sodium hydroxide (lye) NaOH strong
Calcium hydroxide (lime) Ca(OH)2 strong
ammonia NH3 weakPage 4 of 10
pH scaleStrong acids Å more acidic Å weak acids Neutral Weak bases AE More basic AE strong bases
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14
Types of Nuclear Radiation
Radiation Type Symbol Charge Nuclear Equation
Alpha particle 2 4He +2 89 225Ac AE 87 221Fr + 2 4HeBeta particle -1 0e -1 614C AE 7 14N + -1 0e
Gamma Ȗ 0 n/a
Equations
Density = mass ÷ volume (D = m/v) Units: g/cm3 or g/mL Rearranged: mass = Density x Volume Units: grams orVolume = mass ÷ density Units: cm3 or mL
Moles = mass (grams) x Molar Mass (grams / mol) Molar Mass = atomic mass in grams Energy = mass x (speed of light)2 E = mc2 Units: joules Speed = distance ÷ time v = d ÷ t Units: meters / second Rearranged: distance = speed x time Units: meters time = distance ÷ speed Units: seconds Momentum = mass x velocity p = m x v Units: kg . m/sAcceleration = (final velocity - initial velocity) ÷ time a = ǻ÷ t Units: meters / (second)2
Rearrangedǻ
ǻ Units: seconds
Force = mass x acceleration F = m x a Units: kg . m/s2 or Newtons (N) Rearranged: mass = Force ÷ acceleration Units: g or kg acceleration = Force ÷ mass Units: meters / (second)2Page 5 of 10
Weight = mass x gravity (9.8 m/s2 ) Units: kg . m/s2 or Newtons (N) Work = Force x distance W = F x d Units: Joules (J) Rearranged: Force = Work ÷ distance Units: Newtons distance = Work ÷ Force Units: meters Power = Work ÷ time P = W ÷ t Units: J/s or Watts (W) Rearranged: Work = Power x time Units: Joules (J) time = Work ÷ Power Units: seconds (s) Mechanical Advantage = Output Force ÷ Input Force (Resistance Force ÷ Effort Force) orMechanical Advantage = Input Distance ÷ Output Distance (Effort Distance ÷ Resistance Distance)
Gravitational Potential Energy = mass x gravity (9.8 m/s2) x height GPE = m x g x h Units:Joules
Rearranged: m = GPE ÷ (g . h) h = GPE ÷ (m . g) Kinetic Energy = ½ mass x (velocity)2 KE = .5 mv2 Units: JoulesRearranged: m = 2KE ÷ v2 v =
Efficiency of a Machine = (Useful Work Output ÷ Work Input) x 100Temperature Conversions
Celsius-Fahrenheit Conversion:
Fahrenheit temperature = (1.8 x Celsius temperature) + 32.00 F = 1.8 (C) + 320 Celsius temperature = (Fahrenheit temperature 32) ÷ 1.8 C = (F 32) ÷ 1.8Celsius-Kelvin Conversion:
Kelvin = Celsius + 273 Celsius = Kelvin -273
Page 6 of 10
Specific Heat Equation
Energy = mass x Specific Heat Value x change in temperature E = m . c . ǻ Units: Joules Rearranged: mass = Energy ÷ (ǻ Units: kg ǻc x mass ) Units: K or 0CWave Speed Equation
Wave Speed = frequency x wavelength Ȝ Units: m/s Rearranged: Frequency = Wave Speed ÷ wavelength Ȝ Units: Hertz Wavelength = Wave Speed ÷ frequency Ȝ Units: meters / second Speed of light (in a vacuum) = 3.0 x 108 m/s (300,000,000 m/s) Speed of Sound (in air at 25 0C) = 346 m/s Speed of Sound (in water at 25 0C) = 1490 m/sSpeed of Sound (in iron at 25 0C) = 5000 m/s
Law Equation
Current = Voltage ÷ Resistance I = V / R Units: Amperes (A) Rearranged: Voltage = Current x Resistance V = I x R Units: Volts (V)Resistance = Voltage ÷ Current R = V / I ȍ
Electric Power Equation
Power = Current x Voltage P = I x V Units: watts (W) or Kilowatts (kW)Variations: P = I2 x R P = V2 / R
Rearranged: Voltage = Power ÷ Current V = P x I Units: Volts (V) Current = Power ÷ Voltage I = P ÷ V Units: Amperes (A)Page 7 of 10
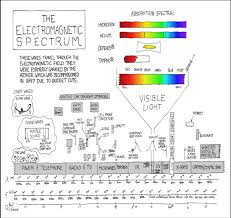
Electromagnetic Spectrum: Relates the energy, frequency and wavelength of various types ofelectromagnetic waves (radio, TV, micro, infrared, visible, ultraviolet, X-ray, and gamma). As energy and
frequency increase the wavelength decreases.Page 8 of 10
Page 9 of 10
AM radio - 535 kilohertz to 1.7 megahertz
Short wave radio - bands from 5.9 megahertz to 26.1 megahertz Citizens band (CB) radio - 26.96 megahertz to 27.41 megahertz Television stations - 54 to 88 megahertz for channels 2 through 6FM radio - 88 megahertz to 108 megahertz
Television stations - 174 to 220 megahertz for channels 7 through 13 Garage door openers, alarm systems, etc. - Around 40 megahertz Standard cordless phones: Bands from 40 to 50 megahertzBaby monitors: 49 megahertz
Radio controlled airplanes: Around 72 megahertz, which is different from...Radio controlled cars: Around 75 megahertz
Wildlife tracking collars: 215 to 220 megahertz
MIR space station: 145 megahertz and 437 megahertzCell phones: 824 to 849 megahertz
New 900-MHz cordless phones: Obviously around 900 megahertz!Air traffic control radar: 960 to 1,215 megahertz
Page 10 of 10
Global Positioning System: 1,227 and 1,575 megahertz Deep space radio communications: 2290 megahertz to 2300 megahertzquotesdbs_dbs11.pdfusesText_17[PDF] 1-es 2016 massachusetts
[PDF] 1. liste des capacités
[PDF] 1.0 bac level
[PDF] 1.3.3 traitement des formalités administratives
[PDF] 1.6 bac
[PDF] 1.9 bac
[PDF] 10 cas de marketing pdf
[PDF] 10 diy sciences experimentales
[PDF] 10 problemas de caida libre resueltos
[PDF] 10 sınıf palme pdf
[PDF] 10-32 unf en mm
[PDF] 10.s?n?f fizik konu anlat?m? pdf indir esen
[PDF] 100 años de lucha libre en mexico pdf
[PDF] 100 common errors in english pdf
